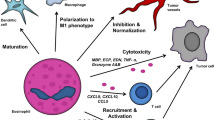
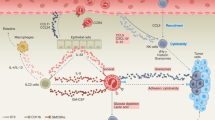
Abstract
Eosinophils have long been associated with allergy and parasitic infections. Today, they are considered as multifunctional leukocytes, which participate both in innate and adaptive immune response though the expression of various receptors and mediators. Although the tumor-associated eosinophilia is observed for a long time in many hematological and solid malignancies, with a generally good prognosis value, there is a lack of knowledge on the different mechanisms involved in this phenomenon. Moreover, the recent discovery in human eosinophils of different receptors and mediators, shared with lymphocytes and involved in anti-tumor defense, suggests that eosinophils can play a role in anti-tumoral immunity. We review in the present paper the current knowledge on epidemiology, recruitment, and mechanisms involved in the response of eosinophils toward tumors.


Similar content being viewed by others
Abbreviations
- DAMP:
-
Damage-associated molecular pattern molecule
- ECP:
-
Eosinophil cationic protein
- EDN:
-
Eosinophil-derived neurotoxin
- EPO:
-
Eosinophil peroxidase
- HL:
-
Hodgkin’s Lymphoma
- HMGB1:
-
High-mobility group box 1
- IL:
-
Interleukin
- MBP:
-
Major basic protein
- RAGE:
-
Receptor for advanced glycation end products
- ROS:
-
Reactive oxygen species
- SCC:
-
Squamous cell carcinoma
- TATE:
-
Tumor-associated tissue eosinophilia
References
Munitz A, Levi-Schaffer F (2004) Eosinophils: ‘new’ roles for ‘old’ cells. Allergy 59(3):268–275
Lowe D, Jorizzo J, Hutt MS (1981) Tumour-associated eosinophilia: a review. J Clin Pathol 34(12):1343–1348
Kita H (2011) Eosinophils: multifaceted biological properties and roles in health and disease. Immunol Rev 242(1):161–177
Driss V, Legrand F, Hermann E, Loiseau S, Guerardel Y, Kremer L, Adam E, Woerly G, Dombrowicz D, Capron M (2009) TLR2-dependent eosinophil interactions with mycobacteria: role of alpha-defensins. Blood 113(14):3235–3244
Legrand F, Driss V, Delbeke M, Loiseau S, Hermann E, Dombrowicz D, Capron M (2010) Human eosinophils exert TNF-alpha and granzyme A-mediated tumoricidal activity toward colon carcinoma cells. J Immunol 185(12):7443–7451
Legrand F, Driss V, Woerly G, Loiseau S, Hermann E, Fournie JJ, Heliot L, Mattot V, Soncin F, Gougeon ML, Dombrowicz D, Capron M (2009) A functional gammadeltaTCR/CD3 complex distinct from gammadeltaT cells is expressed by human eosinophils. PLoS ONE 4(6):e5926
Munitz A, Bachelet I, Fraenkel S, Katz G, Mandelboim O, Simon HU, Moretta L, Colonna M, Levi-Schaffer F (2005) 2B4 (CD244) is expressed and functional on human eosinophils. J Immunol 174(1):110–118
Rothenberg ME, Hogan SP (2006) The eosinophil. Annu Rev Immunol 24:147–174
Gleich GJ (2000) Mechanisms of eosinophil-associated inflammation. J Allergy Clin Immunol 105(4):651–663
Hogan SP, Rosenberg HF, Moqbel R, Phipps S, Foster PS, Lacy P, Kay AB, Rothenberg ME (2008) Eosinophils: biological properties and role in health and disease. Clin Exp Allergy 38(5):709–750
Abu-Ghazaleh RI, Gleich GJ, Prendergast FG (1992) Interaction of eosinophil granule major basic protein with synthetic lipid bilayers: a mechanism for toxicity. J Membr Biol 128(2):153–164
Lucey DR, Nicholson-Weller A, Weller PF (1989) Mature human eosinophils have the capacity to express HLA-DR. Proc Natl Acad Sci USA 86(4):1348–1351
Spencer LA, Weller PF (2010) Eosinophils and Th2 immunity: contemporary insights. Immunol Cell Biol 88(3):250–256
Yang D, Chen Q, Su SB, Zhang P, Kurosaka K, Caspi RR, Michalek SM, Rosenberg HF, Zhang N, Oppenheim JJ (2008) Eosinophil-derived neurotoxin acts as an alarmin to activate the TLR2-MyD88 signal pathway in dendritic cells and enhances Th2 immune responses. J Exp Med 205(1):79–90
Lee JJ, Jacobsen EA, McGarry MP, Schleimer RP, Lee NA (2010) Eosinophils in health and disease: the LIAR hypothesis. Clin Exp Allergy 40(4):563–575
Pretlow TP, Keith EF, Cryar AK, Bartolucci AA, Pitts AM, Pretlow TG II, Kimball PM, Boohaker EA (1983) Eosinophil infiltration of human colonic carcinomas as a prognostic indicator. Cancer Res 43(6):2997–3000
Fernandez-Acenero MJ, Galindo-Gallego M, Sanz J, Aljama A (2000) Prognostic influence of tumor-associated eosinophilic infiltrate in colorectal carcinoma. Cancer 88(7):1544–1548
Dorta RG, Landman G, Kowalski LP, Lauris JR, Latorre MR, Oliveira DT (2002) Tumour-associated tissue eosinophilia as a prognostic factor in oral squamous cell carcinomas. Histopathology 41(2):152–157
Ishibashi S, Ohashi Y, Suzuki T, Miyazaki S, Moriya T, Satomi S, Sasano H (2006) Tumor-associated tissue eosinophilia in human esophageal squamous cell carcinoma. Anticancer Res 26(2B):1419–1424
Fujii M, Yamashita T, Ishiguro R, Tashiro M, Kameyama K (2002) Significance of epidermal growth factor receptor and tumor associated tissue eosinophilia in the prognosis of patients with nasopharyngeal carcinoma. Auris Nasus Larynx 29(2):175–181
Ono Y, Ozawa M, Tamura Y, Suzuki T, Suzuki K, Kurokawa K, Fukabori Y, Yamanaka H (2002) Tumor-associated tissue eosinophilia of penile cancer. Int J Urol 9(2):82–87
Costello R, O’Callaghan T, Sebahoun G (2005) Eosinophils and antitumour response. Rev Med Interne 26(6):479–484
Luna-More S, Florez P, Ayala A, Diaz F, Santos A (1997) Neutral and acid mucins and eosinophil and argyrophil crystalloids in carcinoma and atypical adenomatous hyperplasia of the prostate. Pathol Res Pract 193(4):291–298
von Wasielewski R, Seth S, Franklin J, Fischer R, Hubner K, Hansmann ML, Diehl V, Georgii A (2000) Tissue eosinophilia correlates strongly with poor prognosis in nodular sclerosing Hodgkin’s disease, allowing for known prognostic factors. Blood 95(4):1207–1213
Pinto A, Aldinucci D, Gloghini A, Zagonel V, Degan M, Improta S, Juzbasic S, Todesco M, Perin V, Gattei V, Herrmann F, Gruss HJ, Carbone A (1996) Human eosinophils express functional CD30 ligand and stimulate proliferation of a Hodgkin’s disease cell line. Blood 88(9):3299–3305
Geisinger KR, Steffee CH, McGee RS, Woodruff RD, Buss DH (1998) The cytomorphologic features of sclerosing mucoepidermoid carcinoma of the thyroid gland with eosinophilia. Am J Clin Pathol 109(3):294–301
Fridlender ZG, Simon HU, Shalit M (2003) Metastatic carcinoma presenting with concomitant eosinophilia and thromboembolism. Am J Med Sci 326(2):98–101
Dibbert B, Daigle I, Braun D, Schranz C, Weber M, Blaser K, Zangemeister-Wittke U, Akbar AN, Simon HU (1998) Role for Bcl-xL in delayed eosinophil apoptosis mediated by granulocyte-macrophage colony-stimulating factor and interleukin-5. Blood 92(3):778–783
Teruya-Feldstein J, Jaffe ES, Burd PR, Kingma DW, Setsuda JE, Tosato G (1999) Differential chemokine expression in tissues involved by Hodgkin’s disease: direct correlation of eotaxin expression and tissue eosinophilia. Blood 93(8):2463–2470
Lorena SC, Oliveira DT, Dorta RG, Landman G, Kowalski LP (2003) Eotaxin expression in oral squamous cell carcinomas with and without tumour associated tissue eosinophilia. Oral Dis 9(6):279–283
Thielen C, Radermacher V, Trimeche M, Roufosse F, Goldman M, Boniver J, de Leval L (2008) TARC and IL-5 expression correlates with tissue eosinophilia in peripheral T-cell lymphomas. Leuk Res 32(9):1431–1438
Cormier SA, Taranova AG, Bedient C, Nguyen T, Protheroe C, Pero R, Dimina D, Ochkur SI, O’Neill K, Colbert D, Lombari TR, Constant S, McGarry MP, Lee JJ, Lee NA (2006) Pivotal advance: eosinophil infiltration of solid tumors is an early and persistent inflammatory host response. J Leukoc Biol 79(6):1131–1139
Stenfeldt AL, Wenneras C (2004) Danger signals derived from stressed and necrotic epithelial cells activate human eosinophils. Immunology 112(4):605–614
Lotfi R, Lee JJ, Lotze MT (2007) Eosinophilic granulocytes and damage-associated molecular pattern molecules (DAMPs): role in the inflammatory response within tumors. J Immunother 30(1):16–28
Ito N, DeMarco RA, Mailliard RB, Han J, Rabinowich H, Kalinski P, Stolz DB, Zeh HJ III, Lotze MT (2007) Cytolytic cells induce HMGB1 release from melanoma cell lines. J Leukoc Biol 81(1):75–83
Lotfi R, Herzog GI, DeMarco RA, Beer-Stolz D, Lee JJ, Rubartelli A, Schrezenmeier H, Lotze MT (2009) Eosinophils oxidize damage-associated molecular pattern molecules derived from stressed cells. J Immunol 183(8):5023–5031
Sun P, Ben Q, Tu S, Dong W, Qi X, Wu Y (2011) Serum interleukin-33 levels in patients with gastric cancer. Dig Dis Sci 56(12):3596–3601
Ikutani M, Yanagibashi T, Ogasawara M, Tsuneyama K, Yamamoto S, Hattori Y, Kouro T, Itakura A, Nagai Y, Takaki S, Takatsu K (2011) Identification of innate IL-5-producing cells and their role in lung eosinophil regulation and antitumor immunity. J Immunol 188(2):703–713
Caruso RA, Parisi A, Quattrocchi E, Scardigno M, Branca G, Parisi C, Luciano R, Paparo D, Fedele F (2011) Ultrastructural descriptions of heterotypic aggregation between eosinophils and tumor cells in human gastric carcinomas. Ultrastruct Pathol 35(4):145–149
Huland E, Huland H (1992) Tumor-associated eosinophilia in interleukin-2-treated patients: evidence of toxic eosinophil degranulation on bladder cancer cells. J Cancer Res Clin Oncol 118(6):463–467
Simon HU, Plotz S, Simon D, Seitzer U, Braathen LR, Menz G, Straumann A, Dummer R, Levi-Schaffer F (2003) Interleukin-2 primes eosinophil degranulation in hypereosinophilia and Wells’ syndrome. Eur J Immunol 33(4):834–839
Sosman JA, Bartemes K, Offord KP, Kita H, Fisher SG, Kefer C, Ellis TA, Fisher RI, Higgins TJ, Gleich GJ (1995) Evidence for eosinophil activation in cancer patients receiving recombinant interleukin-4: effects of interleukin-4 alone and following interleukin-2 administration. Clin Cancer Res 1(8):805–812
Tepper RI, Coffman RL, Leder P (1992) An eosinophil-dependent mechanism for the antitumor effect of interleukin-4. Science 257(5069):548–551
Rivoltini L, Viggiano V, Spinazze S, Santoro A, Colombo MP, Takatsu K, Parmiani G (1993) In vitro anti-tumor activity of eosinophils from cancer patients treated with subcutaneous administration of interleukin 2. Role of interleukin 5. Int J Cancer 54(1):8–15
Benatar T, Cao MY, Lee Y, Lightfoot J, Feng N, Gu X, Lee V, Jin H, Wang M, Wright JA, Young AH (2010) IL-17E, a proinflammatory cytokine, has antitumor efficacy against several tumor types in vivo. Cancer Immunol Immunother 59(6):805–817
Mattes J, Hulett M, Xie W, Hogan S, Rothenberg ME, Foster P, Parish C (2003) Immunotherapy of cytotoxic T cell-resistant tumors by T helper 2 cells: an eotaxin and STAT6-dependent process. J Exp Med 197(3):387–393
Simson L, Ellyard JI, Dent LA, Matthaei KI, Rothenberg ME, Foster PS, Smyth MJ, Parish CR (2007) Regulation of carcinogenesis by IL-5 and CCL11: a potential role for eosinophils in tumor immune surveillance. J Immunol 178(7):4222–4229
Turner MC, Chen Y, Krewski D, Ghadirian P (2006) An overview of the association between allergy and cancer. Int J Cancer 118(12):3124–3132
Wang H, Diepgen TL (2005) Is atopy a protective or a risk factor for cancer? A review of epidemiological studies. Allergy 60(9):1098–1111
Jensen-Jarolim E, Achatz G, Turner MC, Karagiannis S, Legrand F, Capron M, Penichet ML, Rodriguez JA, Siccardi AG, Vangelista L, Riemer AB, Gould H (2008) AllergoOncology: the role of IgE-mediated allergy in cancer. Allergy 63(10):1255–1266
Tomassini M, Tsicopoulos A, Tai PC, Gruart V, Tonnel AB, Prin L, Capron A, Capron M (1991) Release of granule proteins by eosinophils from allergic and nonallergic patients with eosinophilia on immunoglobulin-dependent activation. J Allergy Clin Immunol 88(3 Pt 1):365–375
Gounni AS, Lamkhioued B, Ochiai K, Tanaka Y, Delaporte E, Capron A, Kinet JP, Capron M (1994) High-affinity IgE receptor on eosinophils is involved in defence against parasites. Nature 367(6459):183–186
Jonsson UB, Bystrom J, Stalenheim G, Venge P (2002) Polymorphism of the eosinophil cationic protein-gene is related to the expression of allergic symptoms. Clin Exp Allergy 32(7):1092–1095
Molin D (2004) Bystander cells and prognosis in Hodgkin lymphoma. Review based on a doctoral thesis. Ups J Med Sci 109(3):179–228
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is part of the Symposium in Writing: AllergoOncology: The Role of Th2 responses in cancer.
Rights and permissions
About this article
Cite this article
Gatault, S., Legrand, F., Delbeke, M. et al. Involvement of eosinophils in the anti-tumor response. Cancer Immunol Immunother 61, 1527–1534 (2012). https://doi.org/10.1007/s00262-012-1288-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1288-3