Abstract
Purpose
To determine whether rectal filling with ultrasound gel is clinically more beneficial in preoperative T staging of patients with rectal cancer (RC) using radiomics model based on magnetic resonance imaging (MRI).
Methods
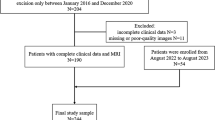
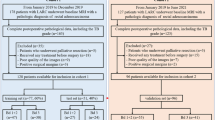
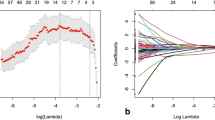
A total of 94 RC patients were assigned to cohort 1 (leave-one-out cross-validation [LOO-CV] set) and 230 RC patients were assigned to cohort 2 (test set). Patients were grouped according to different pathological T stages. The radiomics features were extracted through high-resolution T2-weighted imaging for all volume of interests in the two cohorts. Optimal features were selected using the least absolute shrinkage and selection operator (LASSO) algorithm. Model 1 (without rectal filling) and model 2 (with rectal filling) were constructed. LOO-CV was adopted for radiomics model building in cohort 1. Thereafter, the cohort 2 was used to test and verify the effectiveness of the two models.
Results
Totally, 204 patients were enrolled, including 60 cases in cohort 1 and 144 cases in cohort 2. Finally, seven optimal features with LASSO were selected to build model 1 and nine optimal features were used for model 2. The ROC curves showed an AUC of 0.806 and 0.946 for model 1 and model 2 in cohort 1, respectively, and an AUC of 0.783 and 0.920 for model 1 and model 2 in cohort 2, respectively (p = 0.021).
Conclusion
The radiomics model with rectal filling showed an advantage for differentiating T1 + 2 from T3 and had less inaccurate categories in the test cohort, suggesting that this model may be useful for T-stage evaluation.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
N/A.
Abbreviations
- RC:
-
Rectal cancer
- TME:
-
Total mesorectal excision
- ICC:
-
Intraclass correlation coefficient
- T2WI:
-
T2-weighted imaging
- VOI:
-
Volume of interest
- LASSO:
-
Least absolute shrinkage and selection operator
- XGBoost:
-
Extreme gradient boosting
- LOO-CV:
-
Leave-one-out cross-validation
- ROC:
-
Receiver operating characteristic
- AUC:
-
Area under the ROC curve
- MRF:
-
Mesorectal fascia
- EMVI:
-
Extramural vascular invasion
- LN:
-
Lymph node
References
Gollub MJ, Lall C, Lalwani N, Rosenthal MH. Current controversy, confusion, and imprecision in the use and interpretation of rectal MRI. Abdom Radiol (NY). 2019;44(11):3549-3558.
Oronsky B, Reid T, Larson C, Knox SJ. Locally advanced rectal cancer: The past, present, and future. Semin Oncol. 2020; 47(1):85-92.
Benson AB, Venook AP, Al-Hawary MM, et al. Rectal cancer, version 2.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018; 16(7):874-901.
Horvat N, Carlos Tavares Rocha C, Clemente Oliveira B, Petkovska I, Gollub MJ. MRI of rectal cancer: Tumor staging, imaging techniques, and management. Radiographics. 2019;39(2):367-387.
Nougaret S, Reinhold C, Mikhael HW, Rouanet P, Bibeau F, Brown G. The use of MR imaging in treatment planning for patients with rectal carcinoma: Have you checked the "DISTANCE"? Radiology. 2013;268(2):330-344.
Kaur H, Choi H, You YN, et al. MR imaging for preoperative evaluation of primary rectal cancer: practical considerations. RadioGraphics. 2012; 32:389–409.
Kim MJ, Lim JS, Oh YT, et al. Preoperative MRI of rectal cancer with and without rectal water filling: An intraindividual comparison. AJR 2004; 182:1469–1476.
Ye F, Zhang H, Liang X, Ouyang H, Zhao X, Zhou C. JOURNAL CLUB: Preoperative MRI evaluation of primary rectal cancer: intrasubject comparison with and without rectal distention. AJR Am J Roentgenol. 2016;207(1):32-39.
Beets-Tan RGH, Lambregts DMJ, Maas M, et al. Magnetic resonance imaging for clinical management of rectal cancer: Updated recommendations from the 2016 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol. 2018;28(4):1465-1475.
Gollub MJ, Arya S, Beets-Tan RG, et al. Use of magnetic resonance imaging in rectal cancer patients: Society of Abdominal Radiology (SAR) rectal cancer disease-focused panel (DFP) recommendations 2017. Abdom Radiol (NY). 2018;43(11):2893-2902.
Slater A, Halligan S, Taylor SA, Marshall M. Distance between the rectal wall and mesorectal fascia measured by MRI: Effect of rectal distension and implications for preoperative prediction of a tumour-free circumferential resection margin. Clin Radiol. 2006;61(1):65-70.
Stijns RC, Scheenen TW, de Wilt JH, Fütterer JJ, Beets-Tan RG. The influence of endorectal filling on rectal cancer staging with MRI. Br J Radiol. 2018;91(1089):20180205.
Gillies RJ, Kinahan PE, Hricak H. Radiomics: Images are more than pictures, they are data. Radiology. 2016; 278:563-577.
Bibault JE, Xing L, Giraud P, et al. Radiomics: A primer for the radiation oncologist. Cancer Radiother. 2020; 24:403-410.
Lambin P, Leijenaar RTH, Deist TM, et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol. 2017; 14:749-762.
Amin MB, Greene FL, Edge SB, et al. The eighth edition AJCC cancer staging manual: Continuing to build a bridge from a population-based to a more "personalized" approach to cancer staging. CA Cancer J Clin. 2017; 67(2):93-99.
Ma X, Shen F, Jia Y, Xia Y, Li Q, Lu J. MRI-based radiomics of rectal cancer: preoperative assessment of the pathological features. BMC Med Imaging. 2019;19(1):86.
Liu M, Ma X, Shen F, Xia Y, Jia Y, Lu J. MRI-based radiomics nomogram to predict synchronous liver metastasis in primary rectal cancer patients. Cancer Med. 2020;9(14):5155-5163.
van Griethuysen JJM, Fedorov A, Parmar C, et al. Computational radiomics system to decode the radiographic phenotype. Cancer Res. 2017;77(21): e104-e107.
Torlay L, Perrone-Bertolotti M, Thomas E, Baciu M. Machine learning-XGBoost analysis of language networks to classify patients with epilepsy. Brain Inform. 2017; 4(3):159-169.
Yu D, Liu Z, Su C, et al. Copy number variation in plasma as a tool for lung cancer prediction using Extreme Gradient Boosting (XGBoost) classifier. Thorac Cancer. 2020; 11(1):95-102.
Gronau QF, Wagenmakers EJ. Limitations of Bayesian leave-one-out cross-validation for model selection. Comput Brain Behav. 2019;2(1):1-11.
Zhang H, Mao Y, Chen X, et al. Magnetic resonance imaging radiomics in categorizing ovarian masses and predicting clinical outcome: a preliminary study. Eur Radiol. 2019;29(7):3358-3371.
Zhang Z, Yang J, Ho A, et al. A predictive model for distinguishing radiation necrosis from tumour progression after gamma knife radiosurgery based on radiomic features from MR images. Eur Radiol. 2018;28(6):2255-2263.
Trebeschi S, van Griethuysen JJM, Lambregts DMJ, et al. Deep learning for fully-automated localization and segmentation of rectal cancer on multiparametric MR. Sci Rep. 2017(1); 7:5301.
Pedersen BG. Moran B, Brown G, Blomqvist L, Fenger-Grøn M, Laurberg S. Reproducibility of depth of extramural tumor spread and distance to circumferential resection margin at rectal MRI: enhancement of clinical guidelines for neoadjuvant therapy. AJR 2011; 197:1360–1366.
Horvat N, Petkovska I, Gollub MJ. MR imaging of rectal cancer. Radiol Clin North Am. 2018;56(5):751-774.
Bluemke DA, Moy L, Bredella MA, et al. Assessing radiology research on artificial intelligence: a brief guide for authors, reviewers, and readers-from the radiology editorial board. Radiology. 2020;294(3):487-489.
Funding
N/A.
Author information
Authors and Affiliations
Contributions
YY performed segmentation, analyzed, and is a major contributor in writing the manuscript. HL performed segmentation, analyzed, and is a major contributor in writing the manuscript. XM acquired the data and interpreted the patient data regarding radiomics features. FC interpreted the patient data regarding radiomics features. SZ analyzed and interpreted the patient data regarding radiomics features. MW acquired the data. YX performed statistical analysis. FS conceived the project. JL conceived the project. CS conceived the project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Consent to participate
Informed consent was waived for this retrospective study.
Consent for publication
N/A.
Ethical approval
All methods of the present research were approved by the local Institutional Review Board of Changhai Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplemental
Table 1 Oblique axial High-resolution T2WI sequence parameters (DOCX 21 KB)
Supplemental
Table 2 ROC analysis of XGBoost models and subjective assessment for both cohorts (DOCX 18 KB)
Supplemental
Table 3 Comparison of proposed models in cohort 1 (DOCX 20 KB)
Supplemental Table 4
Logistic regression analyses of associations between potential predictors and pathological T stage in cohort 1 (DOCX 20 KB)
Supplemental
Fig. 1. The repeatability of radiomics features using ICCs on different scanners. (TIF 2879 KB)
Rights and permissions
About this article
Cite this article
Yuan, Y., Lu, H., Ma, X. et al. Is rectal filling optimal for MRI-based radiomics in preoperative T staging of rectal cancer?. Abdom Radiol 47, 1741–1749 (2022). https://doi.org/10.1007/s00261-022-03477-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03477-6