Abstract
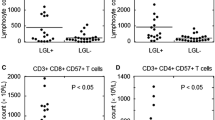
We previously reported that hairy cell leukemia (HCL) patients have high percentages of CD56+/CD57+/CD3+ large granular lymphocytes consistent with cytotoxic T-lymphocytes (CTLs), and other investigators have reported skewing of the T-cell repertoire. In previous studies of up to seven HCL patients, many of the 22 established T-cell receptor (TCR) beta variable region (TRBV) families showed mono- or oligoclonal restriction. To determine whether percentages of CTLs are correlated with TRBV clonal excess, we studied 20 HCL patients with flow cytometry, PCR of TCR gamma and TRBV regions, and fractional gel electrophoresis of PCR-amplified TRBV CDR3 domains (CDR3 spectratyping). Increased percentages of CD3+/CD8+/CD57+ CTLs correlated with more mono/oligoclonal and fewer polyclonal TRBV families (r=0.53; P=0.016). Age correlated with number of mono/oligoclonal TRBV families (r=0.51; P=0.022). Time since last purine analog therapy correlated with number of polyclonal TRBV families (r=0.46; P=0.040), but treatment with the anti-CD22 recombinant immunotoxin BL22 was not related to clonal excess. We conclude that abnormalities in the T-cell repertoire in HCL patients may represent deficient immunity, and may be exacerbated by purine analogs. Increased CD3+/CD57+ T-cells may be a useful marker of abnormal TRBV repertoire in HCL patients, and might prove useful in deciding whether patients should receive biologic antibody-based treatment rather than repeated courses of purine analog for relapsed disease.





Similar content being viewed by others
References
Goodman GR, Bethel KJ, Saven A (2003) Hairy cell leukemia: an update. Curr Opin Hematol 10:258–266
Polliack A (2002) Hairy cell leukemia: biology, clinical diagnosis, unusual manifestations and associated disorders discussion (449–350). Rev Clin Exp Hematol 6:366–388
Robbins BA, Ellison DJ, Spinosa JC, Carey CA, Lukes RJ, Poppema S, Saven A, Piro LD (1993) Diagnostic application of two-color flow cytometry in 161 cases of hairy cell leukemia. Blood 82:1277–1287
Matutes E, Morilla R, Owusu-Ankomah K, Houliham A, Meeus P, Catovsky D (1994) The immunophenotype of hairy cell leukemia (HCL). Proposal for a scoring system to distinguish HCL from B-cell disorders with hairy or villous lymphocytes. Leuk Lymphoma 14(Suppl 1):57–61
Juliusson G, Liliemark J (1992) Rapid recovery from cytopenia in hairy cell leukemia after treatment with 2-chloro-2’-deoxyadenosine (CdA): relation to opportunistic infections. Blood 79:888–894
Hoffman MA, Janson D, Rose E, Rai KR (1997) Treatment of hairy-cell leukemia with cladribine: response, toxicity, and long-term follow-up. J Clin Oncol 15:1138–1142
Blasinska-Morawiec M, Robak T, Krykowski E, Hellmann A, Urbanska-Rys H (1997) Hairy cell leukemia-variant treated with 2-chlorodeoxyadenosine–a report of three cases. Leuk Lymphoma 25:381–385
Matutes E, Wotherspoon A, BritoBabapulle V, Catovsky D (2001) The natural history and clinico-pathological features of the variant form of hairy cell leukemia. Leukemia 15:184–186
Flinn IW, Kopecky KJ, Foucar MK, Head D, Bennett JM, Huchison R, Corbett W, Cassileth P, Habermann T, Golomb H, Rai K, Eisenhauer E, Appelbaum F, Cheson B, Grever MR (2000) Long-term follow-up of remission duration, mortality, and second malignancies in hairy cell leukemia patients treated with pentostatin. Blood 96:2981–2986
Maloisel F, Benboubker L, Gardembas M, Coiffier B, Divine M, Sebban C, Blanc M, Abgrall JF, Lederlin P, Harousseau JL, Blaise AM, Grosbois B, Morice P, Ghandour C, Castaigne S (2003) Long-term outcome with pentostatin treatment in hairy cell leukemia patients. A French retrospective study of 238 patients. Leukemia 17:45–51
Goodman GR, Burian C, Koziol JA, Saven A (2003) Extended follow-up of patients with hairy cell leukemia after treatment with cladribine. J Clin Oncol 21:891–896
Tallman MS, Peterson LC, Hakimian D, Gillis S, Polliack A (1999) Treatment of hairy-cell leukemia: current views. Semin Hematol 36:155–163
Saven A, Burian C, Koziol JA, Piro LD (1998) Long-term follow-up of patients with hairy cell leukemia after cladribine treatment. Blood 92:1918–1926
Cheson BD, Sorensen JM, Vena DA, Montello MJ, Barrett JA, Damasio E, Tallman M, Annino L, Connors J, Coiffier B, Lauria F (1998) Treatment of hairy cell leukemia with 2-chlorodeoxyadenosine via the Group C protocol mechanism of the National Cancer Institute: a report of 979 patients. J Clin Oncol 16:3007–3015
Filleul B, Delannoy A, Ferrant A, Zenebergh A, Van Daele S, Bosly A, Doyen C, Mineur P, Glorieux P, Driesschaert P, Sokal C, Martiat P, Michaux JL (1994) A single course of 2-chloro-deoxyadenosine does not eradicate leukemic cells in hairy cell leukemia patients in complete remission. Leukemia 8:1153–1156
Carbone A, Reato G, Di Celle PF, Lauria F, Foa R (1994) Disease eradication in hairy cell leukemia patients treated with 2- chlorodeoxyadenosine [letter]. Leukemia 8:2019–2020
Seymour JF, Kurzrock R, Freireich EJ, Estey EH (1994) 2-chlorodeoxyadenosine induces durable remissions and prolonged suppression of CD4+ lymphocyte counts in patients with hairy cell leukemia. Blood 83:2906–2911
Seymour JF, Talpaz M, Kurzrock R (1997) Response duration and recovery of CD4+ lymphocytes following deoxycoformycin in interferon-alpha-resistant hairy cell leukemia: 7- year follow-up. Leukemia 11:42–47
Lauria F, Lenoci M, Annino L, Raspadori D, Marotta G, Bocchia M, Forconi F, Gentili S, La Manda M, Marconcini S, Tozzi M, Baldini L, Zinzani PL, Foa R (2001) Efficacy of anti-CD20 monoclonal antibodies (Mabthera) in patients with progressed hairy cell leukemia. Haematologica 86:1046–1050
Hagberg H, Lundholm L (2001) Rituximab, a chimaeric anti-CD20 monoclonal antibody, in the treatment of hairy cell leukaemia. Br J Haematol 115:609–611
Nieva J, Bethel K, Saven A (2003) Phase 2 study of rituximab in the treatment of cladribine-failed patients with hairy cell leukemia. Blood 102:810–813
Thomas DA, O’Brien S, Bueso-Ramos C, Faderl S, Keating MJ, Giles FJ, Cortes J, Kantarjian HM (2003) Rituximab in relapsed or refractory hairy cell leukemia. Blood 102:3906–3911
Kreitman RJ, Squires DR, Stetler-Stevenson M, Noel P, Fitzgerald DJ, Wilson WH, Pastan I (2005) Phase I trial of recombinant immunotoxin RFB4(dsFv)-PE38 (BL22) in patients with B-cell malignancies. J Clin Oncol 23:6719–6729
Kreitman RJ, Wilson WH, Bergeron K, Raggio M, Stetler-Stevenson M, FitzGerald DJ, Pastan I (2001) Efficacy of the Anti-CD22 Recombinant Immunotoxin BL22 in Chemotherapy-Resistant Hairy-Cell Leukemia. New Engl J Med 345:241–247
Ruco LP, Procopio A, Maccallini V, Calogero A, Uccini S, Annino L, Mandelli F, Baroni CD (1983) Severe deficiency of natural killer activity in the peripheral blood of patients with hairy cell leukemia. Blood 61:1132–1137
Smith BR, Rosenthal DS, Ault KA (1985) Natural killer lymphocytes in hairy cell leukemia: presence of phenotypically identifiable cells with defective functional activity. Exp Hematol 13:189–193
Bassan R, Rambaldi A, Allavena P, Abbate M, Marini B, Barbui T (1988) Association of large granular lymphocyte/natural killer cell proliferative disease and second hematologic malignancy. Am J Hematol 29:85–93
Airo P, Rossi G, Facchetti F, Marocolo D, Garza L, Lanfranchi A, Prati E, Brugnoni D, Malacarne F, Cattaneo R (1995) Monoclonal expansion of large granular lymphocytes with a CD4+ CD8dim+/− phenotype associated with hairy cell leukemia. Haematologica 80:146–149
Kluin-Nelemans JC, Kester MG, Melenhorst JJ, Landegent JE, van de Corput L, Willemze R, Falkenburg JH (1996) Persistent clonal excess and skewed T-cell repertoire in T cells from patients with hairy cell leukemia. Blood 87:3795–3802
Kluin-Nelemans HC, Kester MG, van deCorput L, Boor PP, Landegent JE, van Dongen JJ, Willemze R, Falkenburg JH (1998) Correction of abnormal T-cell receptor repertoire during interferon-alpha therapy in patients with hairy cell leukemia. Blood 91:4224–4231
Spaenij-Dekking EH, Van der Meijden ED, Falkenburg JH, Kluin-Nelemans JC (2004) Clonally expanded T cells in hairy cell leukemia patients are not leukemia specific. Leukemia 18:176–178
van de Corput L, Kluin-Nelemans HC, Kester MG, Willemze R, Falkenburg JH (1999) Hairy cell leukemia-specific recognition by multiple autologous HLA-DQ or DP-restricted T-cell clones. Blood 93:251–259
Spaenij-Dekking EH, Van Delft J, Van Der Meijden E, Hiemstra HS, Falkenburg JH, Koning F, Drijfhout JW, Kluin-Nelemans JC (2003) Synaptojanin 2 is recognized by HLA class II-restricted hairy cell leukemia-specific T cells. Leukemia 17:2467–2473
Xie XY, Sorbara L, Kreitman RJ, Fukushima PI, Kingma DW, Stetler-Stevenson M (2000) Development of lymphoproliferative disorder of granular lymphocytes in association with hairy cell leukemia. Leukemia & Lymphoma 37:97–104
McCarthy KP, Sloane JP, Kabarowski JH, Matutes E, Wiedemann LM (1992) A simplified method of detection of clonal rearrangements of the T-cell receptor-gamma chain gene. Diagn Mol Pathol 1:173–179
Nishimura MI, Kawakami Y, Charmley P, O’Neil B, Shilyansky J, Yannelli JR, Rosenberg SA, Hood L (1994) T-cell receptor repertoire in tumor-infiltrating lymphocytes. Analysis of melanoma-specific long-term lines. J Immunother Emphasis Tumor Immunol 16:85–94
Choi YW, Kotzin B, Herron L, Callahan J, Marrack P, Kappler J (1989) Interaction of Staphylococcus aureus toxin “superantigens” with human T cells. Proc Natl Acad Sci USA 86:8941–8945
Pannetier C, Cochet M, Darche S, Casrouge A, Zoller M, Kourilsky P (1993) The sizes of the CDR3 hypervariable regions of the murine T-cell receptor beta chains vary as a function of the recombined germ-line segments. Proc Natl Acad Sci USA 90:4319–4323
Serrano D, Monteiro J, Allen SL, Kolitz J, Schulman P, Lichtman SM, Buchbinder A, Vinciguerra VP, Chiorazzi N, Gregersen PK (1997) Clonal expansion within the CD4+CD57+ and CD8+CD57+ T cell subsets in chronic lymphocytic leukemia. J Immunol 158:1482–1489
Tsutsumi Y, Tanaka J, Miura Y, Toubai T, Kato N, Fujisaw F, Toyoshima N, Ota S, Mori A, Yonezumi M, Chiba K, Kondo T, Hasino S, Kobayasi R, Masauji N, Kasai M, Asaka M, Imamura M (2004) Molecular analysis of T-cell repertoire in patients with graft-versus-host disease after allogeneic stem cell transplantation. Leuk Lymphoma 45:481–488
Van De Corput L, Falkenburg JH, Kluin-Nelemans JC (1998) T-cell dysfunction in hairy cell leukemia: an updated review. Leuk Lymphoma 30:31–39
Rocha B, Tanchot C (2004) CD8 T cell memory. Semin Immunol 16:305–314
Farace F, Orlanducci F, Dietrich PY, Gaudin C, Angevin E, Courtier MH, Bayle C, Hercend T, Triebel F (1994) T cell repertoire in patients with B chronic lymphocytic leukemia. Evidence for multiple in vivo T cell clonal expansions. J Immunol 153:4281–4290
Rezvany MR, Jeddi-Tehrani M, Osterborg A, Kimby E, Wigzell H, Mellstedt H (1999) Oligoclonal TCRBV gene usage in B-cell chronic lymphocytic leukemia: major perturbations are preferentially seen within the CD4 T-cell subset. Blood 94:1063–1069
Alatrakchi N, Farace F, Frau E, Carde P, Munck JN, Triebel F (1998) T-cell clonal expansion in patients with B-cell lymphoproliferative disorders. J Immunother 21:363–370
Brown RD, Yuen E, Nelson M, Gibson J, Joshua D (1997) The prognostic significance of T cell receptor beta gene rearrangements and idiotype-reactive T cells in multiple myeloma. Leukemia 11:1312–1317
Molldrem JJ, LeePP, Wang C, Felio K, Kantarjian HM, Champlin RE, Davis MM (2000) Evidence that specific T lymphocytes may participate in the elimination of chronic myelogenous leukemia. Nat Med 6:1018–1023
Acknowledgments
We thank technicians, nurses, patient care coordinators, and data managers from the Clinical Immunotherapy Section of the Laboratory of Molecular Biology, including Karen Bergeron, Kelly Cahill, Linda Ellison, Sonya Duke, Rita Mincemoyer, Barbara Debrah, and Inger Margulies. This research was supported in part by the Intramural Research Program of the NIH, National Cancer Institute, Center for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arons, E., Sorbara, L., Raffeld, M. et al. Characterization of T-cell repertoire in hairy cell leukemia patients before and after recombinant immunotoxin BL22 therapy. Cancer Immunol Immunother 55, 1100–1110 (2006). https://doi.org/10.1007/s00262-005-0099-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-005-0099-1