Abstract
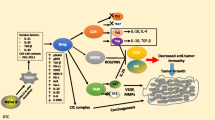
The mouse 4T1 mammary carcinoma is a BALB/c-derived tumor that spontaneously metastasizes and induces immune suppression. Although >95% of wild type BALB/c mice die from metastatic 4T1 tumor even if the primary mammary tumor is surgically removed, >65% of BALB/c mice with a deleted Signal Transducer Activator of Transcription 6 (STAT6) gene survive post-surgery. STAT6-deficiency also confers enhanced immunity against spontaneously developing breast cancer since NeuT+/− mice that are STAT6-deficient develop mammary tumors later and survive longer than NeuT+/− mice that are STAT6-competent. Rejection of metastastic disease and survival of STAT6-deficient mice after removal of primary tumor involve three mechanisms: (1) The generation of M1 type macrophages that produce nitric oxide and are tumoricidal; (2) A decrease to normal in the elevated levels of myeloid suppressor cells that accumulate during primary tumor growth; and (3) CD8+ tumor-specific T lymphocytes. STAT6-deficient, but not wild type BALB/c, mice generate nitric oxide producing macrophages because they lack the STAT6 transcription factor which is necessary for signaling through the type 2 IL-4Rα complex, and which induces the production of arginase instead of nitric oxide.



Similar content being viewed by others
References
Almand B, Clark JI, Nikitina E, van Beynen J, English NR, Knight SC, Carbone DP,Gabrilovich DI (2001) Increased production of immature myeloid cells in cancer patients: a mechanism of immunosuppression in cancer. J Immunol 166:678
Bantounas I, Phylactou LA, Uney JB (2004) RNA interference and the use of small interfering RNA to study gene function in mammalian systems. J Mol Endocrinol 33:545
Boggio K, Nicoletti G, Di Carlo E, Cavallo F, Landuzzi L, Melani C, Giovarelli M, Rossi I, Nanni P, De Giovanni C, Bouchard P, Wolf S, Modesti A, Musiani P, Lollini PL, Colombo MP, Forni G (1998) Interleukin 12-mediated prevention of spontaneous mammary adenocarcinomas in two lines of Her-2/neu transgenic mice. J Exp Med 188:589
Bronte V, Apolloni E, Cabrelle A, Ronca R, Serafini P, Zamboni P, Restifo NP, Zanovello P (2000) Identification of a CD11b(+)/Gr-1(+)/CD31(+) myeloid progenitor capable of activating or suppressing CD8(+) T cells. Blood 96:3838
Bronte V, Serafini P, Apolloni E, Zanovello P (2001) Tumor-induced immune dysfunctions caused by myeloid suppressor cells. J Immunother 24:431
Danna EA, Sinha P, Gilbert M, Clements VK, Pulaski BA, Ostrand-Rosenberg S (2004) Surgical removal of primary tumor reverses tumor-induced immunosuppression despite the presence of metastatic disease. Cancer Res 64:2205
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3:991
Dunn GP, Old LJ, Schreiber RD (2004) The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21:137
Gabrilovich DI, Velders MP, Sotomayor EM, Kast WM (2001) Mechanism of immune dysfunction in cancer mediated by immature Gr-1+ myeloid cells. J Immunol 166:5398
Jensen SM, Meijer SL, Kurt RA, Urba WJ, Hu HM, Fox BA (2003) Regression of a mammary adenocarcinoma in STAT6−/− mice is dependent on the presence of STAT6-reactive T cells. J Immunol 170:2014
Kacha AK, Fallarino F, Markiewicz MA, Gajewski TF (2000) Cutting edge: spontaneous rejection of poorly immunogenic P1.HTR tumors by Stat6-deficient mice. J Immunol 165:6024
Kaplan MH, Schindler U, Smiley ST, Grusby MJ (1996) Stat6 is required for mediating responses to IL-4 and for development of Th2 cells. Immunity 4:313
Kusmartsev S, Gabrilovich DI (2002) Immature myeloid cells and cancer-associated immune suppression. Cancer Immunol Immunother 51:293
Kusmartsev S, Cheng F, Yu B, Nefedova Y, Sotomayor E, Lush R, Gabrilovich D (2003) All-trans-retinoic acid eliminates immature myeloid cells from tumor-bearing mice and improves the effect of vaccination. Cancer Res 63:4441
Kusmartsev S, Gabrilovich DI (2003) Inhibition of myeloid cell differentiation in cancer: the role of reactive oxygen species. J Leukoc Biol 74:186
Liu Y, Van Ginderachter JA, Brys L, De Baetselier P, Raes G, Geldhof AB (2003) Nitric oxide-independent CTL suppression during tumor progression: association with arginase-producing (M2) myeloid cells. J Immunol 170:5064
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23:549
Mazzoni A, Bronte V, Visintin A, Spitzer JH, Apolloni E, Serafini P, Zanovello P, Segal DM (2002) Myeloid suppressor lines inhibit T cell responses by an NO-dependent mechanism. J Immunol 168:689
Miller FR, Miller BE, Heppner GH (1983) Characterization of metastatic heterogeneity among subpopulations of a single mouse mammary tumor: heterogeneity in phenotypic stability. Invasion Metastasis 3:22
Mills CD, Kincaid K, Alt JM, Heilman MJ, Hill AM (2000) M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol 164:6166
Mills CD (2001) Macrophage arginine metabolism to ornithine/urea or nitric oxide/citrulline: a life or death issue. Crit Rev Immunol 21:399
Ostrand-Rosenberg S, Grusby MJ, Clements VK (2000) Cutting edge: STAT6-deficient mice have enhanced tumor immunity to primary and metastatic mammary carcinoma. J Immunol 165:6015
Ostrand-Rosenberg S, Clements VK, Terabe M, Park JM, Berzofsky JA, Dissanayake SK (2002) Resistance to metastatic disease in STAT6-deficient mice requires hemopoietic and nonhemopoietic cells and is IFN-gamma dependent. J Immunol 169:5796
Ostrand-Rosenberg S, Sinha P, Clements V, Dissanayake SI, Miller S, Davis C, Danna E (2004) Signal transducer and activator of transcription 6 (Stat6) and CD1: inhibitors of immunosurveillance against primary tumors and metastatic disease. Cancer Immunol Immunother 53:86
Pulaski BA, Clements VK, Pipeling MR, Ostrand-Rosenberg S (2000a) Immunotherapy with vaccines combining MHC class II/CD80+ tumor cells with interleukin-12 reduces established metastatic disease and stimulates immune effectors and monokine induced by interferon gamma. Cancer Immunol Immunother 49:34
Pulaski BA, Terman DS, Khan S, Muller E, Ostrand-Rosenberg S (2000b) Cooperativity of Staphylococcal aureus enterotoxin B superantigen, major histocompatibility complex class II, and CD80 for immunotherapy of advanced spontaneous metastases in a clinically relevant postoperative mouse breast cancer model. Cancer Res 60:2710
Pulaski BA, Smyth MJ, Ostrand-Rosenberg S (2002) Interferon-gamma-dependent phagocytic cells are a critical component of innate immunity against metastatic mammary carcinoma. Cancer Res 62:4406
Rutschman R, Lang R, Hesse M, Ihle JN, Wynn TA, Murray PJ (2001) Cutting edge: Stat6-dependent substrate depletion regulates nitric oxide production. J Immunol 166:2173
Serafini P, De Santo C, Marigo I, Cingarlini S, Dolcetti L, Gallina G, Zanovello P, Bronte V (2004) Derangement of immune responses by myeloid suppressor cells. Cancer Immunol Immunother 53:64
Shankaran V, Ikeda H, Bruce AT, White JM, Swanson PE, Old LJ, Schreiber RD (2001) IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410:1107
Sinha P, Clements VK, Ostrand-Rosenberg S (2005) Reduction of myeloid-derived suppressor cells and induction of m1 macrophages facilitate the rejection of established metastatic disease. J Immunol 174:636
Terabe M, Matsui S, Noben-Trauth N, Chen H, Watson C, Donaldson DD, Carbone DP, Paul WE, Berzofsky JA (2000) NKT cell-mediated repression of tumor immunosurveillance by IL-13 and the IL-4R-STAT6 pathway. Nat Immunol 1:515
Wei LH, Jacobs AT, Morris SM Jr,Ignarro LJ (2000) IL-4 and IL-13 upregulate arginase I expression by cAMP and JAK/STAT6 pathways in vascular smooth muscle cells. Am J Physiol Cell Physiol 279:C248
Acknowledgements
We thank Ms. Cordula Davis for her help in monitoring tumor progression in the neuT+/− and STAT6−/− neuT+/− mice and Dr. Beth Pulaski for performing the carrageenan experiment. We appreciate the excellent care given to our mouse colony by Ms. Sandra Mason. These studies were supported by NIH grants R01 CA52527 and R01 CA84232, and by U.S. Army Breast Cancer Program Grant DAMD-17-01-1-0312.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is a symposium paper from the conference “Tumor Escape and Its Determinants”, held in Salzburg, Austria, on 10–13 October 2004
Rights and permissions
About this article
Cite this article
Sinha, P., Clements, V.K., Miller, S. et al. Tumor immunity: a balancing act between T cell activation, macrophage activation and tumor-induced immune suppression. Cancer Immunol Immunother 54, 1137–1142 (2005). https://doi.org/10.1007/s00262-005-0703-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-005-0703-4