Abstract
Background
Dendritic cell (DC)-based vaccines have been applied clinically in the setting of cancer, but tumor-associated antigens (TAAs) have not yet been enough identified in various cancers. In this study, we investigated whether preventive vaccination with unpulsed DCs or peptide-pulsed DCs could offer anti-tumor effects against MC38 or BL6 liver tumors.
Methods
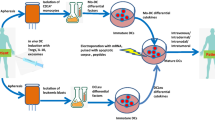
Mice were subcutaneously (s.c.) immunized with unpulsed DCs or the recently defined TAA EphA2 derived peptide-pulsed dendritic cells (Eph-DCs) to treat EphA2-positive MC38 and EphA2-negative BL6 liver tumors. Liver mononuclear cells (LMNCs) from treated mice were subjected to 51Cr release assays against YAC-1 target cells. In some experiments, mice were injected with anti-CD8, anti-CD4 or anti-asialo GM1 antibody to deplete each lymphocyte subsets.
Results
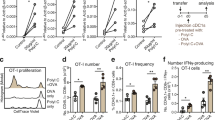
Immunization with unpulsed DCs displayed comparable efficacy against both MC38 and BL6 liver tumors when compared with Eph-DCs. Both DC-based vaccines significantly augmented the cytotoxicity of LMNCs against YAC-1 cells. In vivo antibody depletion studies revealed that NK cells, as well as, CD4+ and CD8+ T cells play critical roles in the anti-tumor efficacy associated with either DC-based modality.
Tumor-specific cytotoxic T lymphocyte (CTL) activity was generally higher if mice had received Eph-DCs versus unpulsed DCs. Importantly, the mice that had been protected from MC38 liver tumor by either unpulsed DCs or Eph-DCs became resistant to s.c. MC38 rechallenge, but not to BL6 rechallenge.
Conclusions
These results demonstrate that unpulsed DC vaccines might serve as an effective therapy for treating metastatic liver tumor, for which TAA has not yet been identified.





Similar content being viewed by others
Abbreviations
- DC:
-
Dendritic cell
- Eph-DCs:
-
EphA2 derived peptide-pulsed dendritic cells
- CTLs:
-
Cytotoxic T cell lymphocytes
- s.c.:
-
Subcutaneously
- SCID:
-
Severe combined immuno-deficiency
- BM:
-
Bone marrow
- GM-CSF:
-
Granulocyte/macrophage-colony stimulating factor
- PBS:
-
Phosphate-buffered saline
- LMNC:
-
Liver mononuclear cell
- TAA:
-
Tumor-associated antigen
References
Steinman RM (1991) The dendritic cell system and its role in immunogenicity. Annu Rev Immunol 9:271–296
Hart DN (1997) Dendritic cells: unique leukocyte populations which control the primary immune response. Blood 90:3245–3287
Smith CM, Wilson NS, Waithman J, Villadangos JA, Carbone FR, Heath WR, Belz GT (2004) Cognate CD4(+) T cell licensing of dendritic cells in CD8(+) T cell immunity. Nat Immunol 5:1143–1148
O’Neill DW, Adams S, Bhardwaj N (2004) Manipulating dendritic cell biology for the active immunotherapy of cancer. Blood 104:2235–2246
Fernandez NC, Lozier A, Flament C, Ricciardi-Castagnoli P, Bellet D, Suter M, Perricaudet M, Tursz T, Maraskovsky E, Zitvogel L (1999) Dendritic cells directly trigger NK cell functions: cross-talk relevant in innate anti-tumor immune responses in vivo. Nat Med 5:405–411
Gerosa F, Baldani-Guerra B, Nisii C, Marchesini V, Carra G, Trinchieri G (2002) Reciprocal activating interaction between natural killer cells and dendritic cells. J Exp Med 195:327–333
Miller G, Lahrs S, Dematteo RP (2003) Overexpression of interleukin–12 enables dendritic cells to activate NK cells and confer systemic antitumor immunity. FASEB J 17:728–730
Piccioli D, Sbrana S, Melandri E, Valiante NM (2002) Contact-dependent stimulation and inhibition of dendritic cells by natural killer cells. J Exp Med 195:335–341
Jinushi M, Takehara T, Kanto T, Tatsumi T, Groh V, Spies T, Miyagi T, Suzuki T, Sasaki Y, Hayashi N (2003) Critical role of MHC class I-related chain A and B expression on IFNα-stimulated dendritic cells in NK cell activation: Impairment in chronic hepatitis C virus infection. J Immunol 170:1249–1256
Kawano T, Cui J, Koezuka Y, Toura Y, Kaneko Y, Motoki K, Ueno H, Nakagawa R, Sato H, Kondo E, Koseki H, Taniguchi M (1997) CD1d-restricted and TCR-mediated activation of Vα14NKT cells by glycosylceramids. Science 278:1626–1629
Kawano T, Cui J, Koezuka Y, Toura I, Kaneko Y, Sato H, Kondo E, Harada M, Koseki H, Tanaka Y, Taniguchi M (1998) Natural killer-like nonspecific tumor cell lysis mediated by specific ligand-activated Vα14NKT cells. Proc Natl Acad Sci USA 95:5690–5693
Kitamura H, Iwakabe K, Yahata T, Nishimura S, Ohta A, Ohmi Y, Sato M, Takeda K, Okumura K, Van Kaer L, Kawano T, Taniguchi M, Nishimura T (1999) The natural killer T (NKT) cell-ligand-Galactosylceramide demonstrates immunopoteniating effects by inducing interleukin (IL)-12 production by dendritic cells and IL-12 receptor expression on NKT cells. J Exp Med 189:1121–1128
Wiltrout RH (2000) Regulation and antimetastatic functions of liver-associated natural killer cells. Immunol Rev 174:63–76
George AP, Catherine AP (2005) Liver immunobiology. Toxicol Pathol 33:52–62
Doherty DG, O’Farrelly C (2000) Innate and adaptive lymphoid cells in human liver. Immunol Rev 174:5–20
Miyagi T, Takehara T, Tatsumi T, Kanto T, Suzuki T, Jinushi M, Sugimoto Y, Sasaki Y, Hori M, Hayashi N (2003) CD1d-mediated stimulation of natural killer T cells selectively activates hepatic natural killer T cells to eliminate experimentally disseminated hepatoma cells in murine liver. Int J Cancer 106:81–89
Nakagawa R, Motoki K, Ueno H, Iijima R, Nakamura H, Kobayashi E, Shimosaka A, Koezuka Y (1998) Treatment of hepatic metastasis of the colon 26 adenocarcinoma with a Galactosylceramide, KRN7000. Cancer Res 58:1202–1207
Mayordome JI, Zorina T, Storkus WJ, Zitvogel L, Celluzzi C, Falo LD, Melief CJ, Ildstad ST, Kast WM, Deleo AB et al (1995) Bone marrow-derived dendritic cells pulsed with synthetic tumor peptide elicits protective and therapeutic antitumor immunity. Nat Med 12:1297–1302
Paglia P, Chiodoni C, Rodolfo M, Colombo MP (1996) Murine dendritic cells loaded in vitro with soluble protein prime cytotoxic T lymphocytes against tumor antigen in vivo. J Exp Med 183:317–322
Mayordomo JI, Loftus DJ, Sakamoto H, De Cesare CM, Appasamy PM, Lotze MT, Storkus WJ, Appella E, DeLeo AB (1996) Therapy of murine tumors with p53 wild-type and mutant sequence peptide-based vaccines. J Exp Med 183:1357–1365
Olson RM, Perencevich NP, Malcolm AW, Chaffey JT, Wilson RE (1980) Patterns of recurrence following curative resection of adenocarcinoma of the colon and rectum. Cancer 45:2969–2974
Malcolm AW, Perencevich NP, Olson RM, Hanley JA, Chaffey JT, Wilson RE (1981) Analysis of recurrence patterns following curative resection for carcinoma of thecolon and rectum. Surg Gynecol Obstet 152:131–136
Ota M, Shimada H, Masui H, Tanaka K, Yamaguchi S, Ichikawa Y, Togo S, Ike H, Oki S (2004) Adjuvant hepatic arterial infusion chemotherapy after curative resection for Dukes colorectal cancer: a pilot study. Hepatogastroenterology 51:124–127
Arkenau HT, Rettig K, Porschen R (2005) Adjuvant chemotherapy in curative resected colon carcinoma: 5-fluorouracil/leucovorin versus high-dose 5- fluorouracil 24-h infusion versus high-dose 5-fluorouracil 24-h infusion. Int J Colorectal Dis 20:258–261
Hayashibe A, Kameyama M, Shinbo M, Makimoto S (2007) Clinical results on intra-arterial adjuvant chemotherapy for prevention of liver metastasis following curative resection of pancreatic cancer. Ann Surg Oncol 14:190–194
Liu KJ, Wang CC, Chen LT, Cheng AL, Lin DT, Wu YC, Yu WL, Hung YM, Yang HY, Juang SH, Whang-Peng J (2004) Generation of carcinoembryonic antigen (CEA)- specific T-cell responses in HLA-A*0201 and HLA-A*2402 late-stage colorectal cancer patients after vaccination with dendritic cells loaded with CEA peptides. Clin Cancer Res 10:2645–2651
Sadanaga N, Nagashima H, Mashino K, Tahara K, Yamaguchi H, Ohta M, Fujie T, Tanaka F, Inoue H, Takesako K, Akiyoshi T, Mori M (2001) Dendritic cell vaccination with MAGE peptide is a novel therapeutic approach for gastrointestinal carcinomas. Clin Cancer Res 7:2277–2284
Timmerman JM, Levy R (1999) Dendritic cell vaccines for cancer immunotherapy. Annu Rev Med 50:507–529
Nevellino L, Castelli C, Parmiani G (2005) A listing of human tumor antigens recognized by T cells: March 2004 update. Cancer Immunol Immunother 54:187–207
Simone M, Carlo RR, Mario L, Donato N (2004) Colorectal cancer vaccines: principles, results, and perspectives. Gastroenterology 27:1821–1837
Oka Y, Tsuboi A, Taguchi T, Osaki T, Kyo T, Nakajima H, Elisseeva OA, Oji Y, Kawakami M, Ikegame K, Hosen N, Yoshihara S, Wu Y, Fujiki F, Murukami M, Masuda T, Nishida S, Shirakata T, Nakatsuka S, Sasaki A, Udaka K, Dohy H, Aozasa K, Noguchi S, Kawase I, Sugiyama H (2004) Induction of WT1 (Wilms’ tumor gene)-specific cytotoxic T lymphocytes by WT1 peptide vaccine and the resultant cancer regression. Pro Natl Acad Sci USA 138:13885–13890
Yamaguchi S, Tatsumi T, Takehara T, Sakamori R, Uemura A, Mizushima T, Ohkawa K, Hayashi N (2007) Immunotherapy of murine colon cancer using receptor tyrosine kinase EphA2-derived peptide-pulsed dendritic cell vaccines. Cancer 110(7):1469–1477
Felazzo G, Munz C (2004) NK cell compartments and their activation by dendritic cells. J Immunol 172:1333–1339
Nieda M, Okai M, Tazbirkova A, Lin H, Yamamura A, Ide K, Abraham R, Juji T, Macfarlane DJ, Nicol AJ (2004) Therapeutic activation of Vα24 + Vβ11 + NKT cells in human subjects results in highly coordinates secondary activation of acquired and innate immunity. Blood 103:383–389
Acknowledgments
The authors wish to thank Dr. Walter J. Storkus (The University of Pittsburgh) for careful review and comments provided during the preparation of this manuscript. This work was supported by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan and a Grant-in-Aid for Research on Hepatitis and BSE from the Ministry of Health, Labor and Welfare of Japan [N.H.].
Author information
Authors and Affiliations
Corresponding author
Additional information
Shinjiro Yamaguchi and Tomohide Tatsumi contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yamaguchi, S., Tatsumi, T., Takehara, T. et al. Dendritic cell-based vaccines suppress metastatic liver tumor via activation of local innate and acquired immunity. Cancer Immunol Immunother 57, 1861–1869 (2008). https://doi.org/10.1007/s00262-008-0514-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-008-0514-5