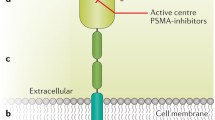
Abstract
Prostate cancer is the second leading cause of cancer-related deaths in men in the United States. Imaging techniques such as CT, MRI, and bone scans have traditionally been used for diagnosis and staging. Molecular imaging modalities targeting the prostate-specific membrane antigen (PSMA) have recently gained attention due to their high affinity and accuracy. PSMA PET has been combined with other modalities such as multiparametric MRI for better diagnostic and prognostic performance. PSMA imaging has been studied at different clinical settings with a wide range of disease aggressiveness. In this review we will explore the role of PSMA PET in high-risk prostate cancer staging, biochemical recurrence, and castration-resistant prostate cancer. The primary focus of this review article is to examine the latest developments in the use of PSMA imaging and emphasize the clinical situations where its effectiveness has been demonstrated to significantly impact the treatment of prostate cancer. In addition, we will touch upon the potential future advancements of PSMA PET imaging and its evolving significance in the management of prostate cancer.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prostate cancer is the most common cancer in men in the United States and second leading cause of cancer-related death in men [1]. CT, MRI and bone scan are the conventionally used imaging techniques for diagnosis, staging and restaging of prostate cancer [2]. Prostate specific membrane antigen (PSMA) is a membrane glycoprotein, expressed 100 to 1000 times higher on prostate cancer cells compared to normal prostate [3]. Imaging agents targeting PSMA are now widely available and have drawn attention due to their high affinity and accuracy in diagnosis, staging, restaging of prostate cancer [4]. Numerous PSMA agents using different radiotracers and imaging modalities such as SPECT and PET have been developed and studied in the literature [5, 6]. PSMA PET has been combined with other modalities such as multiparametric MRI, for a better diagnostic and prognostic performance [5].
The role of PSMA has been studied at different clinical settings with a wide range of disease aggressiveness [4]. Recently, a multidisciplinary panel of healthcare providers and prostate cancer imaging experts developed the appropriate use criteria for PSMA imaging [2]. Patients with newly diagnosed unfavorable intermediate, high, or very-high-risk prostate cancer, biochemical recurrence, and castration-resistant prostate cancer (CRPC) represent the scenarios with the highest scores for PSMA imaging utilization (see Table 1) [2].
This review article focuses on recent advances in the applications of PSMA imaging and highlights the clinical scenarios where its effectiveness has been proven to significantly alter the management of prostate cancer.
PSMA-targeting radioligands
Since its discovery in 1987, multiple iterations of PSMA-targeting molecules for the purpose of diagnosis and treatment of prostate cancer have been developed [7]. For a long time, prostascint (ProstaScint, capromab pendetide; EUSA Pharma) which is a radiolabeled anti PSMA antibody, was the only agent which was FDA approved [8]. Emergence of small molecule PSMA ligands has enabled a more efficient targeting by attaching to an extracellular domain of the PSMA molecule [8]. While PSMA expression tends to increase with aggressiveness, it has been shown to decrease in highly de-differentiated tumors [9]. In addition, higher PSMA uptake is associated with castration resistance and disease progression [3]. Among numerous PSMA-targeting molecules, 68Ga‐PSMA‐11 and 18F-DCFPyL have recently received FDA approval [2]. These new generation PSMA-targeting ligands are made in form of small molecules which clear more rapidly from blood and are more efficient with better tumor to background ratio compared to the older antibody-based counterparts [10]. There is a long list of PSMA-targeting agents which are currently in active clinical trials [11] such as 18F-PSMA-1007, 68Ga-PSMA-617, 68Ga-PSMA-I&T, and 18F-rhPSMA-7.3 [5]. 18F-rhPSMA-7.3 is a radiohybrid radiotracer with high affinity to PSMA and reported lower urinary excretion compared to 68Ga-PSMA-11 and 18F-DCFPyL, with favorable results in the SPOTLIGHT trial [12].
PSMA scan
The European Association of Nuclear Medicine (EANM) and the Society of Nuclear Medicine and Molecular Imaging’s (SNMMI) joint procedure standard for 68Ga-PSMA-11 [13] recommends a 60-minute uptake time with voiding immediately before imaging to minimize bladder uptake. Delayed imaging is recommended in BCR patients with low PSA (< 1ng/mL) [14]. To avoid artifact from residual activity in collecting system, administration of furosemide or hyper hydration has been suggested [15]. Intravenous contrast administration and imaging at urographic phase has also been a solution for delineation of urinary collecting system from tumor burden [16]. Conventionally accepted uptake time for 18F-PSMA compounds range between 60 and 120 minutes [17]. The normal biodistribution of PSMA is seen on Fig. 1. Frequently, either cervical, celiac and/or presacral ganglia can be seen. It is important to know that these are variant of normal biodistribution. Additionally, PSMA shows specific uptake in astrocytes, proximal renal tubules, small intestine, salivary and lacrimal gland [18].
The estimated coefficient for effective dose from PSMA PET ligands ranges between 0.0116 and 0.022 mSv/MBq, resulting in calculated effective doses ranging between 3.4 and 6.6 mSv depending on radioligand type and injection dose [13]. These numbers are comparable to other PET radiotracers such as 18F-choline (0.01mSv/MBq) [19]. Kidneys and lacrimal glands receive the highest absorbed dose in 68Ga-PSMA PET [19]. Radiation exposure from the CT portion of the scan varies and depends on the protocol with effective dose ranging from 1 to 20 mSv [13].
PSMA vs. other compounds
Choline PET
The increased turnover and production of phosphatidyl choline in cellular membranes is the basis of this radiotracer [20]. Choline is either conjugated with 11C or 18F. Studies have shown promising results in patients with BCR despite lower detection rates compared to PSMA PET, with the difference more pronounced on lower PSA levels [21, 22]. Although FDA approved in 2012 for BCR, short half-life of 11C limits availability of this radiotracer to centers with on-site cyclotron. 18F Choline has a longer half-life, enabling wider adoption. This radiotracer was not adopted in the United States due to regulatory issues [4].
Fluciclovine PET
Fluciclovine PET targets amino acid transporters for L-lerucine[4]. Fluciclovine was shown to be at least comparable to PSMA in the earlier studies of patients with advanced disease [23, 24]. It has been FDA approved in 2016 for BCR of prostate cancer. In the more recent clinical trials, Fluciclovine was compared to PSMA in a head-to-head prospective trial of 50 patients with BCR and low PSA (< 2.0 ng/mL) [25]. The detection rate for PSMA was at least twice (56% vs. 26%).
Staging of high-risk prostate cancer
The initial staging of prostate cancer is a crucial step in determining the most appropriate treatment plan, including options such as prostatectomy, radiation therapy with or without local lymph node treatment, or systemic therapy [4] (Fig 2). Risk stratification schemes, based on criteria such as histologic pattern, PSA level, and clinical stage, have been developed to optimize treatment planning [26]. The latest update on cancer center network guideline defines a 5-tier risk stratification method for clinically localized prostate cancer [27]. Imaging is generally not indicated for low and very low-risk prostate cancer, as it is unlikely to extend beyond the prostate [2, 27].
74-year-old with high-risk prostate cancer. Pre-surgical planning 68Ga-PSMA-11 PET/CT performed. Single PSMA avid 2 mm right internal iliac chain lymph node (white arrow in panels a and d; SUVmax 4.5), not well seen on CT (white arrow on panel c). Maximum intensity projection PET a is scaled at 0–5 SUV
The majority of studies examining the role of PSMA in prostate cancer staging have reported favorable specificity and predictive values. Hope et al. [28] in their prospective multicenter single-arm phase 3 trial of 277 patients with high-risk prostate cancer with Ga68-PSMA-11 PET prior to prostatectomy, 27% of patients were positive for pelvic lymph node metastasis with sensitivity of 40%, specificity of 95%, positive predictive value of 75%, and negative predictive value of 81%. The findings of this study formed the foundation for new drug application of 68Ga-PSMA-11. The OSPREY study [29] was a multicenter prospective clinical trial with 18F-DCFPyL that looked at 252 patients undergoing radical prostatectomy, which found a median specificity of 97.9% and a not much favorable sensitivity of 40.3% was achieved for detection of pelvic lymph nodes. The positive predictive value was 86.7% and negative predictive value was 83.2% [29].
Some studies have compared the performance of PSMA PET versus conventional imaging. In the proPSMA study, a multicentric randomized clinical trial of 302 patients aimed at investigating the accuracy of Ga68-PSMA-11 in staging prostate cancer in high-risk patients at initial presentation compared to conventional imaging, PSMA PET showed superior accuracy (92% compared to 65% in conventional imaging), sensitivity (85% versus 38%) [30]. Another study of 160 high-risk prostate cancer patients evaluated with F18-DCFPyL for initial staging, 90% of patients with distant metastasis were correctly identified, where 48% of these patients did not have enlarged lymph node on CT [31]. Nearly all the patients with evidence of metastasis on CT, were found to have additional sites on F18-DCFPyL [31]. Based on the results of these studies, PSMA PET appears to be superior to conventional imaging [32, 33] and appropriate for initial staging of the unfavorable intermediate, high, or very-high-risk prostate cancer [2]. A meta-analysis that evaluated 257 patients for initial evaluation of prostate cancer (primary and metastatic lymph node combined) in a per-lesion analysis, found sensitivity of 83% and specificity of 81% in diagnosis of primary prostate cancer [34].
Biochemical recurrence
Biochemical recurrence occurs when the serum PSA level increases to a specific threshold following definitive therapy. In radiation therapy, this threshold is more than 2.0 ng/mL above nadir, while in radical prostatectomy, it is 0.2 ng/mL on two consecutive reads [35, 36]. PSA recurrence after definitive therapy is common [37] with about 50% of patients experiencing PSA recurrence within 10 years (Figs. 3 and 4 ) [37].
a–c Example of single PSMA lymph node disease in a 54-year-old with recent resection for prostate cancer with persistently elevated PSA. F18-DCFPyL PET/CT showed a single focus of PSMA uptake in right pelvis (panel a and b, SUVmax 4.4) corresponding to a 4 mm right pelvic side wall lymph node (panel c). Original preprostatectomy pathology showed up to Gleason 4+5 prostate adenocarcinoma in left prostate lobe and 10/17 cores were positive for cancer. Uptake in the left pelvis corresponds to ureteric activity. Maximum intensity projection view a SUV scaled at 0–5
Early detection of biochemical recurrence (BCR) may provide a window of opportunity for effective treatment with directed radiation therapy and/or surgery before the onset of clinical recurrence. For example, salvage radiation therapy which has been shown to be the most effective treatment plan after radical prostatectomy and BCR, is usually done without conventional imaging, potentially missing distant lesions [38]. Therefore, a sensitive imaging technique to detect disease out of expected radiation field for optimal treatment is critical for management of BCR [4]. In a recent post hoc analysis involving 270 patients, PSMA PET imaging was found to alter patient management in 19% of cases [39]. As a result, a clinical trial by Calais et al. is currently investigating the potential value of PSMA PET imaging in radiation planning for these patients [40].
There is a large body of evidence supporting use of PSMA PET in BCR [39, 41,42,43,44,45,46,47,48,49,50]. Hope et al. in their meta-analysis of 256 patients, found sensitivity of 99%, specificity of 76% in pelvic lymph nodes in patients with BCR using pathologic correlate as gold standard [51]. In a more recent systematic review and meta-analysis, Pozdnyakov et al. [50] found that PSMA PET altered treatment plans in 56.4% of the population from 34 studies (3680 patients) and resulted in BCR-free survival of 60.2% at median follow up of 20 months for 1057 patients. In another meta-analysis of 4790 patients [52], Ga68-PSMA PET was found to improve detection rates with rising PSA levels from 33% for PSA levels below 0.19 ng/mL and to 95% for PSA levels > 2.0 ng/mL. This study found high sensitivity and specificity in detection of nodal metastasis in BCR patients (75% and 99%, respectively) comparable with conventional imaging [32, 52]. PSMA PET was able to predict three-year freedom from disease progression in the multicenter clinical trial on prostate cancer patients with BCR undergoing salvage radiotherapy [44]. PSMA is now incorporated in biochemical recurrence guidelines of American Urology Association Radiographic Assessments for Detection of Advanced Recurrence (RADAR III) consensus group [53], European association of urology [54] and the NCCN guidelines for prostate cancer [55].
Castration-resistant prostate cancer and PSMA-targeted treatment of prostate cancer
Castration resistance in prostate cancer occurs when the disease continues to progress, even in the presence of low levels of prostate-specific antigen (PSA) and despite treatment with androgen deprivation therapy. Distinguishing between metastatic and nonmetastatic castration-resistant prostate cancer (CRPC) is crucial for determining the appropriate treatment plan [2]. PSMA PET imaging is highly effective in accurately evaluating the extent of disease in both patient groups. In a multicentric retrospective study of 200 patients with CRPC, PSMA PET was able to detect metastatic disease in 55% of patients who were designated as M0 based on conventional imaging [56]. Neuroendocrine differentiation, a rare but increasingly aggressive subtype of prostate cancer, can be observed in the advanced stages of CRPC [57]. However, recent advancements in PSMA-targeted treatments for prostate cancer have demonstrated promise in effectively treating these types of prostate cancers.
The life prolonging treatments for CRPC are diverse and include chemotherapy, hormonal therapy, immunotherapy and radioligand therapy (Fig 5). With recent approval from the food and drug administration on 177Lu-PSMA-617 (Pluvicto, 177Lu-vipivotide tetraxetan; Novartis [Basel, Switzerland]/Advanced Accelerator Applications USA, Inc. [Millburn, NJ]) based on the findings of clinical trials showing effectiveness of radiopharmaceutical therapy (RPT) [58, 59], PSMA imaging workgroup has recently updated the AUC to recommend PSMA imaging for evaluation of eligibility for patients being considered for PSMA-targeted RPT [60]. A recently published procedure guideline by EANM/SNMMI outlines the expert recommendations for patient selection, treatment protocol and management of side effects [61].
a–e 73-year-old with castration-resistant metastatic prostate cancer initially treated with RT and ADT in 2004, bone metastasis in 2019, treated with chemotherapy and RT to multiple lesions, started on 177Lu-PSMA treatment given recurrent disease and rise in PSA after baseline 18F-DCFPyL PET/CT (a). Subsequent post therapy whole-body uptake images (b–e) demonstrate gradual decrease in conspicuity of the osseous lesions, corresponding to interval treatment of the bone metastases. Overall decline in PSA levels was also noted during treatment (f). Maximum intensity projection 18F-DCFPyL PET a is scaled at 0–10 SUV
Intraprostatic localization and pre biopsy evaluation
Tumor localization within the prostate can be helpful in biopsy targeting, targeted therapy and potentially eliminating the need for biopsy in patients with clinically insignificant prostate cancer. For the latter aim, PRIMARY trial [62] has been conducted which assesses the potential added value of PSMA PET to mpMRI in intraprostatic localization and possibly elimination of biopsies. A total of 296 patients with suspected prostate cancer without prior MRI or tissue sampling were included, then underwent MRI, biopsy and PSMA PET imaging. Combination of PSMA+MRI improved the negative predictive value to 91% versus 72% for MRI, improved sensitivity to 97% versus 83% while specificity decreased to 40% versus 53% [62]. The authors concluded 19% of patients could have potentially avoided biopsy by only risking 3.1% of patients with clinically significant prostate cancer. However, the MRI read at this study was not central and was comparable to similar studies with multiple central readers [63]. Therefore, further validation with larger sample size and potential incorporation of multivariate risk calculators would be warranted [64].
PSMA in cancers other than prostate
Despite the name, PSMA is not specifically expressed on prostate and a wide range of other cancers have PSMA uptake including renal cell carcinoma, transitional cell carcinoma, primary brain tumors, thyroid carcinoma, breast cancer, hepatocellular carcinoma, and lung (Fig. 6) [65]. Perry et al. retrospectively reviewed 1445 prostate cancer patients with atypical findings on 18F-DCFPyL PSMA PET and found non-prostate cancer tumors only in 1.2% of them with nearly all non-prostate cancer tumors showing no or low PSMA uptake, except renal cell carcinoma [66].
a-c 74-year-old man with a PSA 8.1, Gleason 4+3 prostate cancer. Staging Ga68-PSMA-11 PET findings consistent with T3bN1bMx disease and spiculated 2.6 cm right upper lobe nodule with low radiotracer uptake (SUVmax 5.8) on a background of emphysema (white arrow in panels a–c). Subsequent FNA was consistent with SCC of lung. There is increased uptake within the prostate with known cancer and metastatic pelvic lymph nodes. Several foci of low-level radiotracer uptake without CT correlate in the posterior left sixth rib without a CT or subsequent FDG correlate, favored to be benign. Maximum intensity projection a SUV scale 0–5
Limitations of PSMA
Although PSMA-targeted imaging has been very effective in detection of PSMA expressing tumors, prostate cancers with neuroendocrine differentiation do not express PSMA due to FOLH1 gene suppression and thus, making this imaging technique ineffective [9]. Alternative molecular imaging targets including Fibroblast Activating Protein (FAP), Somatostatin receptor type 2, gastrin-releasing peptide receptor and FDG have been proposed as alternative diagnostic tests in these aggressive tumors [9, 67,68,69]. However, further studies are needed to show which, if any, of these alternatives are effective for imaging this subtype.
Androgen blockade is an integral part of the several treatment options for prostate cancer due to its significant role in improving survival rate and treatment effectiveness [70]. PSMA expression on prostate cancer cells is heavily modulated by treatments that alter androgen receptor [71, 72]. This response heterogeneity has implications for upcoming imaging and therapeutic interventions and differs depending on the tumor phenotype (hormone sensitive vs. CRPC) and the timing after treatment [71, 72].
Determination of malignancy or benignity of solitary bone lesions can be challenging in PSMA PET as this could change patient management and false positive interpretations would cause unnecessary harm to the patient [73]. Solitary rib lesions are common and mostly due to benign causes such as fracture or benign etiologies such as fibrous dysplasia (Fig 7). In a retrospective study of patients with prostate cancer, 62 patients with solitary Ga68-PSMA PET rib lesions were selected and their malignant potential was determined based on imaging follow up, PSA level and biopsy [73]. With these criteria, only one patient had a false negative finding of a rib lesion which resulted in worsening of metastatic disease and majority of lesions were classified as benign without need for follow up [73]. The findings of a recent study involving 48 high-risk primary or recurrent prostate cancer patients suggest that indeterminate 18F-DCFPyL PSMA PET bone lesions with a standardized uptake value maximum (SUVmax) exceeding 5 and coexisting bone metastasis in other areas are likely to be malignant, whereas lesions with SUVmax below 5 and no concurrent suspicious findings are more likely to be benign [74].
Future directives
PSMA PET imaging in metastatic-directed therapy of oligometastatic prostate cancer
The utilization of sensitive prostate-specific antigen (PSA) detection methods and improved PET radiotracers has resulted in an increased identification of “oligometastatic disease,” thereby raising interest in metastasis-directed therapy (MDT) [75, 76] (Fig 8). Emerging evidence indicates that localized MDT may defer disease progression, prolong the time before systemic therapies become necessary, and mitigate-associated toxicities [75]. Multiple studies have studied the role of PSMA PET in treatment guidance of the oligometastatic prostate cancer with findings favoring improved progression-free survival [77]. A challenge in evaluation of the effectiveness of new more sensitive PSMA-directed oligometastatic disease treatment is the stage migration caused by improved sensitivity of these new techniques compared to conventional imaging which will limits use of historical data from previously performed trials [76].
a–c Example of oligometastatic osseous disease in a 68-year-old man with history prostatectomy now presenting with biochemical recurrence and PSA of 1.3 ng/mL. 68 Ga-PSMA-11 PET/CT showed a single focus of PSMA uptake (white arrow in panels a and b, SUVmax 4) corresponding to a small sclerotic focus in the T8 vertebral body (white arrow in panel c). Activity below bladder corresponds to urinary contamination. Maximum intensity projection a SUV scaled at 0–5
Assessment of response to treatment of CRPC using 177Lu-PSMA
In addition to its predictive role in determining the degree of response to 177Lu-PSMA therapy and aiding in patient selection, PSMA PET may also be utilized for evaluating treatment response [61]. The LuPIN trial investigated the response to 177Lu-PSMA-617 therapy and radiation sensitizer NOX66 in 37 patients with castration-resistant prostate cancer (CRPC). This prospective study employed 68Ga-PSMA and 18F-FDG PET scans both before and after treatment [78]. The study identified quantitative PSMA total tumor volume and PSA progression as the only two independently prognostic variables for overall survival among other variables [78]. Further studies with larger patient populations would be beneficial in further evaluating the role of post treatment PSMA PET as an imaging biomarker.
Reporting frameworks for PSMA PET
PROMISE is a molecular imaging TNM staging system which is proposed to facilitate reporting and study design of prostate cancer molecular imaging [79, 80]. Under this system, local disease is categorized into T stages, ranging from miT0 (no local tumor) to miT4 (tumor invades structures other than seminal vesicles) [79]. Regional nodal disease is assigned N stages, ranging from miN0 (no regional lymph nodes) to miN2 (multiple pelvic nodal regions affected) [79]. Metastatic disease is classified with an M stage, which spans from miM0 (no distant metastases) to miM1 (distant metastases), further categorized as diss (disseminated), dmi (diffuse marrow involvement), oligo (oligiometastatic), and uni (unifocal) [79]. PSMA uptake levels are assessed using a visual scoring system based on the mean uptake in the blood pool, liver, and parotid gland, and the results are reported as 0, 1, 2, or 3, indicating no, low, intermediate, or high PSMA expression, respectively [79]. The recently introduced PROMISE V2 [80] comes with two hierarchical levels of assessment including an updated TNM staging schema and reporting of PSMA expression score which is now integrated with PRIMARY score [80]. PRIMARY score is a five-point scoring system, validated for detection of clinically significant prostate cancer in biopsy naïve patients [62]. PROMISE V2 has new recommendations for reporting sequential studies, lesion distribution and tumor volume assessment. These new changes facilitate use of specific PSMA response criteria such as PSMA PET Progression criteria (PPP) which focuses on a single lesion or Response Evaluation Criteria in PSMA-PET/CT (RECIP) which mostly focuses on total tumor volume in patients with extensive disease [62].
Prostate-specific Membrane Antigen-Reporting and Data System (PSMA-RADS) is a proposed reporting and data system with ultimate goal of establishing a uniform and structured reporting system for PSMA-targeted PET studies [81]. This framework assigns a PSMA-RADS value of 1–5 to both individual lesions and the overall scan. A value of 1 indicates a normal scan or a known benign lesion based on pathological examination or pathognomonic imaging. A value of 2 suggests a likely benign lesion, although biopsy results may be unavailable, or imaging findings may not be pathognomonic. Category 3 is multifaceted and includes lesions that are indeterminate for prostate cancer or suspicious for a non-prostate malignancy. A value of 4 indicates a likely prostate cancer, while 5 signifies almost certain prostate cancer with a classic imaging appearance [81]. The aim of this framework is to reflect the level of confidence of the clinician in presence of prostate cancer and clarify the need for further workup. Different categories of PSMA-RADS specifically category 3 which is reflects more equivocal findings, has been validated in recent studies [82,83,84].
Concluding remarks
PSMA PET is considered a valuable tool for various aspects of patient care in the clinical management of prostate cancer and has shown promising results in initial staging of prostate cancer, localization of recurrent or persistent disease, and staging before PSMA-directed radioligand therapy. It also has potential applications in guiding prostate biopsy, guiding metastatic-directed therapy, and monitoring systemic and radioligand treatment response. While the impact on patient outcomes and management is still being assessed, PSMA PET has been included in clinical guidelines and consensus documents, highlighting its superior accuracy and additional value in prostate cancer staging. However, further research and evaluation are needed to fully establish its role in treatment monitoring and patient outcomes.
References
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA: A Cancer Journal for Clinicians. 2023;73(1):17-48. doi: https://doi.org/10.3322/caac.21763.
Jadvar H, Calais J, Fanti S, Feng F, Greene KL, Gulley JL, et al. Appropriate Use Criteria for Prostate-Specific Membrane Antigen PET Imaging. J Nucl Med. 2022;63(1):59-68. doi: https://doi.org/10.2967/jnumed.121.263262.
Ristau BT, O'Keefe DS, Bacich DJ. The prostate-specific membrane antigen: lessons and current clinical implications from 20 years of research. Urol Oncol. 2014;32(3):272-9. doi: https://doi.org/10.1016/j.urolonc.2013.09.003.
Lawhn-Heath C, Salavati A, Behr SC, Rowe SP, Calais J, Fendler WP, et al. Prostate-specific Membrane Antigen PET in Prostate Cancer. Radiology. 2021;299(2):248-60. doi: https://doi.org/10.1148/radiol.2021202771.
Farolfi A, Calderoni L, Mattana F, Mei R, Telo S, Fanti S, et al. Current and Emerging Clinical Applications of PSMA PET Diagnostic Imaging for Prostate Cancer. J Nucl Med. 2021;62(5):596-604. doi: https://doi.org/10.2967/jnumed.120.257238.
Walker SM, Lim I, Lindenberg L, Mena E, Choyke PL, Turkbey B. Positron emission tomography (PET) radiotracers for prostate cancer imaging. Abdom Radiol (NY). 2020;45(7):2165-75. doi: https://doi.org/10.1007/s00261-020-02427-4.
Horoszewicz JS, Kawinski E, Murphy GP. Monoclonal antibodies to a new antigenic marker in epithelial prostatic cells and serum of prostatic cancer patients. Anticancer Res. 1987;7(5B):927-35.
Schwarzenboeck SM, Rauscher I, Bluemel C, Fendler WP, Rowe SP, Pomper MG, et al. PSMA Ligands for PET Imaging of Prostate Cancer. J Nucl Med. 2017;58(10):1545-52. doi: https://doi.org/10.2967/jnumed.117.191031.
Bakht MK, Derecichei I, Li Y, Ferraiuolo RM, Dunning M, Oh SW, et al. Neuroendocrine differentiation of prostate cancer leads to PSMA suppression. Endocr Relat Cancer. 2018;26(2):131-46. doi: https://doi.org/10.1530/ERC-18-0226.
Hope TA, Afshar-Oromieh A, Eiber M, Emmett L, Fendler WP, Lawhn-Heath C, et al. Imaging Prostate Cancer With Prostate-Specific Membrane Antigen PET/CT and PET/MRI: Current and Future Applications. AJR Am J Roentgenol. 2018;211(2):286-94. doi: https://doi.org/10.2214/AJR.18.19957.
Meher N, VanBrocklin HF, Wilson DM, Flavell RR. PSMA-Targeted Nanotheranostics for Imaging and Radiotherapy of Prostate Cancer. Pharmaceuticals (Basel). 2023;16(2). doi: https://doi.org/10.3390/ph16020315.
Jani AB, Ravizzini GC, Gartrell BA, Siegel BA, Twardowski P, Saltzstein D, et al. Diagnostic Performance and Safety of (18)F-rhPSMA-7.3 Positron Emission Tomography in Men With Suspected Prostate Cancer Recurrence: Results From a Phase 3, Prospective, Multicenter Study (SPOTLIGHT). J Urol. 2023:101097ju0000000000003493. doi: https://doi.org/10.1097/ju.0000000000003493.
Fendler WP, Eiber M, Beheshti M, Bomanji J, Calais J, Ceci F, et al. PSMA PET/CT: joint EANM procedure guideline/SNMMI procedure standard for prostate cancer imaging 2.0. Eur J Nucl Med Mol Imaging. 2023;50(5):1466-86. doi: https://doi.org/10.1007/s00259-022-06089-w.
Beheshti M, Manafi-Farid R, Geinitz H, Vali R, Loidl W, Mottaghy FM, et al. Multiphasic (68)Ga-PSMA PET/CT in the Detection of Early Recurrence in Prostate Cancer Patients with a PSA Level of Less Than 1 ng/mL: A Prospective Study of 135 Patients. J Nucl Med. 2020;61(10):1484-90. doi: https://doi.org/10.2967/jnumed.119.238071.
Lawhn-Heath C, Flavell RR, Korenchan DE, Deller T, Lake S, Carroll PR, et al. Scatter Artifact with Ga-68-PSMA-11 PET: Severity Reduced With Furosemide Diuresis and Improved Scatter Correction. Mol Imaging. 2018;17:1536012118811741. doi: https://doi.org/10.1177/1536012118811741.
Iravani A, Hofman MS, Mulcahy T, Williams S, Murphy D, Parameswaran BK, et al. (68)Ga PSMA-11 PET with CT urography protocol in the initial staging and biochemical relapse of prostate cancer. Cancer Imaging. 2017;17(1):31. doi: https://doi.org/10.1186/s40644-017-0133-5.
Rahbar K, Afshar-Oromieh A, Seifert R, Wagner S, Schafers M, Bogemann M, et al. Diagnostic performance of (18)F-PSMA-1007 PET/CT in patients with biochemical recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2018;45(12):2055-61. doi: https://doi.org/10.1007/s00259-018-4089-x.
Prasad V, Steffen IG, Diederichs G, Makowski MR, Wust P, Brenner W. Biodistribution of [(68)Ga]PSMA-HBED-CC in Patients with Prostate Cancer: Characterization of Uptake in Normal Organs and Tumour Lesions. Mol Imaging Biol. 2016;18(3):428-36. doi: https://doi.org/10.1007/s11307-016-0945-x.
Sandgren K, Johansson L, Axelsson J, Jonsson J, Ögren M, Ögren M, et al. Radiation dosimetry of [(68)Ga]PSMA-11 in low-risk prostate cancer patients. EJNMMI Phys. 2019;6(1):2. doi: https://doi.org/10.1186/s40658-018-0239-2.
Ghafoor S, Burger IA, Vargas AH. Multimodality Imaging of Prostate Cancer. J Nucl Med. 2019;60(10):1350-8. doi: https://doi.org/10.2967/jnumed.119.228320.
Treglia G, Pereira Mestre R, Ferrari M, Bosetti DG, Pascale M, Oikonomou E, et al. Radiolabelled choline versus PSMA PET/CT in prostate cancer restaging: a meta-analysis. Am J Nucl Med Mol Imaging. 2019;9(2):127-39.
Wang R, Shen G, Huang M, Tian R. The Diagnostic Role of (18)F-Choline, (18)F-Fluciclovine and (18)F-PSMA PET/CT in the Detection of Prostate Cancer With Biochemical Recurrence: A Meta-Analysis. Front Oncol. 2021;11:684629. doi: https://doi.org/10.3389/fonc.2021.684629.
Pernthaler B, Kulnik R, Gstettner C, Salamon S, Aigner RM, Kvaternik H. A Prospective Head-to-Head Comparison of 18F-Fluciclovine With 68Ga-PSMA-11 in Biochemical Recurrence of Prostate Cancer in PET/CT. Clin Nucl Med. 2019;44(10):e566-e73. doi: https://doi.org/10.1097/RLU.0000000000002703.
Schuster DM, Savir-Baruch B, Nieh PT, Master VA, Halkar RK, Rossi PJ, et al. Detection of recurrent prostate carcinoma with anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid PET/CT and 111In-capromab pendetide SPECT/CT. Radiology. 2011;259(3):852-61. doi: https://doi.org/10.1148/radiol.11102023.
Calais J, Ceci F, Eiber M, Hope TA, Hofman MS, Rischpler C, et al. (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: a prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol. 2019;20(9):1286-94. doi: https://doi.org/10.1016/S1470-2045(19)30415-2.
Litwin MS, Tan HJ. The Diagnosis and Treatment of Prostate Cancer: A Review. JAMA. 2017;317(24):2532-42. doi: https://doi.org/10.1001/jama.2017.7248.
Mohler JL, Antonarakis ES, Armstrong AJ, D'Amico AV, Davis BJ, Dorff T, et al. Prostate Cancer, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2019;17(5):479-505. doi: https://doi.org/10.6004/jnccn.2019.0023.
Hope TA, Eiber M, Armstrong WR, Juarez R, Murthy V, Lawhn-Heath C, et al. Diagnostic Accuracy of 68Ga-PSMA-11 PET for Pelvic Nodal Metastasis Detection Prior to Radical Prostatectomy and Pelvic Lymph Node Dissection: A Multicenter Prospective Phase 3 Imaging Trial. JAMA Oncol. 2021;7(11):1635-42. doi: https://doi.org/10.1001/jamaoncol.2021.3771.
Pienta KJ, Gorin MA, Rowe SP, Carroll PR, Pouliot F, Probst S, et al. A Phase 2/3 Prospective Multicenter Study of the Diagnostic Accuracy of Prostate Specific Membrane Antigen PET/CT with (18)F-DCFPyL in Prostate Cancer Patients (OSPREY). J Urol. 2021;206(1):52-61. doi: https://doi.org/10.1097/JU.0000000000001698.
Hofman MS, Lawrentschuk N, Francis RJ, Tang C, Vela I, Thomas P, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395(10231):1208-16. doi: https://doi.org/10.1016/S0140-6736(20)30314-7.
Wondergem M, van der Zant FM, Broos WAM, Roeleveld TA, Donker R, Ten Oever D, et al. (18)F-DCFPyL PET/CT for primary staging in 160 high-risk prostate cancer patients; metastasis detection rate, influence on clinical management and preliminary results of treatment efficacy. Eur J Nucl Med Mol Imaging. 2021;48(2):521-31. doi: https://doi.org/10.1007/s00259-020-04782-2.
Hovels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL, et al. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol. 2008;63(4):387-95. doi: https://doi.org/10.1016/j.crad.2007.05.022.
Woo S, Suh CH, Kim SY, Cho JY, Kim SH. The Diagnostic Performance of MRI for Detection of Lymph Node Metastasis in Bladder and Prostate Cancer: An Updated Systematic Review and Diagnostic Meta-Analysis. AJR Am J Roentgenol. 2018;210(3):W95-W109. doi: https://doi.org/10.2214/AJR.17.18481.
Wang R, Shen G, Yang R, Ma X, Tian R. (68)Ga-PSMA PET/MRI for the diagnosis of primary and biochemically recurrent prostate cancer: A meta-analysis. Eur J Radiol. 2020;130:109131. doi: https://doi.org/10.1016/j.ejrad.2020.109131.
Roach M, 3rd, Hanks G, Thames H, Jr., Schellhammer P, Shipley WU, Sokol GH, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int J Radiat Oncol Biol Phys. 2006;65(4):965-74. doi: https://doi.org/10.1016/j.ijrobp.2006.04.029.
Cookson MS, Aus G, Burnett AL, Canby-Hagino ED, D'Amico AV, Dmochowski RR, et al. Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol. 2007;177(2):540-5. doi: https://doi.org/10.1016/j.juro.2006.10.097.
Kupelian PA, Mahadevan A, Reddy CA, Reuther AM, Klein EA. Use of different definitions of biochemical failure after external beam radiotherapy changes conclusions about relative treatment efficacy for localized prostate cancer. Urology. 2006;68(3):593-8. doi: https://doi.org/10.1016/j.urology.2006.03.075.
Briganti A, Karnes RJ, Joniau S, Boorjian SA, Cozzarini C, Gandaglia G, et al. Prediction of outcome following early salvage radiotherapy among patients with biochemical recurrence after radical prostatectomy. Eur Urol. 2014;66(3):479-86. doi: https://doi.org/10.1016/j.eururo.2013.11.045.
Calais J, Czernin J, Cao M, Kishan AU, Hegde JV, Shaverdian N, et al. (68)Ga-PSMA-11 PET/CT Mapping of Prostate Cancer Biochemical Recurrence After Radical Prostatectomy in 270 Patients with a PSA Level of Less Than 1.0 ng/mL: Impact on Salvage Radiotherapy Planning. J Nucl Med. 2018;59(2):230-7. doi: https://doi.org/10.2967/jnumed.117.201749.
Calais J, Czernin J, Fendler WP, Elashoff D, Nickols NG. Randomized prospective phase III trial of (68)Ga-PSMA-11 PET/CT molecular imaging for prostate cancer salvage radiotherapy planning [PSMA-SRT]. BMC Cancer. 2019;19(1):18. doi: https://doi.org/10.1186/s12885-018-5200-1.
Fendler WP, Calais J, Eiber M, Flavell RR, Mishoe A, Feng FY, et al. Assessment of 68Ga-PSMA-11 PET Accuracy in Localizing Recurrent Prostate Cancer: A Prospective Single-Arm Clinical Trial. JAMA Oncol. 2019;5(6):856-63. doi: https://doi.org/10.1001/jamaoncol.2019.0096.
Lawhn-Heath C, Flavell RR, Behr SC, Yohannan T, Greene KL, Feng F, et al. Single-Center Prospective Evaluation of (68)Ga-PSMA-11 PET in Biochemical Recurrence of Prostate Cancer. AJR Am J Roentgenol. 2019;213(2):266-74. doi: https://doi.org/10.2214/AJR.18.20699.
Hope TA, Aggarwal R, Chee B, Tao D, Greene KL, Cooperberg MR, et al. Impact of (68)Ga-PSMA-11 PET on Management in Patients with Biochemically Recurrent Prostate Cancer. J Nucl Med. 2017;58(12):1956-61. doi: https://doi.org/10.2967/jnumed.117.192476.
Emmett L, Tang R, Nandurkar R, Hruby G, Roach P, Watts JA, et al. 3-Year Freedom from Progression After (68)Ga-PSMA PET/CT-Triaged Management in Men with Biochemical Recurrence After Radical Prostatectomy: Results of a Prospective Multicenter Trial. J Nucl Med. 2020;61(6):866-72. doi: https://doi.org/10.2967/jnumed.119.235028.
Sonni I, Eiber M, Fendler WP, Alano RM, Vangala SS, Kishan AU, et al. Impact of (68)Ga-PSMA-11 PET/CT on Staging and Management of Prostate Cancer Patients in Various Clinical Settings: A Prospective Single-Center Study. J Nucl Med. 2020;61(8):1153-60. doi: https://doi.org/10.2967/jnumed.119.237602.
Santos A, Mattiolli A, Carvalheira JB, Ferreira U, Camacho M, Silva C, et al. PSMA whole-body tumor burden in primary staging and biochemical recurrence of prostate cancer. Eur J Nucl Med Mol Imaging. 2021;48(2):493-500. doi: https://doi.org/10.1007/s00259-020-04981-x.
Afshar-Oromieh A, da Cunha ML, Wagner J, Haberkorn U, Debus N, Weber W, et al. Performance of [(68)Ga]Ga-PSMA-11 PET/CT in patients with recurrent prostate cancer after prostatectomy-a multi-centre evaluation of 2533 patients. Eur J Nucl Med Mol Imaging. 2021;48(9):2925-34. doi: https://doi.org/10.1007/s00259-021-05189-3.
Han S, Woo S, Kim YJ, Suh CH. Impact of (68)Ga-PSMA PET on the Management of Patients with Prostate Cancer: A Systematic Review and Meta-analysis. Eur Urol. 2018;74(2):179-90. doi: https://doi.org/10.1016/j.eururo.2018.03.030.
Kimura S, Abufaraj M, Janisch F, Iwata T, Parizi MK, Foerster B, et al. Performance of [(68)Ga] Ga-PSMA 11 PET for detecting prostate cancer in the lymph nodes before salvage lymph node dissection: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2020;23(1):1-10. doi: https://doi.org/10.1038/s41391-019-0156-z.
Pozdnyakov A, Kulanthaivelu R, Bauman G, Ortega C, Veit-Haibach P, Metser U. The impact of PSMA PET on the treatment and outcomes of men with biochemical recurrence of prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2022. doi: https://doi.org/10.1038/s41391-022-00544-3.
Hope TA, Goodman JZ, Allen IE, Calais J, Fendler WP, Carroll PR. Metaanalysis of (68)Ga-PSMA-11 PET Accuracy for the Detection of Prostate Cancer Validated by Histopathology. J Nucl Med. 2019;60(6):786-93. doi: https://doi.org/10.2967/jnumed.118.219501.
Perera M, Papa N, Roberts M, Williams M, Udovicich C, Vela I, et al. Gallium-68 Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer-Updated Diagnostic Utility, Sensitivity, Specificity, and Distribution of Prostate-specific Membrane Antigen-avid Lesions: A Systematic Review and Meta-analysis. Eur Urol. 2020;77(4):403-17. doi: https://doi.org/10.1016/j.eururo.2019.01.049.
Crawford ED, Koo PJ, Shore N, Slovin SF, Concepcion RS, Freedland SJ, et al. A Clinician's Guide to Next Generation Imaging in Patients With Advanced Prostate Cancer (RADAR III). J Urol. 2019;201(4):682-92. doi: https://doi.org/10.1016/j.juro.2018.05.164.
Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021;79(2):243-62. doi: https://doi.org/10.1016/j.eururo.2020.09.042.
Schaeffer EM, Srinivas S, Adra N, An Y, Barocas D, Bitting R, et al. NCCN Guidelines® Insights: Prostate Cancer, Version 1.2023. J Natl Compr Canc Netw. 2022;20(12):1288-98. doi: https://doi.org/10.6004/jnccn.2022.0063.
Fendler WP, Weber M, Iravani A, Hofman MS, Calais J, Czernin J, et al. Prostate-Specific Membrane Antigen Ligand Positron Emission Tomography in Men with Nonmetastatic Castration-Resistant Prostate Cancer. Clin Cancer Res. 2019;25(24):7448-54. doi: https://doi.org/10.1158/1078-0432.CCR-19-1050.
Kemble J, Kwon ED, Karnes RJ. Addressing the need for more therapeutic options in neuroendocrine prostate cancer. Expert Rev Anticancer Ther. 2023;23(2):177-85. doi: https://doi.org/10.1080/14737140.2023.2173174.
Hofman MS, Emmett L, Sandhu S, Iravani A, Joshua AM, Goh JC, et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397(10276):797-804. doi: https://doi.org/10.1016/s0140-6736(21)00237-3.
Sartor O, de Bono J, Chi KN, Fizazi K, Herrmann K, Rahbar K, et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2021;385(12):1091-103. doi: https://doi.org/10.1056/NEJMoa2107322.
Hope TA, Jadvar H. Updates to Appropriate Use Criteria for PSMA PET. J Nucl Med. 2022;63(5):14N.
Kratochwil C, Fendler WP, Eiber M, Hofman MS, Emmett L, Calais J, et al. Joint EANM/SNMMI procedure guideline for the use of 177Lu-labeled PSMA-targeted radioligand-therapy (177Lu-PSMA-RLT). European Journal of Nuclear Medicine and Molecular Imaging. 2023. doi: https://doi.org/10.1007/s00259-023-06255-8.
Emmett L, Buteau J, Papa N, Moon D, Thompson J, Roberts MJ, et al. The Additive Diagnostic Value of Prostate-specific Membrane Antigen Positron Emission Tomography Computed Tomography to Multiparametric Magnetic Resonance Imaging Triage in the Diagnosis of Prostate Cancer (PRIMARY): A Prospective Multicentre Study. Eur Urol. 2021;80(6):682-9. doi: https://doi.org/10.1016/j.eururo.2021.08.002.
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389(10071):815-22. doi: https://doi.org/10.1016/S0140-6736(16)32401-1.
Hagens MJ, van Leeuwen PJ. A Future Prebiopsy Imaging-guided Pathway to Safely Omit Systematic Biopsies and Prevent Diagnosis of Indolent Prostate Cancer. Eur Urol. 2021;80(6):690-2. doi: https://doi.org/10.1016/j.eururo.2021.09.007.
Van de Wiele C, Sathekge M, de Spiegeleer B, de Jonghe PJ, Beels L, Maes A. PSMA-Targeting Positron Emission Agents for Imaging Solid Tumors Other Than Non-Prostate Carcinoma: A Systematic Review. Int J Mol Sci. 2019;20(19). doi: https://doi.org/10.3390/ijms20194886.
Perry E, Talwar A, Sharma S, O'Connor D, Wong LM, Taubman K, et al. Non-prostate cancer tumours: incidence on (18)F-DCFPyL PSMA PET/CT and uptake characteristics in 1445 patients. Eur J Nucl Med Mol Imaging. 2022;49(9):3277-88. doi: https://doi.org/10.1007/s00259-022-05721-z.
Bakht MK, Lovnicki JM, Tubman J, Stringer KF, Chiaramonte J, Reynolds MR, et al. Differential Expression of Glucose Transporters and Hexokinases in Prostate Cancer with a Neuroendocrine Gene Signature: A Mechanistic Perspective for (18)F-FDG Imaging of PSMA-Suppressed Tumors. J Nucl Med. 2020;61(6):904-10. doi: https://doi.org/10.2967/jnumed.119.231068.
Pang Y, Meng T, Xu W, Shang Q, Chen H. 68 Ga-FAPI PET/CT Detected Non-PSMA/FDG-Avid Primary Tumor in De Novo Metastatic Prostate Cancer. Clin Nucl Med. 2022;47(12):1108-11. doi: https://doi.org/10.1097/RLU.0000000000004349.
Gorica J, De Feo MS, Filippi L, Frantellizzi V, Schillaci O, De Vincentis G. Gastrin-releasing peptide receptor agonists and antagonists for molecular imaging of breast and prostate cancer: from pre-clinical studies to translational perspectives. Expert Rev Mol Diagn. 2022;22(11):991-6. doi: https://doi.org/10.1080/14737159.2022.2145187.
Onal C, Guler OC, Torun N, Reyhan M, Yapar AF. The effect of androgen deprivation therapy on (68)Ga-PSMA tracer uptake in non-metastatic prostate cancer patients. Eur J Nucl Med Mol Imaging. 2020;47(3):632-41. doi: https://doi.org/10.1007/s00259-019-04581-4.
Emmett L, Yin C, Crumbaker M, Hruby G, Kneebone A, Epstein R, et al. Rapid Modulation of PSMA Expression by Androgen Deprivation: Serial (68)Ga-PSMA-11 PET in Men with Hormone-Sensitive and Castrate-Resistant Prostate Cancer Commencing Androgen Blockade. J Nucl Med. 2019;60(7):950-4. doi: https://doi.org/10.2967/jnumed.118.223099.
Ettala O, Malaspina S, Tuokkola T, Luoto P, Loyttyniemi E, Bostrom PJ, et al. Prospective study on the effect of short-term androgen deprivation therapy on PSMA uptake evaluated with (68)Ga-PSMA-11 PET/MRI in men with treatment-naive prostate cancer. Eur J Nucl Med Mol Imaging. 2020;47(3):665-73. doi: https://doi.org/10.1007/s00259-019-04635-7.
Chen MY, Franklin A, Yaxley J, Gianduzzo T, McBean R, Wong D, et al. Solitary rib lesions showing prostate-specific membrane antigen (PSMA) uptake in pre-treatment staging (68) Ga-PSMA-11 positron emission tomography scans for men with prostate cancer: benign or malignant? BJU Int. 2020;126(3):396-401. doi: https://doi.org/10.1111/bju.15152.
Phelps TE, Harmon SA, Mena E, Lindenberg L, Shih JH, Citrin DE, et al. Predicting Outcomes of Indeterminate Bone Lesions on (18)F-DCFPyL PSMA PET/CT Scans in the Setting of High-Risk Primary or Recurrent Prostate Cancer. J Nucl Med. 2023;64(3):395-401. doi: https://doi.org/10.2967/jnumed.122.264334.
Rogowski P, Roach M, 3rd, Schmidt-Hegemann NS, Trapp C, von Bestenbostel R, Shi R, et al. Radiotherapy of oligometastatic prostate cancer: a systematic review. Radiat Oncol. 2021;16(1):50. doi: https://doi.org/10.1186/s13014-021-01776-8.
Galgano SJ, McDonald AM, West JT, Rais-Bahrami S. Defining Oligometastatic Disease in the New Era of PSMA-PET Imaging for Primary Staging of Prostate Cancer. Cancers. 2022;14(14):3302.
Jadvar H, Abreu AL, Ballas LK, Quinn DI. Oligometastatic Prostate Cancer: Current Status and Future Challenges. Journal of Nuclear Medicine. 2022;63(11):1628-35. doi: https://doi.org/10.2967/jnumed.121.263124.
Pathmanandavel S, Crumbaker M, Nguyen A, Yam AO, Wilson P, Niman R, et al. The Prognostic Value of Posttreatment (68)Ga-PSMA-11 PET/CT and (18)F-FDG PET/CT in Metastatic Castration-Resistant Prostate Cancer Treated with (177)Lu-PSMA-617 and NOX66 in a Phase I/II Trial (LuPIN). J Nucl Med. 2023;64(1):69-74. doi: https://doi.org/10.2967/jnumed.122.264104.
Eiber M, Herrmann K, Calais J, Hadaschik B, Giesel FL, Hartenbach M, et al. Prostate Cancer Molecular Imaging Standardized Evaluation (PROMISE): Proposed miTNM Classification for the Interpretation of PSMA-Ligand PET/CT. J Nucl Med. 2018;59(3):469-78. doi: https://doi.org/10.2967/jnumed.117.198119.
Seifert R, Emmett L, Rowe SP, Herrmann K, Hadaschik B, Calais J, et al. Second Version of the Prostate Cancer Molecular Imaging Standardized Evaluation Framework Including Response Evaluation for Clinical Trials (PROMISE V2). European Urology. 2023;83(5):405-12. doi: https://doi.org/https://doi.org/10.1016/j.eururo.2023.02.002.
Rowe SP, Pienta KJ, Pomper MG, Gorin MA. Proposal for a Structured Reporting System for Prostate-Specific Membrane Antigen-Targeted PET Imaging: PSMA-RADS Version 1.0. J Nucl Med. 2018;59(3):479-85. doi: https://doi.org/10.2967/jnumed.117.195255.
Kuten J, Dekalo S, Mintz I, Yossepowitch O, Mano R, Even-Sapir E. The significance of equivocal bone findings in staging PSMA imaging in the preoperative setting: validation of the PSMA-RADS version 1.0. EJNMMI Res. 2021;11(1):3. doi: https://doi.org/10.1186/s13550-020-00745-8.
Letang A, Crombé A, Rousseau C, Sargos P, Merlin C, Cantarel C, et al. Bone Uptake in Prostate Cancer Patients: Diagnostic Performances of PSMA-RADS v1.0, Clinical, Biological, and 68 Ga-PSMA-11 PET Features to Predict Metastasis After Biochemical Recurrence. Clin Nucl Med. 2022;47(8):e529-e39. doi: https://doi.org/10.1097/rlu.0000000000004259.
Yin Y, Werner RA, Higuchi T, Lapa C, Pienta KJ, Pomper MG, et al. Follow-up of Lesions with Equivocal Radiotracer Uptake on PSMA-Targeted PET in Patients with Prostate Cancer: Predictive Values of the PSMA-RADS-3A and PSMA-RADS-3B Categories. J Nucl Med. 2019;60(4):511-6. doi: https://doi.org/10.2967/jnumed.118.217653.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None
Research involving human and animal rights
Not applicable
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Houshmand, S., Lawhn-Heath, C. & Behr, S. PSMA PET imaging in the diagnosis and management of prostate cancer. Abdom Radiol 48, 3610–3623 (2023). https://doi.org/10.1007/s00261-023-04002-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-023-04002-z