Abstract
Purpose
To evaluate whether PET/CT could be used to assess the extent of colorectal peritoneal metastases.
Methods
All patients who underwent a PET/CT scan before a CRS-HIPEC procedure between January 1, 2010 and December 31, 2013 were retrospectively included (n = 35). Two nuclear medicine physicians (observer 1 and observer 2) separately reviewed the scans on intraperitoneal abnormalities. A simplified PCI was used to compare the extent of rPCI versus sPCI.
Results
Included patients had a median age of 60.6 years. Histology of primary tumors were 51.5% adenocarcinomas, 37.1% mucinous adenocarcinoma, and 11.4% SRCC. Median sPCI was 9.5 (5.0–11.8) and median rPCI was 5.0 (3.0–7.0) for observer 1 and 4.0 (3.0–6.0) for observer 2 (p = 0.02 and p = 0.01, respectively). When compared to the surgical data, PET/CT showed a poor correlation for assessing the extent of PC for both adenocarcinoma (observer 1 rho – 0.17, p = 0.51 and observer 2 rho 0.13, p = 0.61) as well as mucinous carcinoma or SRCC (observer 1 rho 0.44, p = 0.08 and observer 2 rho 0.38, p = 0.14).
Conclusion
PET/CT underestimates the extent of PC during surgery in both mucinous and non-mucinous CRC and is not recommended for intraperitoneal tumor scoring.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Peritoneal carcinomatosis (PC) is a common manifestation of colorectal cancer (CRC) with a prevalence of up to 7% at the time of diagnosis of the primary tumor [1, 2]. PC has long been considered an incurable condition with very limited treatment options. Patients with colorectal PC have a poor prognosis with a median survival of 5–9 months without (palliative) therapy [3]. In the past years, studies have shown that treatment with cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) can increase survival and even cure the condition [4, 5]. However, CRS-HIPEC is associated with high-therapy-related morbidity (30–40%), mortality (3%), and high costs [5]. It is also a procedure which requires a long surgery time and often a long revalidation process, with an impaired quality of life scoring until 3 to 4 months post-operative [6, 7]. Therefore, patient selection is important to prevent unnecessary surgery and costs. Besides age and general condition, the presence of extraperitoneal metastases, the extent of peritoneal involvement, and the prediction of degree of successful cytoreduction are the most important selection criteria for the complete procedure [8]. In patients with colorectal PC, computed tomography (CT) of the thorax, abdomen, and pelvis is generally used to exclude extraperitoneal disease [9]. However, CT has shown poor results in assessing the extent of PC [10, 11]. Extent of PC has a major prognostic impact, high tumor load is an important reason for discontinuation of CRS-HIPEC [12, 13]. Historically, in the Radboud University Medical Centre Nijmegen a PET/CT scan was performed to rule out extraperitoneal disease in all patients with colorectal cancer scheduled for HIPEC procedure. Currently, contrast-enhanced CT is used to stage patients with colorectal PC, which is in line with national and international guidelines. The primary aim of the current retrospective study was to investigate whether PET/CT can also have a role in the non-invasive assessment of the extent of PC and to compare these results with the surgical data.
Methods
A retrospective evaluation of [18F]F-FDG-PET/CT (PET/CT) results by two independent nuclear medicine physicians was performed.
Patients
This study was performed in accordance with local medical ethical guidelines. Sixty-two patients underwent a PET/CT scan between January 1, 2010 and December 31, 2013 prior to a CRS-HIPEC procedure. After exclusion 35 patients were retrospectively included, patients were included if the PET/CT was performed to rule out extraperitoneal disease in order to determine whether the patient would be a suitable CRS-HIPEC candidate. Patients who underwent a PET/CT scan before CRS-HIPEC with a different indication were excluded from the current study (n = 19). Six patients were excluded because the PET/CT showed extraperitoneal disease. Two patients who had undergone a PET/CT were excluded because that the clinical condition progressed so that a CRS-HIPEC procedure was not indicated. None of the included patients were diabetics that used metformin. As the study did not require any patient-related interventions or experiments, assessment by the Institutional Review Board (IRB) was waived.
[18F]F-FDG PET/CT examination
Patients included before November 2011(n = 11) were scanned on the Biograph duo PET/CT scanners (Siemens Medical Solutions USA, Inc., Knoxville, TN, USA). Scanning parameters included 40 mAs (50 mAs for patient weight > 100 kg (220 lbs) and 60 mAs for > 120 kg (260 lbs)), 130 kV, 5 mm (0.20 in) slice collimation, 0.8 s rotation time and pitch of 1.5, reconstructed to 3 mm (0.12 in) slices for smooth coronal representation. [18F]F-FDG PET from proximal femora to skull base was combined with a low-dose CT for localization and attenuation correction purposes. For [18F]F-FDG-PET, the acquisition time per bed position was 4 min. Image reconstruction was performed with 2 iterations and 8 subsets. [18F]F-FDG PET/CT scans (n = 24) acquired after November 2011 were performed on the Siemens Biograph mCT scanner (4 ring, 40 slice- PET/CT scanner: with time-of-flight (Siemens, Knoxville TN)). Scanning parameters were CARE Dose4D on (ref. mAs 50), 120 kV, 3 mm (0.12 in) slice collimation, 0.5 s rotation time and pitch of 1.0, reconstructed to 5 mm (0.20 in) slices for smooth coronal visual representation. [18F]F-FDG PET from proximal femora to skull base was combined with a low-dose CT for localization and attenuation correction purposes. For [18F]F-FDG-PET, the acquisition time per bed position was 4 min. Image reconstruction was performed using TrueX and TOF (UltraHD-PET) with 3 iterations and 21 subsets and a Gausse PRF with 3.0 mm (0.12 in) FWHM. With both imaging protocols, patients fasted for 6 h before the examination and were prehydrated upon arrival with 500 mL (0.13 gal) of water. Patients had a glucose level below 8.0 mmol/L (144 mg/dL) prior to [18F]F-FDG -injection. After injection of 10 mg (0.15 g) furosemide, approximately 3.5 Mbq/kg (285 mCI/lbs) [18F]F-FDG was injected intravenously 60 min before the scan. Both imaging protocols were in accordance with the EANM guidelines [14].
Surgical peritoneal cancer index
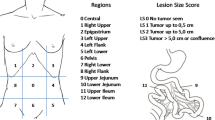
To determine the extent of PC we used the Peritoneal Cancer Index (PCI) as reported by Sugarbaker et al. [15]. The PCI divides the abdomen/pelvis into 13 regions. Each region is given a lesion size score (LSS) depending on the size of the largest lesion. The LSS can range from 0 to 3: LSS-0, no tumor seen; LSS-1, tumor up to 0.5 cm (0.2 in); LSS-2, tumor up to 5.0 cm (2.0 in); LSS-3, tumor > 5.0 cm (2.0 in) or confluent growth. The PCI score is the sum of each score given to each region and it can range from 0 to 39. Surgical PCI (sPCI) is based on visual an tactile assessment. No comparison was made with histological PCI.
PET/CT based peritoneal cancer index
The PET/CT scans were separately evaluated by an experienced nuclear medicine physician (observer 1) with 15 years of PET experience and a nuclear medicine resident (observer 2) with 4 years of PET experience. Both physicians were blinded to clinical, surgical, histopathological and other radiological data regarding the patients. To assess the extent of PC on the [18F]F-FDG scans a PCI PET/CT score (rPCI) was calculated for each patient (Figs. 1, 2). In each region the largest tumor was determined by measuring the transaxial diameter and each region was given a score from 0 to 3 (Score 0; no tumor seen; score 1, tumor up to 0.5 cm (0.2 in); score 2, tumor up to 5.0 (2.0 in) cm; score 3, tumor > 5 cm (2.0 in)). Because we expected difficulty in distinguishing whether the tumor was located in the small bowel or the peritoneal cavity, we decided to simplify the rPCI by limiting the evaluation of the PET/CT scan to the nine abdominal regions as proposed by Berthelot et al. [16]. The rPCI ranged from 0 to 27 as only the regions 0–8 were taken into account.
Comparison of radiological and surgical PCI
Because the rPCI only takes 9 regions into account and sPCI all 13 regions, the sPCI was simplified to compare both results. As proposed by Berthelot et al. [16] the simplified surgical PCI score combined the regions 9–10 with region 0 and the regions 11 and 12 with region 7. The largest tumor from regions 0, 9, and 10 and the largest tumor from regions 7, 11, and 12 were scored. The rPCI, as well as the sPCI ranged from 0 to 27.
Statistical analysis
The results consisted of nominal and continuous variables. The continuous variables were expressed as mean and standard deviation. Continuous variables without normal distribution were expressed as median and interquartile ranges. We used the Wilcoxon signed-rank test to compare continuous variables without normal distribution. The Pearson product moment correlation coefficient was used to assess the correlation between rPCI and sPCI. The interclass correlation coefficient was used to determine interobserver correlation. The correlation coefficient was calculated for two groups based on tumor histology: (1) adenocarcinoma and (2) mucinous or signet ring cell carcinoma (SRCC). Statistical analyses were performed using the Statistical Package for Social Sciences, Version 20.0 (IBM Corp., Armonk, NY, USA). The level of significance was set at p < 0.05.
Results
Table 1 shows the baseline characteristics of the included patients. Median interval between PET/CT scan and the operation was 33 (25–53) days. For the patients with adenocarcinoma, the median sPCI was 9.5 (5.0–11.8), the median rPCI was 5.0 (3.0–7.0) for observer 1, and 4.0 (3.0–6.0) for observer 2. There was a significant difference between the median sPCI and the median rPCI for both observers (p = 0.03 for observer 1 and p = 0.003 for observer 2). For the patients with mucinous carcinoma or SRCC, the median sPCI was 11.0 (5.5–15.0), the median rPCI was 3.0 (1.5–6.0) for observer 1, and 4.0 (2.5–5.5) for observer 2. There was a significant difference between the median sPCI and the rPCI for both observers (p = 0.02 for observer 1 and p = 0.01 for observer 2). A poor correlation between both PCI scores was found in the group of patients with adenocarcinoma (observer 1 rho – 0.17, p = 0.51 and observer 2 rho 0.13, p = 0.61) as well as the group of patients with mucinous carcinoma or SRCC (observer 1 rho 0.44, p = 0.08 and observer 2 rho 0.38, p = 0.14). However, a good interclass correlation coefficient was found between the rPCI of both observers (ICC 0.83 p < 0.001). Two patients underwent an explorative laparotomy but had too extensive PC for a successful cytoreduction and the procedure was aborted. The pre-operative PET/CT scan poorly predicted the tumor load for both patients. The first patient had rPCI of 2 (observer 1) and 4 (observer 2). The second patient had rPCI of 6 (observer 1) and 3 (observer 2).
To gain information about the specificity of detecting macroscopic tumor load, a subgroup analysis was performed in which patients who had at least 1 peritoneal implant of > 5 cm (2.0 in) detected during surgery were included (n = 21). The subgroup analysis also showed a poor correlation between sPCI and rPCI (observer 1 rho 0.06; p = 0.85 and observer 2 rho 0.05 p = 0.87). The rPCI of patients scanned before November 2011 did not differ from the patients scanned after November 2011.
Discussion
The present study demonstrated that PET/CT underestimates the extent of PC when compared to the intraoperative findings, which is in line with the results reported in the literature [11, 16,17,18]. An earlier study evaluating metastatic patterns of colorectal carcinoma showed that mucinous carcinoma and SRCC more frequently metastasize to the peritoneum compared to adenocarcinoma (48%, 51%, and 20%, respectively) [19]. Especially in mucinous carcinoma or SRCC an underestimation might occur due to the fact that these histological subtypes are generally less [18F]F-FDG avid, probably due to the relative hypocellularity of these tumors [20]. Therefore, it seems more difficult to assess the extent of the PC of these histological tumor types, which unfortunately are common subtypes in patients scheduled for CRS-HIPEC [21]. The PCI score was underestimated by PET/CT regardless of the histological typing. Small peritoneal metastases are easily missed by PET/CT due to its limited image resolution. Secondly, it is difficult to distinguish peritoneal metastases from increased physiological FDG-uptake in the intestines. Another reason for underestimation might be the median time between the PET/CT scan and surgery (33 days). Surgical assessment has also been shown to overestimate PCI scoring, possibly adding to the gap between scoring methods [16]. Further limitations of the current study are the retrospective character and relatively small number of cases. Three studies showed similar results compared to the present study [11, 20, 22]. Dromain et al. [11] compared surgical PCI scores with PET/CT scores and found an ICC of 0.12. No difference in the value of [18F]F-FDG PET/CT between mucinous and non-mucinous PC (ICC of 0.11 vs. 0.14) was found. On a contrary, De Vos et al. [20] included 55 patients with PC from colorectal origin. PC was detected by PET/CT in 96% of the patients with non-mucinous histology and in 60% of the patients with mucinous histology. When comparing the surgical results with the PET/CT results for the extent of PC, an adequate correlation (ρ = 0.623) for patients with non-mucinous tumors and a poor correlation for patients with mucinous tumors (ρ = − 0180) was found. The difference between patients with non-mucinous histology and mucinous histology is in contrast with results of the present study. De Vos et al. quantified the extent of PC by using the modified 7 region count (7RC), whereas the present study used the modified PCI score. The PCI is the most accepted method in scoring PC, furthermore it has been identified as a separate prognostic factor for patients with PC [23,24,25]. This might explain the difference in results between both studies. In a retrospective study of 58 patients, Pasqual et al. [26] found that PET/CT did not detect PC in 17% of the patients and underestimated the extent of PC in 43% of the cases. Four studies showed more favorable results for PET/CT in the detection of PC [16,17,18, 27].These studies included patients with various primary tumors (colorectal, gastric, ovarian, pseudomyxoma peritonei, mesothelioma), [16,17,18] while our study focused on colorectal PC. Colorectal PC has a different biology and prognosis compared to other origins of disease. The inclusion of different disease origins might cause heterogeneity in the study population when investigating limits of detection methods like PET/CT. Pfannenberg et al. [17] compared both PET and CT and fused PET/CT for the extent of PC, using a contrast-enhanced PET/CT scan where abnormalities not related to FDG-uptake were also interpreted as positive. In the present study, however, patients underwent a PET/CT scan with a low-dose CT. Using a contrast-enhanced PET/CT (especially with mucinous tumors) might improve the accuracy of rPCI. Different outcomes might also be explained by the fact that Pfannenberg et al. had a mean sPCI of 18.6 ± 11.6, whereas in the current study the mean sPCI was 11.8 ± 6.8 (p < 0.001). It might be more difficult to correctly score patients based on imaging with less extensive peritoneal disease, probably consisting of smaller lesions. CT tends to have lower sensitivity in smaller lesions and is deemed not reliable in lesions smaller than 5 cm [28, 29]. Also, the evaluation of CT is dependent of the radiologist, possibly leading to outcome bias [10]. Klumpp et al. [18] compared the diagnostic accuracy of contrast-enhanced PET/CT and MRI in relation to the surgical results. A diagnostic accuracy of 94% for PET/CT and 88% for MRI was found. Dubreuil et al. [30] assessed the prognostic impact of contrast-enhanced PET/CT in peritoneal mesothelioma. A diagnostic accuracy of 87% was found with a sensitivity and specificity of 86% and 89%, respectively. Liberale et al. [27] focused on colorectal PC. Their study found a correlation of 77% between sPCI and rPCI, but specific PCI scores were not described. Berthelot et al. [16] showed a good correlation between the PET/CT and the surgical assessment of the extent of PC (ρ 0.615; p = 0.0005). They also found a good correlation between rPCI and histological data. Compared to the histological PCI (hPCI), the sPCI was overestimated (hPCI 9.5 ± 5.6 vs. sPCI 12.3 ± 8.0, p < 0.05). Reasons for surgical overestimation might be due fibrous non-malignant tissue or adhesions mimicking visual and tactile characteristics of PC during surgery. Therefore, the rPCI might correlate better with hPCI than with sPCI. This might explain the difference in PCI scoring in the current study.
Conclusion
Defining which lesions are malignant and which are non-malignant before surgery might reduce the amount of resected tissue and potentially the impact of CRS. PET/CT has been shown to have a better diagnostic value compared to PET, CT and MRI [17, 18]. However, our findings suggest that the rPCI does not correlate with the sPCI in colorectal PC. It might deem useful to further study the correlation between histological and PET/CT scoring of peritoneal malignancies and its clinical relevance. Based on our findings, we do not recommend using [18F]F-FDG-PET/CT for pre-operative assessment of extensiveness of peritoneal malignancies.
Abbreviations
- PET/CT:
-
[18F]F-FDG PET/CT
- CRS-HIPEC:
-
Cytoreductive surgery with hyperthermic intraperitoneal chemotherapy
- PCI:
-
Peritoneal cancer index
- rPCI:
-
Peritoneal carcinoma found with PET/CT
- sPCI:
-
Surgical PCI
- SRCC:
-
Signet ring cell carcinomas
- CRC:
-
Colorectal carcinoma
References
Jayne DG, Fook S, Loi C, Seow-Choen F. Peritoneal carcinomatosis from colorectal cancer. The British journal of surgery. 2002;89(12):1545-50.
Segelman J, Granath F, Holm T, Machado M, Mahteme H, Martling A. Incidence, prevalence and risk factors for peritoneal carcinomatosis from colorectal cancer. The British journal of surgery. 2012;99(5):699-705.
de Bree E, Witkamp AJ, Zoetmulder FA. Intraperitoneal chemotherapy for colorectal cancer. Journal of surgical oncology. 2002;79(1):46-61.
Verwaal VJ, van Ruth S, de Bree E, van Sloothen GW, van Tinteren H, Boot H, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2003;21(20):3737-43.
Kuijpers AM, Mirck B, Aalbers AG, Nienhuijs SW, de Hingh IH, Wiezer MJ, et al. Cytoreduction and HIPEC in the Netherlands: nationwide long-term outcome following the Dutch protocol. Annals of surgical oncology. 2013;20(13):4224-30.
Kopanakis N, Argyriou EO, Vassiliadou D, Sidera C, Chionis M, Kyriazanos J, et al. Quality of life after cytoreductive surgery and HIPEC: A single centre prospective study. Journal of BUON: official journal of the Balkan Union of Oncology. 2018;23(2):488-93.
Shan LL, Saxena A, Shan BL, Morris DL. Quality of life after cytoreductive surgery and hyperthermic intra-peritoneal chemotherapy for peritoneal carcinomatosis: A systematic review and meta-analysis. Surgical oncology. 2014;23(4):199-210.
Harmon RL, Sugarbaker PH. Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer. International seminars in surgical oncology: ISSO. 2005;2(1):3.
Riss S, Mohamed F, Dayal S, Cecil T, Stift A, Bachleitner-Hofmann T, et al. Peritoneal metastases from colorectal cancer: patient selection for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2013;39(9):931-7.
de Bree E, Koops W, Kroger R, van Ruth S, Verwaal VJ, Zoetmulder FA. Preoperative computed tomography and selection of patients with colorectal peritoneal carcinomatosis for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2006;32(1):65-71.
Dromain C, Leboulleux S, Auperin A, Goere D, Malka D, Lumbroso J, et al. Staging of peritoneal carcinomatosis: enhanced CT vs. PET/CT. Abdominal imaging. 2008;33(1):87-93.
Elias D, Mariani A, Cloutier AS, Blot F, Goere D, Dumont F, et al. Modified selection criteria for complete cytoreductive surgery plus HIPEC based on peritoneal cancer index and small bowel involvement for peritoneal carcinomatosis of colorectal origin. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2014;40(11):1467-73.
Goere D, Souadka A, Faron M, Cloutier AS, Viana B, Honore C, et al. Extent of colorectal peritoneal carcinomatosis: attempt to define a threshold above which HIPEC does not offer survival benefit: a comparative study. Annals of surgical oncology. 2015;22(9):2958-64.
Boellaard R, O’Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. European journal of nuclear medicine and molecular imaging. 2010;37(1):181-200.
Sugarbaker PH. Successful management of microscopic residual disease in large bowel cancer. Cancer chemotherapy and pharmacology. 1999;43 Suppl:S15-25.
Berthelot C, Morel O, Girault S, Verriele V, Poirier AL, Moroch J, et al. Use of FDG-PET/CT for peritoneal carcinomatosis before hyperthermic intraperitoneal chemotherapy. Nuclear medicine communications. 2011;32(1):23-9.
Pfannenberg C, Konigsrainer I, Aschoff P, Oksuz MO, Zieker D, Beckert S, et al. (18)F-FDG-PET/CT to select patients with peritoneal carcinomatosis for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Annals of surgical oncology. 2009;16(5):1295-303.
Klumpp BD, Schwenzer N, Aschoff P, Miller S, Kramer U, Claussen CD, et al. Preoperative assessment of peritoneal carcinomatosis: intraindividual comparison of 18F-FDG PET/CT and MRI. Abdominal imaging. 2013;38(1):64-71.
Hugen N, van de Velde CJ, de Wilt JH, Nagtegaal ID. Metastatic pattern in colorectal cancer is strongly influenced by histological subtype. Annals of oncology: official journal of the European Society for Medical Oncology. 2014;25(3):651-7.
De Vos N, Goethals I, Ceelen W. Clinical Value of (18)F-FDG- PET-CT in the Preoperative Staging of Peritoneal Carcinomatosis from Colorectal Origin. Acta chirurgica Belgica. 2014;114(6):370-5.
Berger KL, Nicholson SA, Dehdashti F, Siegel BA. FDG PET evaluation of mucinous neoplasms: correlation of FDG uptake with histopathologic features. AJR American journal of roentgenology. 2000;174(4):1005-8.
Brendle C, Schwenzer NF, Rempp H, Schmidt H, Pfannenberg C, la Fougere C, et al. Assessment of metastatic colorectal cancer with hybrid imaging: comparison of reading performance using different combinations of anatomical and functional imaging techniques in PET/MRI and PET/CT in a short case series. European journal of nuclear medicine and molecular imaging. 2016;43(1):123-32.
Yan TD, Sim J, Morris DL. Selection of patients with colorectal peritoneal carcinomatosis for cytoreductive surgery and perioperative intraperitoneal chemotherapy. Annals of surgical oncology. 2007;14(6):1807-17.
Ng JL, Ong WS, Chia CS, Tan GH, Soo KC, Teo MC. Prognostic Relevance of the Peritoneal Surface Disease Severity Score Compared to the Peritoneal Cancer Index for Colorectal Peritoneal Carcinomatosis. International journal of surgical oncology. 2016;2016:2495131.
Cavaliere F, De Simone M, Virzi S, Deraco M, Rossi CR, Garofalo A, et al. Prognostic factors and oncologic outcome in 146 patients with colorectal peritoneal carcinomatosis treated with cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy: Italian multicenter study S.I.T.I.L.O. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2011;37(2):148-54.
Pasqual EM, Bertozzi S, Bacchetti S, Londero AP, Basso SM, Santeufemia DA, et al. Preoperative assessment of peritoneal carcinomatosis in patients undergoing hyperthermic intraperitoneal chemotherapy following cytoreductive surgery. Anticancer research. 2014;34(5):2363-8.
Liberale G, Lecocq C, Garcia C, Muylle K, Covas A, Deleporte A, et al. Accuracy of FDG-PET/CT in Colorectal Peritoneal Carcinomatosis: Potential Tool for Evaluation of Chemotherapeutic Response. Anticancer research. 2017;37(2):929-34.
Koh JL, Yan TD, Glenn D, Morris DL. Evaluation of preoperative computed tomography in estimating peritoneal cancer index in colorectal peritoneal carcinomatosis. Annals of surgical oncology. 2009;16(2):327-33.
Jacquet P, Jelinek JS, Steves MA, Sugarbaker PH. Evaluation of computed tomography in patients with peritoneal carcinomatosis. Cancer. 1993;72(5):1631-6.
Dubreuil J, Giammarile F, Rousset P, Rubello D, Bakrin N, Passot G, et al. The role of 18F-FDG-PET/ceCT in peritoneal mesothelioma. Nuclear medicine communications. 2017;38(4):312-8.
Funding
The authors have no funding to declare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors have nothing to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Elekonawo, F.M.K., Starremans, B., Laurens, S.T. et al. Can [18F]F-FDG PET/CT be used to assess the pre-operative extent of peritoneal carcinomatosis in patients with colorectal cancer?. Abdom Radiol 45, 301–306 (2020). https://doi.org/10.1007/s00261-019-02268-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-019-02268-w