Abstract
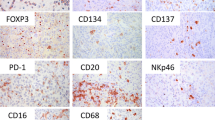
Studies on the prognostic importance of tumor-infiltrating lymphocytes have mainly focused on T cells, while little is known about the role of tumor-infiltrating B lymphocytes. We investigated the prevalence of CD20+ B cells by immunohistochemistry in primary melanoma samples of 106 patients and analyzed in relation to clinicopathological parameters and patients’ survival. The majority of samples contained a significant amount of B lymphocytes, predominantly dispersed in the stroma surrounding tumor deposits (mean peritumoral and intratumoral densities: 178.7 ± 156.1 vs. 4.9 ± 6.9 cells/mm2, respectively). B cells organized in follicle-like aggregates were also observed in 26% of the samples. B-cell density correlated with that of activated (CD25+ or OX40+) T lymphocytes. Infiltration by CD20+ lymphocytes did not correlate with tumor thickness, while the presence of B-cell aggregates was observed more frequently in thick melanomas. On the other hand, B-cell infiltration was more pronounced in nonmetastatic or lymph node metastatic tumors, compared to visceral metastatic ones. Accordingly, high number of these cells provided significant survival advantage (P = 0.0391 and P = 0.0136 for intra- and peritumoral infiltration, respectively). Furthermore, combination of peritumoral B-cell density with the number of activated T lymphocytes identified patient subgroups with different disease outcome, which was most favorable in the case of high density, while very poor in the case of low density of both cell types. Multivariate survival analysis identified tumor thickness and CD20+/OX40+ cell density combination as significant independent prognostic factors. Taken together, our results show correlation between low number of CD20+ B lymphocytes and melanoma progression, indicating a possible role of tumor-infiltrating B cells in antitumoral immune response. It was also reflected in better outcome of the disease since the density of B lymphocytes alone as well as in combination with that of activated T cells proved of prognostic importance in patients with malignant melanoma.



Similar content being viewed by others
References
Vesalainen S, Lipponen P, Talja M, Syrjanen K (1994) Histological grade, perineural infiltration, tumour-infiltrating lymphocytes and apoptosis as determinants of long-term prognosis in prostatic adenocarcinoma. Eur J Cancer 30A:1797–1803
Setälä LP, Kosma VM, Marin S, Lipponen PK, Eskelinen MJ, Syrjanen KJ, Alhava EM (1996) Prognostic factors in gastric cancer: the value of vascular invasion, mitotic rate and lymphoplasmacytic infiltration. Br J Cancer 74:766–772
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohué F, Bruneval P, Cugnenc P-H, Trajanoski Z, Fridman W-H, Pagès F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313:1960–1964
Al-Shibli KI, Donnem T, Al-Saad S, Persson M, Bremnes RM, Busund L-T (2008) Prognostic effect of epithelial and stromal lymphocyte infiltration in non-small cell lung cancer. Clin Cancer Res 14:5220–5227
Lee HE, Chae SW, Lee YJ, Kim MA, Lee HS, Lee BL, Kim WH (2008) Prognostic implications of type and density of tumour-infiltrating lymphocytes in gastric cancer. Br J Cancer 99:1704–1711
Naito Y, Saito K, Shiiba K, Ohuchi A, Saigenji K, Nagura H, Ohtani H (1998) CD8+ T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Cancer Res 58:3491–3494
Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, Kepner J, Odunsi T, Ritter G, Lele S, Chen Y-T, Ohtani H, Old LJ, Odunsi K (2005) Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci USA 102:18538–18543
Clark WH Jr, Elder DE, Guerry D IV, Braitman LE, Trock BJ, Schultz D, Synnestvedt M, Halpern AC (1989) Model predicting survival in stage I melanoma based on tumor progression. J Natl Cancer Inst 81:1893–1904
Clemente CG, Mihm MC Jr, Bufalino R, Zurrida S, Collini P, Cascinelli N (1996) Prognostic value of tumor infiltrating lymphocytes in the vertical growth phase of primary cutaneous melanoma. Cancer 77:1303–1310
Barnhill RL, Fine JA, Roush GC, Berwick M (1996) Predicting five-year outcome for patients with cutaneous melanoma in a population-based study. Cancer 78:427–432
Taylor RC, Patel A, Panageas KS, Busam KJ, Brady MS (2007) Tumor-infiltrating lymphocytes predict sentinel lymph node positivity in patients with cutaneous melanoma. J Clin Oncol 25:869–875
Hillen F, Beaten CIM, van de Winkel A, Creytens D, van der Schaft DWJ, Winnepenninckx V, Griffioen AW (2008) Leukocyte infiltration and tumor cell plasticity are parameters of aggressiveness in primary cutaneous melanoma. Cancer Immunol Immunother 57:97–106
Ridolfi RL, Rosen PP, Port A, Kinne D, Miké V (1977) Medullary carcinoma of the breast: a clinicopathologic study with 10 year follow-up. Cancer 40:1365–1385
Whelchel JC, Farah SE, McLean IW, Burnier MN (1993) Immunohistochemistry of infiltrating lymphocytes in uveal malignant melanoma. Invest Ophthalmol Vis Sci 34:2603–2606
Coronella-Wood JA, Hersh EM (2003) Naturally occurring B-cell responses to breast cancer. Cancer Immunol Immunother 52:715–738
Nelson BH (2010) CD20+ B cells: the other tumor-infiltrating lymphocytes. J Immunol 185:4977–4982
Hersey P, Murray E, Grace J, McCarthy WH (1985) Current research on immunopathology of melanoma: analysis of lymphocyte populations in relation to antigen expression and histological features of melanoma. Pathology 17:385–391
Ralfkaier E, Hou-Jensen K, Gatter KC, Drzewiecki KT, Mason DY (1987) Immunohistological analysis of the lymphoid infiltrate in cutaneous malignant melanomas. Virchow Arch A 410:355–361
Hussein MR, Elsers DAH, Fadel SA, Omar A-EM (2006) Immunohistological characterization of tumour infiltrating lymphocytes in melanocytic skin lesions. J Clin Pathol 59:316–324
Ladányi A, Somlai B, Gilde K, Fejös Z, Gaudi I, Tímár J (2004) T-cell activation marker expression on tumor-infiltrating lymphocytes as prognostic factor in cutaneous malignant melanoma. Clin Cancer Res 10:521–530
Ladányi A, Kiss J, Somlai B, Gilde K, Fejős Z, Mohos A, Gaudi I, Tímár J (2007) Density of DC-LAMP+ mature dendritic cells in combination with activated T lymphocytes infiltrating primary cutaneous melanoma is a strong independent prognostic factor. Cancer Immunol Immunother 56:1459–1469
Balch CM, Buzaid AC, Soong S-J, Atkins MB, Cascinelli N, Coit DG, Fleming ID, Gershenwald JE, Houghton A Jr, Kirkwood JM, McMasters KM, Mihm MF, Morton DL, Reintgen DS, Ross MI, Sober A, Thompson JA, Thompson JF (2001) Final version of the American Joint Committee on Cancer staging system for cutaneous melanoma. J Clin Oncol 19:3635–3648
Kiss J, Tímár J, Somlai B, Gilde K, Fejős Z, Gaudi I, Ladányi A (2007) Association of microvessel density with infiltrating cells in human cutaneous malignant melanoma. Pathol Oncol Res 13:21–31
Hansen MH, Nielsen H, Ditzel HJ (2001) The tumor-infiltrating B cell response in medullary breast cancer is oligoclonal and directed against the autoantigen actin exposed on the surface of apoptotic cancer cells. Proc Natl Acad Sci USA 98:12659–12664
O’Brien PM, Tsirimonaki E, Coomber DW, Millan DW, Davis JA, Campo MS (2001) Immunoglobulin genes expressed by B-lymphocytes infiltrating cervical carcinoma show evidence of antigen-driven selection. Cancer Immunol Immunother 50:523–532
Coronella JA, Tellemann P, Kingsbury GA, Truong TD, Hays S, Junghans RP (2001) Evidence for an antigen-driven humoral immune response in medullary ductal breast cancer. Cancer Res 61:7889–7899
Coronella JA, Spier C, Welch M, Trevor KT, Stopeck AT, Villar H, Hersh EM (2002) Antigen-driven oligoclonal expansion of tumor-infiltrating B cells in infiltrating ductal carcinoma of the breast. J Immunol 169:1829–1836
Nzula S, Going JJ, Stott DI (2003) Antigen-driven clonal proliferation, somatic hypermutation, and selection of B lymphocytes infiltrating human ductal breast carcinomas. Cancer Res 63:3275–3280
Yeilding NM, Gerstner C, Kirkwood JM (1992) Analysis of two human monoclonal antibodies against melanoma. Int J Cancer 52:967–973
Punt CJA, Barbuto JAM, Zhang H, Grimes WJ, Hatch KD, Hersh EM (1994) Anti-tumor antibody produced by human tumor-infiltrating and peripheral blood B lymphocytes. Cancer Immunol Immunother 38:225–232
Kotlan B, Simsa P, Teillaud J-L, Fridman WH, Toth J, McKnight M, Glassy MC (2005) Novel ganglioside antigen identified by B cells in human medullary breast carcinomas: the proof of principle concerning the tumor-infiltrating B lymphocytes. J Immunol 175:2278–2285
Dieu-Nosjean M-C, Antoine M, Danel C, Heudes D, Wislez M, Poulot V, Rabbe N, Laurans L, Tartour E, de Chaisemartin L, Lebecque S, Fridman W-H, Cadranel J (2008) Long-term survival for patients with non–small-cell lung cancer with intratumoral lymphoid structures. J Clin Oncol 26:4410–4417
Suzuki A, Masuda A, Nagata H, Kameoka S, Kikawada Y, Yamakawa M, Kasajima T (2002) Mature dendritic cells make clusters with T cells in the invasive margin of colorectal carcinoma. J Pathol 196:37–43
de Visser KE, Korets LV, Coussens LM (2005) De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell 7:411–423
Brodt P, Gordon J (1978) Anti-tumor immunity in B lymphocyte-deprived mice. I. immunity to a chemically induced tumor. J Immunol 121:359–362
Qin Z, Richter G, Schüler T, Ibe S, Cao X, Blankenstein T (1998) B cells inhibit induction of T cell-dependent tumor immunity. Nat Med 4:627–630
Shah S, Divekar AA, Hilchey SP, Cho H-M, Newman CL, Shin S-U, Nechustan H, Challita-Eid PM, Segal BM, Yi KH (2005) Increased rejection of primary tumors in mice lacking B cells: inhibition of anti-tumor CTL and TH1 cytokine responses by B cells. Int J Cancer 117:574–586
Schultz KR, Klarnet JP, Gieni RS, HayGlass KT, Greenberg PD (1990) The role of B cells for in vivo T cell responses to a Friend virus-induced leukemia. Science 249:921–923
DiLillo DJ, Yanaba K, Tedder TF (2010) B cells are required for optimal CD4+ and CD8+ T cell tumor immunity: therapeutic B cell depletion enhances B16 melanoma growth in mice. J Immunol 184:4006–4016
Kaliss N (1958) Immunological enhancement of tumor homografts in mice: a review. Cancer Res 18:992–1003
Salinas FA, Wee KH (1986) Prognostic and pathogenetic implications of immune complexes in human cancer. Adv Immun Cancer Ther 2:189–209
Johansson M, DeNardo DG, Coussens LM (2008) Polarized immune responses differentially regulate cancer development. Immunol Rev 222:145–154
Lapointe R, Bellemare-Pelletier A, Housseau F, Thibodeau J, Hwu P (2003) CD40-stimulated B lymphocytes pulsed with tumor antigens are effective antigen-presenting cells that can generate specific T cells. Cancer Res 63:2836–2843
Li Q, Grover AC, Donald EJ, Carr A, Yu J, Whitfield J, Nelson M, Takeshita N, Chang AE (2005) Simultaneous targeting of CD3 on T cells and CD40 on B or dendritic cells augments the antitumor reactivity of tumor-primed lymph node cells. J Immunol 175:1424–1432
Linton P-J, Harbertson J, Bradley LM (2000) A critical role for B cells in the development of memory CD4 cells. J Immunol 165:5558–5565
Rivera A, Chen CC, Ron N, Dougherty JP, Ron Y (2001) Role of B cells as antigen-presenting cells in vivo revisited: antigen-specific B cells are essential for T cell expansion in lymph nodes and for systemic T cell responses to low antigen concentrations. Int Immunol 13:1583–1593
Linton P-J, Bautista B, Biederman E, Bradley ES, Harbertson J, Kondrack RM, Padrick RC, Bradley LM (2003) Costimulation via OX40L expressed by B cells is sufficient to determine the extent of primary CD4 cell expansion and Th2 cytokine secretion in vivo. J Exp Med 197:875–883
Reuschenbach M, von Knebel Doeberitz M, Wentzensen N (2009) A systematic review of humoral immune response against tumor antigens. Cancer Immunol Immunother 58:1535–1544
Milne K, Köbel M, Kalloger SE, Barnes RO, Gao D, Gilks CB, Watson PH, Nelson BH (2009) Systematic analysis of immune infiltrates in high-grade serous ovarian cancer reveals CD20, FoxP3 and TIA-1 as positive prognostic factors. PLoS One 4:e6412
Acknowledgments
The authors thank K. Derecskei, I. Sinka and M. Kónya (National Institute of Oncology, Budapest) for their excellent technical assistance. The study was supported by the National Scientific Research Fund OTKA 72836 (AL), Hungarian Ministry of Health grant ETT 308/2003 (AL), and NKFP1a-0024-05 (JT).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ladányi, A., Kiss, J., Mohos, A. et al. Prognostic impact of B-cell density in cutaneous melanoma. Cancer Immunol Immunother 60, 1729–1738 (2011). https://doi.org/10.1007/s00262-011-1071-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-011-1071-x