Abstract
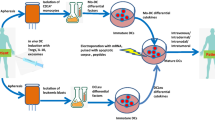
Tumor-associated peptides isolated by acid elution are frequently used for therapeutic immunization against various tumors both in mice and in humans. In acute myeloid leukemia (AML), the frequent accessibility of a large tumor burden allows for extraction of peptides from leukemia cells by using either citrate–phosphate (CP) or trifluoroacetic acid (TFA) buffer. To develop an optimal immunotherapeutic protocol for AML patients, we evaluated both in mice and in humans, the immunogenicity of peptides eluted from leukemia cells with the two acids (TFA or CP). Although ex vivo studies in mice showed that both prophylactic immunizations with mature dendritic cells (DC) loaded with TFA-peptides (DC/TFA), or CP-peptides (DC/CP), were able to stimulate specific antileukemia immune responses, only vaccination with DC/TFA was able to prevent leukemia outgrowth. Moreover, in humans, only DC/TFA generated significant antileukemia CD4+ and cytotoxic CD8+ T cell responses in vitro. In summary, these data demonstrate that the choice of the acid elution procedure to isolate immunogenic peptides strongly influences the efficacy of the antileukemia immune responses. These finding raise essential considerations for the development of immunotherapeutic protocols for cancer patients. In our model, our results argue for the use of the TFA elution method to extract immunogenic AML-associated peptides.





Similar content being viewed by others
Abbreviations
- Mature DC:
-
Mature dendritic cells
- TFA:
-
Trifluoroacetic acid
- CP:
-
Citrate–phosphate
- DC/TFA:
-
Mature dendritic cells loaded with peptides extracted from leukemia cells using TFA buffer
- DC/CP:
-
Mature dendritic cells loaded with peptides extracted from leukemia cells using CP buffer
- MST:
-
Median survival time
References
Avigan D, Vasir B, Gong J, Borges V, Wu Z, Uhl L, Atkins M, Mier J, McDermott D, Smith T, Giallambardo N, Stone C, Schadt K, Dolgoff J, Tetreault JC, Villarroel M, Kufe D (2004) Fusion cell vaccination of patients with metastatic breast and renal cancer induces immunological and clinical responses. Clin Cancer Res 10:4699–4708
Bellone M, Iezzi G, Imro MA, Protti MP (1999) Cancer immunotherapy: synthetic and natural peptides in the balance. Immunol Today 20:457–462
Bellone M, Iezzi G, Manfredi AA, Protti MP, Dellabona P, Casorati G, Rugarli C (1994) In vitro priming of cytotoxic T lymphocytes against poorly immunogenic epitopes by engineered antigen-presenting cells. Eur J Immunol 24:2691–2698
Boyer MW, Orchard PJ, Gorden KB, Anderson PM, McLvor RS, Blazar BR (1995) Dependency on intercellular adhesion molecule recognition and local interleukin-2 provision in generation of an in vivo CD8+ T-cell immune response to murine myeloid leukemia. Blood 85:2498–2506
Bradner WT, Pindell MH (1966) Myeloid leukemia C-1498 as a screen for cancer chemotherapeutic agents. Cancer Res 26:375–390
Burroughs L, Storb R (2005) Low-intensity allogeneic hematopoietic stem cell transplantation for myeloid malignancies: separating graft-versus-leukemia effects from graft-versus-host disease. Curr Opin Hematol 12:45–54
Celluzzi CM, Mayordomo JI, Storkus WJ, Lotze MT, Falo LD Jr (1996) Peptide-pulsed dendritic cells induce antigen-specific CTL-mediated protective tumor immunity. J Exp Med 183:283–287
Chaput N, Schartz NE, Andre F, Taieb J, Novault S, Bonnaventure P, Aubert N, Bernard J, Lemonnier F, Merad M, Adema G, Adams M, Ferrantini M, Carpentier AF, Escudier B, Tursz T, Angevin E, Zitvogel L (2004) Exosomes as potent cell-free peptide-based vaccine. II. Exosomes in CpG adjuvants efficiently prime naive Tc1 lymphocytes leading to tumor rejection. J Immunol 172:2137–2146
Delluc S, Tourneur L, Michallet AS, Boix C, Varet B, Fradelizi D, Guillet JG, Buzyn A (2005) Autologous peptides eluted from acute myeloid leukemia cells can be used to generate specific antileukemic CD4 helper and CD8 cytotoxic T lymphocyte responses in vitro. Haematologica 90:1050–1062
Dorfel D, Appel S, Grunebach F, Weck MM, Muller MR, Heine A, Brossart P (2005) Processing and presentation of HLA class I and II epitopes by dendritic cells after transfection with in vitro-transcribed MUC1 RNA. Blood 105:3199–3205
Falk K, Rotzschke O, Deres K, Metzger J, Jung G, Rammensee HG (1991) Identification of naturally processed viral nonapeptides allows their quantification in infected cells and suggests an allele-specific T cell epitope forecast. J Exp Med 174:425–434
Fukui M, Nakano-Hashimoto T, Okano K, Maruta Y, Suehiro Y, Hamanaka Y, Yamashita H, Imai K, Kawano MM, Hinoda Y (2004) Therapeutic effect of dendritic cells loaded with a fusion mRNA encoding tyrosinase-related protein 2 and enhanced green fluorescence protein on B16 melanoma. Tumour Biol 25:252–257
Galea-Lauri J, Darling D, Mufti G, Harrison P, Farzaneh F (2002) Eliciting cytotoxic T lymphocytes against acute myeloid leukemia-derived antigens: evaluation of dendritic cell-leukemia cell hybrids and other antigen-loading strategies for dendritic cell-based vaccination. Cancer Immunol Immunother 51:299–310
Galea-Lauri J, Wells JW, Darling D, Harrison P, Farzaneh F (2004) Strategies for antigen choice and priming of dendritic cells influence the polarization and efficacy of antitumor T-cell responses in dendritic cell-based cancer vaccination. Cancer Immunol Immunother 53:963–977
Greiner J, Ringhoffer M, Simikopinko O, Szmaragowska A, Huebsch S, Maurer U, Bergmann L, Schmitt M (2000) Simultaneous expression of different immunogenic antigens in acute myeloid leukemia. Exp Hematol 28:1413–1422
Greiner J, Ringhoffer M, Taniguchi M, Li L, Schmitt A, Shiku H, Dohner H, Schmitt M (2004) mRNA expression of leukemia-associated antigens in patients with acute myeloid leukemia for the development of specific immunotherapies. Int J Cancer 108:704–711
Gritzapis AD, Perez SA, Baxevanis CN, Papamichail M (2005) Pooled peptides from HER-2/neu-overexpressing primary ovarian tumours induce CTL with potent antitumour responses in vitro and in vivo. Br J Cancer 92:72–79
Herr W, Ranieri E, Olson W, Zarour H, Gesualdo L, Storkus WJ (2000) Mature dendritic cells pulsed with freeze-thaw cell lysates define an effective in vitro vaccine designed to elicit EBV-specific CD4(+) and CD8(+) T lymphocyte responses. Blood 96:1857–1864
Heslop HE, Stevenson FK, Molldrem JJ (2003) Immunotherapy of hematologic malignancy. Hematology. Am Soc Hematol Educ Program 331–349
Houghton AN, Mintzer D, Cordon-Cardo C, Welt S, Fliegel B, Vadhan S, Carswell E, Melamed MR, Oettgen HF, Old LJ (1985) Mouse monoclonal IgG3 antibody detecting GD3 ganglioside: a phase I trial in patients with malignant melanoma. Proc Natl Acad Sci USA 82:1242–1246
Kim KJ, Kanellopoulos-Langevin C, Merwin RM, Sachs DH, Asofsky R (1979) Establishment and characterization of BALB/c lymphoma lines with B cell properties. J Immunol 122:549–554
Kurokawa T, Oelke M, Mackensen A (2001) Induction and clonal expansion of tumor-specific cytotoxic T lymphocytes from renal cell carcinoma patients after stimulation with autologous dendritic cells loaded with tumor cells. Int J Cancer 91:749–756
Lambert LA, Gibson GR, Maloney M, Barth RJ Jr (2001) Equipotent generation of protective antitumor immunity by various methods of dendritic cell loading with whole cell tumor antigens. J Immunother 24:232–36
Liau LM, Prins RM, Kiertscher SM, Odesa SK, Kremen TJ, Giovannone AJ, Lin JW, Chute DJ, Mischel PS, Cloughesy TF, Roth MD (2005) Dendritic cell vaccination in glioblastoma patients induces systemic and intracranial T-cell responses modulated by the local central nervous system tumor microenvironment. Clin Cancer Res 11:5515–5525
Mailander V, Scheibenbogen C, Thiel E, Letsch A, Blau IW, Keilholz U (2004) Complete remission in a patient with recurrent acute myeloid leukemia induced by vaccination with WT1 peptide in the absence of hematological or renal toxicity. Leukemia 18:165–166
Merle-Beral H, Nguyen Cong Duc L, Leblond V, Boucheix C, Michel A, Chastang C, Debre P (1989) Diagnostic and prognostic significance of myelomonocytic cell surface antigens in acute myeloid leukaemia. Br J Haematol 73:323–330
Ostankovitch M, Buzyn A, Bonhomme D, Connan F, Bouscary D, Heshmati F, Dreyfus F, Choppin J, Guillet JG (1998) Antileukemic HLA-restricted T-cell clones generated with naturally processed peptides eluted from acute myeloblastic leukemia blasts. Blood 92:19–24
Parkhurst MR, DePan C, Riley JP, Rosenberg SA, Shu S (2003) Hybrids of dendritic cells and tumor cells generated by electrofusion simultaneously present immunodominant epitopes from multiple human tumor-associated antigens in the context of MHC class I and class II molecules. J Immunol 170:5317–5325
Prasad SJ, Farrand KJ, Matthews SA, Chang JH, McHugh RS, Ronchese F (2005) Dendritic cells loaded with stressed tumor cells elicit long-lasting protective tumor immunity in mice depleted of CD4+CD25+ regulatory T cells. J Immunol 174:90–98
Rastetter W, Molina A, White CA (2004) Rituximab: expanding role in therapy for lymphomas and autoimmune diseases. Annu Rev Med 55:477–503
Rawson P, Hermans IF, Huck SP, Roberts JM, Pircher H, Ronchese F (2000) Immunotherapy with dendritic cells and tumor major histocompatibility complex class I-derived peptides requires a high density of antigen on tumor cells. Cancer Res 60:4493–4498
Ribas A, Butterfield LH, Hu B, Dissette VB, Meng WS, Koh A, Andrews KJ, Lee M, Amar SN, Glaspy JA, McBride WH, Economou JS (2000) Immune deviation and Fas-mediated deletion limit antitumor activity after multiple dendritic cell vaccinations in mice. Cancer Res 60:2218–2224
Rotzschke O, Falk K, Wallny HJ, Faath S, Rammensee HG (1990) Characterization of naturally occurring minor histocompatibility peptides including H-4 and H-Y. Science 249:283–287
Santin AD, Bellone S, Ravaggi A, Pecorelli S, Cannon MJ, Parham GP (2000) Induction of ovarian tumor-specific CD8+ cytotoxic T lymphocytes by acid-eluted peptide-pulsed autologous dendritic cells. Obstet Gynecol 96:422–430
Sauer MG, Ericson ME, Weigel BJ, Herron MJ, Panoskaltsis-Mortari A, Kren BT, Levine BL, Serody JS, June CH, Taylor PA, Blazar BR (2004) A novel system for simultaneous in vivo tracking and biological assessment of leukemia cells and ex vivo generated leukemia-reactive cytotoxic T cells. Cancer Res 64:3914–3921
Srivastava PK (1996) Do human cancers express shared protective antigens? Or the necessity of remembrance of things past. Semin Immunol 8:295–302
Storkus WJ, Zeh HJ 3rd, Salter RD, Lotze MT (1993) Identification of T-cell epitopes: rapid isolation of class I-presented peptides from viable cells by mild acid elution. J Immunother 14:94–103
Tanaka Y, Tevethia SS (1988) In vitro selection of SV40 T antigen epitope loss variants by site-specific cytotoxic T lymphocyte clones. J Immunol 140:4348–4354
Turk MJ, Wolchok JD, Guevara-Patino JA, Goldberg SM, Houghton AN (2002) Multiple pathways to tumor immunity and concomitant autoimmunity. Immunol Rev 188:122–135
Vegh Z, Mazumder A (2003) Generation of tumor cell lysate-loaded dendritic cells preprogrammed for IL-12 production and augmented T cell response. Cancer Immunol Immunother 52:67–79
Wetzler M, Baer MR, Stewart SJ, Donohue K, Ford L, Stewart CC, Repasky EA, Ferrone S (2001) HLA class I antigen cell surface expression is preserved on acute myeloid leukemia blasts at diagnosis and at relapse. Leukemia 15:128–133
Yang S, Vervaert CE, Burch J Jr, Grichnik J, Seigler HF, Darrow TL (1999) Murine dendritic cells transfected with human GP100 elicit both antigen-specific CD8(+) and CD4(+) T-cell responses and are more effective than DNA vaccines at generating anti-tumor immunity. Int J Cancer 83:532–540
Yu JS, Wheeler CJ, Zeltzer PM, Ying H, Finger DN, Lee PK, Yong WH, Incardona F, Thompson RC, Riedinger MS, Zhang W, Prins RM, Black KL (2001) Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Res 61:842–847
Zeng Y, Graner MW, Thompson S, Marron M, Katsanis E (2005) Induction of BCR-ABL-specific immunity following vaccination with chaperone-rich cell lysates derived from BCR-ABL+ tumor cells. Blood 105:2016–2022
Zitvogel L, Mayordomo JI, Tjandrawan T, DeLeo AB, Clarke MR, Lotze MT, Storkus WJ (1996) Therapy of murine tumors with tumor peptide-pulsed dendritic cells: dependence on T cells, B7 costimulation, and T helper cell 1-associated cytokines. J Exp Med 183:87–97
Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, Ricciardi-Castagnoli P, Raposo G, Amigorena S (1998) Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med 4:594–600
Acknowledgements
The authors are grateful to Dr Francoise Audat who suggested patients for inclusion in the study, and to Emilie Floch’ for her technical support. We thank Franck Lagger for the intravenously injection of leukemia cells in mice. This work was supported by the “Institut National de la Santé et de la Recherche Médicale” (INSERM). The laboratory is associated with the “ligue contre le cancer”, Comité Ile de France. S. Delluc and L. Tourneur are supported by the “Fondation de France” (FDF), Comité Leucémie. S. Delluc received financial support from “France Intergroupe de la Leucémie Myéloïde Chronique” (FI LMC). The “Délégation régionale à la Recherche Clinique” (DRRC), and the “Société Française de Greffe de Moelle et de Thérapie Cellulaire” (SFGM-TC) promoted this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors of submitted papers declare no conflict of interest or financial interest in the product or in potentially competing products held by them, their spouses and/or minor children.
Rights and permissions
About this article
Cite this article
Delluc, S., Tourneur, L., Fradelizi, D. et al. DC-based vaccine loaded with acid-eluted peptides in acute myeloid leukemia: the importance of choosing the best elution method. Cancer Immunol Immunother 56, 1–12 (2007). https://doi.org/10.1007/s00262-006-0170-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-006-0170-6