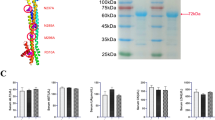
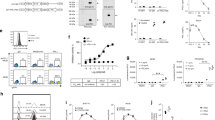
Abstract
Adoptive transfer of tumor-specific T cells has shown some success for treating metastatic melanoma. We evaluated a novel strategy to improve adoptive therapy by administering both T cells and oncolytic myxoma virus to mice with syngeneic B16.SIY melanoma brain tumors. Adoptive transfer of activated CD8+ 2C T cells that recognize SIY peptide doubled survival time, but SIY-negative tumors recurred. Myxoma virus killed B16.SIY cells in vitro, and intratumoral injection of virus led to selective and transient infection of the tumor. Virus treatment recruited innate immune cells to the tumor and induced IFNβ production in the brain, resulting in limited oncolytic effects in vivo. To counter this, we evaluated the safety and efficacy of co-administering 2C T cells, myxoma virus, and either rapamycin or neutralizing antibodies against IFNβ. Mice that received either triple combination therapy survived significantly longer with no apparent side effects, but eventually relapsed. Importantly, rapamycin treatment did not impair T cell-mediated tumor destruction, supporting the feasibility of combining adoptive immunotherapy and rapamycin-enhanced virotherapy. Myxoma virus may be a useful vector for transient delivery of therapeutic genes to a tumor to enhance T cell responses.






Similar content being viewed by others
References
Sampson JH, Carter JH Jr, Friedman AH, Seigler HF (1998) Demographics, prognosis, and therapy in 702 patients with brain metastases from malignant melanoma. J Neurosurg 88(1):11–20. doi:10.3171/jns.1998.88.1.0011
Rosenberg SA, Dudley ME (2009) Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr Opin Immunol 21(2):233–240. doi:10.1016/j.coi.2009.03.002
Dudley ME, Yang JC, Sherry R, Hughes MS, Royal R, Kammula U, Robbins PF, Huang J, Citrin DE, Leitman SF, Wunderlich J, Restifo NP, Thomasian A, Downey SG, Smith FO, Klapper J, Morton K, Laurencot C, White DE, Rosenberg SA (2008) Adoptive cell therapy for patients with metastatic melanoma: evaluation of intensive myeloablative chemoradiation preparative regimens. J Clin Oncol 26(32):5233–5239. doi:10.1200/JCO.2008.16.5449
Hong JJ, Rosenberg SA, Dudley ME, Yang JC, White DE, Butman JA, Sherry RM (2010) Successful Treatment of Melanoma Brain Metastases With Adoptive Cell Therapy. Clin Cancer Res. doi:10.1158/1078-0432.CCR-10-1507
Jager E, Ringhoffer M, Karbach J, Arand M, Oesch F, Knuth A (1996) Inverse relationship of melanocyte differentiation antigen expression in melanoma tissues and CD8 + cytotoxic-T-cell responses: evidence for immunoselection of antigen-loss variants in vivo. Int J Cancer 66(4):470–476. doi: 10.1002/(SICI)1097-0215(19960516)66:4<470:AID-IJC10>3.0.CO;2-C
Riker A, Cormier J, Panelli M, Kammula U, Wang E, Abati A, Fetsch P, Lee KH, Steinberg S, Rosenberg S, Marincola F (1999) Immune selection after antigen-specific immunotherapy of melanoma. Surgery 126(2):112–120. doi:S0039606099002044
Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, Celis E, Greenberg PD (2002) Adoptive T cell therapy using antigen-specific CD8 + T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci USA 99(25):16168–16173
Goldberger O, Volovitz I, Machlenkin A, Vadai E, Tzehoval E, Eisenbach L (2008) Exuberated numbers of tumor-specific T cells result in tumor escape. Cancer Res 68(9):3450–3457. doi:10.1158/0008-5472.CAN-07-5006
Dunn GP, Koebel CM, Schreiber RD (2006) Interferons, immunity and cancer immunoediting. Nat Rev Immunol 6(11):836–848. doi:10.1038/nri1961
Sampson JH, Archer GE, Mitchell DA, Heimberger AB, Bigner DD (2008) Tumor-specific immunotherapy targeting the EGFRvIII mutation in patients with malignant glioma. Semin Immunol 20(5):267–275
Thomas DL, Kim M, Bowerman NA, Narayanan S, Kranz DM, Schreiber H, Roy EJ (2009) Recurrence of intracranial tumors following adoptive T cell therapy can be prevented by direct and indirect killing aided by high levels of tumor antigen cross-presented on stromal cells. J Immunol 183(3):1828–1837. doi:10.4049/jimmunol.0802322
Diaz RM, Galivo F, Kottke T, Wongthida P, Qiao J, Thompson J, Valdes M, Barber G, Vile RG (2007) Oncolytic immunovirotherapy for melanoma using vesicular stomatitis virus. Cancer Res 67(6):2840–2848. doi:10.1158/0008-5472.CAN-06-3974
Qiao J, Wang H, Kottke T, Diaz RM, Willmon C, Hudacek A, Thompson J, Parato K, Bell J, Naik J, Chester J, Selby P, Harrington K, Melcher A, Vile RG (2008) Loading of oncolytic vesicular stomatitis virus onto antigen-specific T cells enhances the efficacy of adoptive T-cell therapy of tumors. Gene Ther 15(8):604–616. doi:10.1038/sj.gt.3303098
Zhang YQ, Tsai YC, Monie A, Wu TC, Hung CF (2010) Enhancing the therapeutic effect against ovarian cancer through a combination of viral oncolysis and antigen-specific immunotherapy. Mol Ther 18(4):692–699. doi:10.1038/mt.2009.318
Sypula WFJ, Ma Y, Bell J, McFadden G (2004) Myxoma virus tropism in human tumor cells. Gene Ther Mol Biol 8:103–114
Lun X, Yang W, Alain T, Shi ZQ, Muzik H, Barrett JW, McFadden G, Bell J, Hamilton MG, Senger DL, Forsyth PA (2005) Myxoma virus is a novel oncolytic virus with significant antitumor activity against experimental human gliomas. Cancer Res 65(21):9982–9990
Lun XQ, Zhou H, Alain T, Sun B, Wang L, Barrett JW, Stanford MM, McFadden G, Bell J, Senger DL, Forsyth PA (2007) Targeting human medulloblastoma: oncolytic virotherapy with myxoma virus is enhanced by rapamycin. Cancer Res 67(18):8818–8827
Woo Y, Kelly KJ, Stanford MM, Galanis C, Chun YS, Fong Y, McFadden G (2008) Myxoma virus is oncolytic for human pancreatic adenocarcinoma cells. Ann Surg Oncol 15(8):2329–2335. doi:10.1245/s10434-008-9924-z
Wu Y, Lun X, Zhou H, Wang L, Sun B, Bell JC, Barrett JW, McFadden G, Biegel JA, Senger DL, Forsyth PA (2008) Oncolytic efficacy of recombinant vesicular stomatitis virus and myxoma virus in experimental models of rhabdoid tumors. Clin Cancer Res 14(4):1218–1227. doi:10.1158/1078-0432.CCR-07-1330
Kim M, Madlambayan GJ, Rahman MM, Smallwood SE, Meacham AM, Hosaka K, Scott EW, Cogle CR, McFadden G (2009) Myxoma virus targets primary human leukemic stem and progenitor cells while sparing normal hematopoietic stem and progenitor cells. Leukemia 23(12):2313–2317. doi:10.1038/leu.2009.219
Stanford MM, Shaban M, Barrett JW, Werden SJ, Gilbert PA, Bondy-Denomy J, Mackenzie L, Graham KC, Chambers AF, McFadden G (2008) Myxoma virus oncolysis of primary and metastatic B16F10 mouse tumors in vivo. Mol Ther 16(1):52–59
Lun X, Alain T, Zemp FJ, Zhou H, Rahman MM, Hamilton MG, McFadden G, Bell J, Senger DL, Forsyth PA (2010) Myxoma virus virotherapy for glioma in immunocompetent animal models: optimizing administration routes and synergy with rapamycin. Cancer Res 70(2):598–608. doi:10.1158/0008-5472.CAN-09-1510
McFadden G (2005) Poxvirus tropism. Nat Rev Microbiol 3(3):201–213. doi:10.1038/nrmicro1099
Jackson EW, Dorn CR, Saito JK, McKercher DG (1966) Absence of serological evidence of myxoma virus infection in humans exposed during an outbreak of myxomatosis. Nature 211(5046):313–314
Homicsko K, Lukashev A, Iggo RD (2005) RAD001 (everolimus) improves the efficacy of replicating adenoviruses that target colon cancer. Cancer Res 65(15):6882–6890. doi:10.1158/0008-5472.CAN-05-0309
Alonso MM, Gomez-Manzano C, Jiang H, Bekele NB, Piao Y, Yung WK, Alemany R, Fueyo J (2007) Combination of the oncolytic adenovirus ICOVIR-5 with chemotherapy provides enhanced anti-glioma effect in vivo. Cancer Gene Ther 14(8):756–761. doi:10.1038/sj.cgt.7701067
Alonso MM, Jiang H, Yokoyama T, Xu J, Bekele NB, Lang FF, Kondo S, Gomez-Manzano C, Fueyo J (2008) Delta-24-RGD in combination with RAD001 induces enhanced anti-glioma effect via autophagic cell death. Mol Ther 16(3):487–493. doi:10.1038/sj.mt.6300400
Lun XQ, Jang JH, Tang N, Deng H, Head R, Bell JC, Stojdl DF, Nutt CL, Senger DL, Forsyth PA, McCart JA (2009) Efficacy of systemically administered oncolytic vaccinia virotherapy for malignant gliomas is enhanced by combination therapy with rapamycin or cyclophosphamide. Clin Cancer Res 15(8):2777–2788. doi:10.1158/1078-0432.CCR-08-2342
Alain T, Lun X, Martineau Y, Sean P, Pulendran B, Petroulakis E, Zemp FJ, Lemay CG, Roy D, Bell JC, Thomas G, Kozma SC, Forsyth PA, Costa-Mattioli M, Sonenberg N (2010) Vesicular stomatitis virus oncolysis is potentiated by impairing mTORC1-dependent type I IFN production. Proc Natl Acad Sci USA 107(4):1576–1581. doi:10.1073/pnas.0912344107
Lun X, Chan J, Zhou H, Sun B, Kelly JJ, Stechishin OO, Bell JC, Parato K, Hu K, Vaillant D, Wang J, Liu TC, Breitbach C, Kirn D, Senger DL, Forsyth PA (2010) Efficacy and safety/toxicity study of recombinant vaccinia virus JX-594 in two immunocompetent animal models of glioma. Mol Ther 18(11):1927–1936. doi:10.1038/mt.2010.183
Blank C, Brown I, Peterson AC, Spiotto M, Iwai Y, Honjo T, Gajewski TF (2004) PD-L1/B7H–1 inhibits the effector phase of tumor rejection by T cell receptor (TCR) transgenic CD8 + T cells. Cancer Res 64(3):1140–1145
Liu J, Wennier S, Reinhard M, Roy E, MacNeill A, McFadden G (2009) Myxoma virus expressing interleukin-15 fails to cause lethal myxomatosis in European rabbits. J Virol 83(11):5933–5938. doi:10.1128/JVI.00204-09
Zhang B, Bowerman NA, Salama JK, Schmidt H, Spiotto MT, Schietinger A, Yu P, Fu YX, Weichselbaum RR, Rowley DA, Kranz DM, Schreiber H (2007) Induced sensitization of tumor stroma leads to eradication of established cancer by T cells. J Exp Med 204(1):49–55
Calzascia T, Di Berardino-Besson W, Wilmotte R, Masson F, de Tribolet N, Dietrich PY, Walker PR (2003) Cutting edge: cross-presentation as a mechanism for efficient recruitment of tumor-specific CTL to the brain. J Immunol 171(5):2187–2191
Kline J, Brown IE, Zha YY, Blank C, Strickler J, Wouters H, Zhang L, Gajewski TF (2008) Homeostatic proliferation plus regulatory T-cell depletion promotes potent rejection of B16 melanoma. Clin Cancer Res 14(10):3156–3167. doi:10.1158/1078-0432.CCR-07-4696
Surh CD, Sprent J (2008) Homeostasis of naive and memory T cells. Immunity 29(6):848–862. doi:10.1016/j.immuni.2008.11.002
Ge Q, Hu H, Eisen HN, Chen J (2002) Different contributions of thymopoiesis and homeostasis-driven proliferation to the reconstitution of naive and memory T cell compartments. Proc Natl Acad Sci USA 99(5):2989–2994. doi:10.1073/pnas.05271409999/5/2989
Wang F, Ma Y, Barrett JW, Gao X, Loh J, Barton E, Virgin HW, McFadden G (2004) Disruption of Erk-dependent type I interferon induction breaks the myxoma virus species barrier. Nat Immunol 5(12):1266–1274
Bartee E, Mohamed MR, Lopez MC, Baker HV, McFadden G (2009) The addition of tumor necrosis factor plus beta interferon induces a novel synergistic antiviral state against poxviruses in primary human fibroblasts. J Virol 83(2):498–511. doi:10.1128/JVI.01376-08
Fujita M, Scheurer ME, Decker SA, McDonald HA, Kohanbash G, Kastenhuber ER, Kato H, Bondy ML, Ohlfest JR, Okada H (2010) Role of type 1 IFNs in antiglioma immunosurveillance–using mouse studies to guide examination of novel prognostic markers in humans. Clin Cancer Res 16(13):3409–3419. doi:10.1158/1078-0432.CCR-10-0644
Willmon CL, Saloura V, Fridlender ZG, Wongthida P, Diaz RM, Thompson J, Kottke T, Federspiel M, Barber G, Albelda SM, Vile RG (2009) Expression of IFN-beta enhances both efficacy and safety of oncolytic vesicular stomatitis virus for therapy of mesothelioma. Cancer Res 69(19):7713–7720. doi:10.1158/0008-5472.CAN-09-1013
Xu B, Grander D, Sangfelt O, Einhorn S (1994) Primary leukemia cells resistant to alpha-interferon in vitro are defective in the activation of the DNA-binding factor interferon-stimulated gene factor 3. Blood 84(6):1942–1949
Wong LH, Krauer KG, Hatzinisiriou I, Estcourt MJ, Hersey P, Tam ND, Edmondson S, Devenish RJ, Ralph SJ (1997) Interferon-resistant human melanoma cells are deficient in ISGF3 components, STAT1, STAT2, and p48-ISGF3gamma. J Biol Chem 272(45):28779–28785
Dunn GP, Sheehan KC, Old LJ, Schreiber RD (2005) IFN unresponsiveness in LNCaP cells due to the lack of JAK1 gene expression. Cancer Res 65(8):3447–3453. doi:10.1158/0008-5472.CAN-04-4316
Stanford MM, Barrett JW, Nazarian SH, Werden S, McFadden G (2007) Oncolytic virotherapy synergism with signaling inhibitors: Rapamycin increases myxoma virus tropism for human tumor cells. J Virol 81(3):1251–1260
Araki K, Turner AP, Shaffer VO, Gangappa S, Keller SA, Bachmann MF, Larsen CP, Ahmed R (2009) mTOR regulates memory CD8 T-cell differentiation. Nature 460(7251):108–112. doi:10.1038/nature08155
Acknowledgments
We thank the anonymous donor that funded this research, Thomas Gajewski and Maciej Lesniak for the B16.SIY cells, and the Flow Cytometry Facility at University of Illinois at Urbana–Champaign.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thomas, D.L., Doty, R., Tosic, V. et al. Myxoma virus combined with rapamycin treatment enhances adoptive T cell therapy for murine melanoma brain tumors. Cancer Immunol Immunother 60, 1461–1472 (2011). https://doi.org/10.1007/s00262-011-1045-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-011-1045-z