Abstract
Purpose
To compare the ability of a clinical-computed tomography (CT) model vs. 2D and 3D radiomics models for predicting occult peritoneal metastasis (PM) in patients with advanced gastric cancer (AGC).
Methods
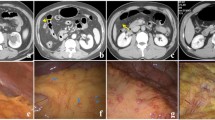
In this retrospective study, we included 49 patients with occult PM and 49 control patients (without PM) who underwent preoperative CT and subsequent surgery between January 2016 and December 2018. Clinical information and CT semantic features were collected, and CT radiomics features were extracted. A predictive clinical-CT model was created using multivariate logistic regression. The least absolute shrinkage and selection operator algorithm and logistic regression were used for constructing 2D and 3D radiomics models. These models were validated with an external cohort (n = 30). Receiver operating characteristics curve with area under the curve (AUC), sensitivity, and specificity were used to evaluate predictive performance.
Results
Tumor size, mild ascites, and serum CA125 were independent factors predictive of occult PM. The clinical-CT model of these independent factors showed better diagnostic performance than 2D and 3D radiomics models. In the external validation cohort, the AUCs of different models were as follows—clinical-CT model: 0.853 (sensitivity, 66.7%; specificity, 93.3%); 2D radiomics model: 0.622 (sensitivity, 80.0%; specificity, 46.7%); and 3D radiomics model: 0.676 (sensitivity, 60.0%; specificity, 86.0%). The clinical-CT model nomogram showed good clinical predictive efficiency to assess occult PM.
Conclusion
The clinical-CT model was better than the radiomics models in predicting occult PM in AGC.
Graphic abstract





Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394-424.doi:https://doi.org/10.3322/caac.21492
Fujitani K, Yang HK, Mizusawa J et al (2016) Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): a phase 3, randomised controlled trial. Lancet Oncol 17:309-318.doi:https://doi.org/10.1016/s1470-2045(15)00553-7
Thomassen I, van Gestel YR, van Ramshorst B et al (2014) Peritoneal carcinomatosis of gastric origin: a population-based study on incidence, survival and risk factors. Int J Cancer 134:622-628.doi:https://doi.org/10.1002/ijc.28373
Ajani JA, D'Amico TA, Almhanna K et al (2016) Gastric Cancer, Version 3.2016, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 14:1286-1312.doi:https://doi.org/10.6004/jnccn.2016.0137
Hallinan JT, Venkatesh SK (2013) Gastric carcinoma: imaging diagnosis, staging and assessment of treatment response. Cancer Imaging 13:212-227.doi:https://doi.org/10.1102/1470-7330.2013.0023
de Bree E, Koops W, Kröger R, van Ruth S, Witkamp AJ, Zoetmulder FA (2004) Peritoneal carcinomatosis from colorectal or appendiceal origin: correlation of preoperative CT with intraoperative findings and evaluation of interobserver agreement. J Surg Oncol 86:64-73.doi:https://doi.org/10.1002/jso.20049
Liu S, He J, Liu S et al (2020) Radiomics analysis using contrast-enhanced CT for preoperative prediction of occult peritoneal metastasis in advanced gastric cancer. Eur Radiol 30:239-246.doi:https://doi.org/10.1007/s00330-019-06368-5
Kim HY, Kim YH, Yun G, Chang W, Lee YJ, Kim B (2018) Could texture features from preoperative CT image be used for predicting occult peritoneal carcinomatosis in patients with advanced gastric cancer? PLoS One 13:e0194755.doi:https://doi.org/10.1371/journal.pone.0194755
Li K, Cannon JGD, Jiang SY et al (2018) Diagnostic staging laparoscopy in gastric cancer treatment: A cost-effectiveness analysis. J Surg Oncol 117:1288-1296.doi:https://doi.org/10.1002/jso.24942
Kim SJ, Kim HH, Kim YH et al (2009) Peritoneal metastasis: detection with 16- or 64-detector row CT in patients undergoing surgery for gastric cancer. Radiology 253:407-415.doi:https://doi.org/10.1148/radiol.2532082272
Huang B, Sun Z, Wang Z et al (2013) Factors associated with peritoneal metastasis in non-serosa-invasive gastric cancer: a retrospective study of a prospectively-collected database. BMC Cancer 13:57.doi:https://doi.org/10.1186/1471-2407-13-57
Hur H, Lee HH, Jung H, Song KY, Jeon HM, Park CH (2010) Predicting factors of unexpected peritoneal seeding in locally advanced gastric cancer: indications for staging laparoscopy. J Surg Oncol 102:753-757.doi:https://doi.org/10.1002/jso.21685
Huang C, Liu Z, Xiao L et al (2019) Clinical Significance of Serum CA125, CA19-9, CA72-4, and Fibrinogen-to-Lymphocyte Ratio in Gastric Cancer With Peritoneal Dissemination. Front Oncol 9:1159.doi:https://doi.org/10.3389/fonc.2019.01159
Dong D, Tang L, Li ZY et al (2019) Development and validation of an individualized nomogram to identify occult peritoneal metastasis in patients with advanced gastric cancer. Ann Oncol 30:431-438.doi:https://doi.org/10.1093/annonc/mdz001
Huang Z, Liu D, Chen X et al (2020) Deep Convolutional Neural Network Based on Computed Tomography Images for the Preoperative Diagnosis of Occult Peritoneal Metastasis in Advanced Gastric Cancer. Front Oncol 10:601869.doi:https://doi.org/10.3389/fonc.2020.601869
Jiang Y, Liang X, Wang W et al (2021) Noninvasive Prediction of Occult Peritoneal Metastasis in Gastric Cancer Using Deep Learning. JAMA Netw Open 4:e2032269.doi:https://doi.org/10.1001/jamanetworkopen.2020.32269
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: Images Are More than Pictures, They Are Data. Radiology 278:563-577.doi:https://doi.org/10.1148/radiol.2015151169
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res 77:e104-e107.doi:https://doi.org/10.1158/0008-5472.Can-17-0339
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749-762.doi:https://doi.org/10.1038/nrclinonc.2017.141
Jiang Y, Chen C, Xie J et al (2018) Radiomics signature of computed tomography imaging for prediction of survival and chemotherapeutic benefits in gastric cancer. EBioMedicine 36:171-182.doi:https://doi.org/10.1016/j.ebiom.2018.09.007
Jiang Y, Wang H, Wu J et al (2020) Noninvasive imaging evaluation of tumor immune microenvironment to predict outcomes in gastric cancer. Ann Oncol 31:760-768.doi:https://doi.org/10.1016/j.annonc.2020.03.295
Zhang L, Dong D, Zhang W et al (2020) A deep learning risk prediction model for overall survival in patients with gastric cancer: A multicenter study. Radiother Oncol 150:73-80.doi:https://doi.org/10.1016/j.radonc.2020.06.010
Wang Y, Liu W, Yu Y et al (2020) Potential value of CT radiomics in the distinction of intestinal-type gastric adenocarcinomas. Eur Radiol 30:2934-2944.doi:https://doi.org/10.1007/s00330-019-06629-3
Ng F, Kozarski R, Ganeshan B, Goh V (2013) Assessment of tumor heterogeneity by CT texture analysis: can the largest cross-sectional area be used as an alternative to whole tumor analysis? Eur J Radiol 82:342-348.doi:https://doi.org/10.1016/j.ejrad.2012.10.023
Wang F, Meng W, Wang B, Qiao L (2014) Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett 345:196-202.doi:https://doi.org/10.1016/j.canlet.2013.08.016
Bernatz S, Ackermann J, Mandel P et al (2020) Comparison of machine learning algorithms to predict clinically significant prostate cancer of the peripheral zone with multiparametric MRI using clinical assessment categories and radiomic features. Eur Radiol 30:6757-6769.doi:https://doi.org/10.1007/s00330-020-07064-5
Yang L, Yang J, Zhou X et al (2019) Development of a radiomics nomogram based on the 2D and 3D CT features to predict the survival of non-small cell lung cancer patients. Eur Radiol 29:2196-2206.doi:https://doi.org/10.1007/s00330-018-5770-y
Ortiz-Ramón R, Larroza A, Ruiz-España S, Arana E, Moratal D (2018) Classifying brain metastases by their primary site of origin using a radiomics approach based on texture analysis: a feasibility study. Eur Radiol 28:4514-4523.doi:https://doi.org/10.1007/s00330-018-5463-6
Liu Y, Zhang Y, Cheng R et al (2019) Radiomics analysis of apparent diffusion coefficient in cervical cancer: A preliminary study on histological grade evaluation. J Magn Reson Imaging 49:280-290.doi:https://doi.org/10.1002/jmri.26192
Houseni M, Mahmoud MA, Saad S, ElHussiny F, Shihab M (2021) Advanced intra-tumoural structural characterisation of hepatocellular carcinoma utilising FDG-PET/CT: a comparative study of radiomics and metabolic features in 3D and 2D. Pol J Radiol 86:e64-e73.doi:https://doi.org/10.5114/pjr.2021.103239
Lubner MG, Smith AD, Sandrasegaran K, Sahani DV, Pickhardt PJ (2017) CT Texture Analysis: Definitions, Applications, Biologic Correlates, and Challenges. Radiographics 37:1483-1503.doi:https://doi.org/10.1148/rg.2017170056
Tran B, Dancey JE, Kamel-Reid S et al (2012) Cancer genomics: technology, discovery, and translation. J Clin Oncol 30:647-660.doi:https://doi.org/10.1200/jco.2011.39.2316
Welch ML, McIntosh C, Haibe-Kains B et al (2019) Vulnerabilities of radiomic signature development: The need for safeguards. Radiother Oncol 130:2-9.doi:https://doi.org/10.1016/j.radonc.2018.10.027
Chiang MF, Tseng TK, Shih CW, Yang TH, Wu SY (2020) Clinical and contrast-enhanced image features in the prediction model for the detection of small hepatocellular carcinomas. J Cancer 11:7166-7175.doi:https://doi.org/10.7150/jca.47245
Kim M, Jeong WK, Lim S, Sohn TS, Bae JM, Sohn IS (2020) Gastric cancer: development and validation of a CT-based model to predict peritoneal metastasis. Acta Radiol 61:732-742.doi:https://doi.org/10.1177/0284185119882662
Chang DK, Kim JW, Kim BK et al (2005) Clinical significance of CT-defined minimal ascites in patients with gastric cancer. World J Gastroenterol 11:6587-6592.doi:https://doi.org/10.3748/wjg.v11.i42.6587
Yajima K, Kanda T, Ohashi M et al (2006) Clinical and diagnostic significance of preoperative computed tomography findings of ascites in patients with advanced gastric cancer. Am J Surg 192:185-190.doi:https://doi.org/10.1016/j.amjsurg.2006.05.007
Hwang GI, Yoo CH, Sohn BH et al (2004) Predictive value of preoperative serum CEA, CA19-9 and CA125 levels for peritoneal metastasis in patients with gastric carcinoma. Cancer Res Treat 36:178-181.doi:https://doi.org/10.4143/crt.2004.36.3.178
Zhou P, Qu H, Shi H, Sun G, He Q (2014) Predictive value of CA125 in peritoneal metastasis and prognosis of patients with gastric carcinoma. Chinese Journal of Gastrointestinal Surgery. https://doi.org/10.3760/cma.j.issn.1671-0274.2014.10.017:1027-1030.doi:https://doi.org/10.3760/cma.j.issn.1671-0274.2014.10.017
Fidler IJ (2003) The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat Rev Cancer 3:453-458.doi:https://doi.org/10.1038/nrc1098
Liang H (2019) [The Precised Management of Surgical Treatment for Gastric Cancer: Interpretation of the 5th edition of Japanese Gastric Cancer Treatment Guideline and the 15th edition of Japanese Classification for Gastric Cancer]. Zhonghua Zhong Liu Za Zhi 41:168–172.doi:https://doi.org/10.3760/cma.j.issn.0253-3766.2019.03.002
Funding
We have no financial relationships to disclose.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, J., Chen, Y., Zhang, Y. et al. Comparison of clinical-computed tomography model with 2D and 3D radiomics models to predict occult peritoneal metastases in advanced gastric cancer. Abdom Radiol 47, 66–75 (2022). https://doi.org/10.1007/s00261-021-03287-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03287-2