Abstract
Background
To investigate the utility of CT volumetry for primary gastric lesions in the prediction of pathologic response to neoadjuvant chemotherapy in patients with resectable advanced gastric cancer (AGC).
Materials and methods
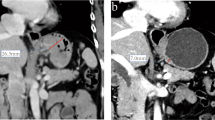
Thirty-three consecutive patients with resectable AGC stage ≥T2 and N1), who had been treated with neoadjuvant chemotherapy and radical gastric resection, were prospectively enrolled in this study. There were 30 men and 3 women with a mean age of 53.8 years. Contrast-enhanced CT was obtained after gastric distention with air before and after chemotherapy using a MDCT scanner. Pre- and post-chemotherapy thickness or short diameter and volume of the primary gastric tumor and largest lymph node (LN), were measured using a dedicated 3D software by two radiologists in consensus. PET/CT was also performed and the peak standardized uptake value (SUV) of primary gastric tumor and largest LN before and after chemotherapy was measured. The percentage diameter, volume, and SUV reduction rates for both the primary gastric tumor and the LN, were calculated and correlated with the histopathologic grades of regression using the Spearman correlation test. Differentiation between pathologic responders and nonresponders was assessed using receiver operating characteristic (ROC) analysis.
Results
Among the three CT parameters which showed significant correlation with the histopathologic grades of regression, the correlation factor was highest in the percentage volume reduction rate of primary gastric tumor (ρ = 0.484, P = 0.004) followed by percentage volume reduction of the index node (ρ = 0.397, P = 0.022), and percentage diameter reduction of the index node (ρ = 0.359, P = 0.04). However, the percentage thickness decrease rate (P = 0.208) and the percentage SUV reduction rate (P = 0.619) of primary gastric tumor were not significantly correlated with the histopathologic grades of regression. When the optimal cutoff value of the percentage volume reduction rate of primary gastric tumor was determined to be 35.6%, a sensitivity of 100% (16/16) and a specificity of 58.8% (10/17) were achieved.
Conclusion
CT volumetry for primary gastric tumor may be the most accurate tool in the prediction of pathologic response following neoadjuvant chemotherapy in patients with resectable AGC.




Similar content being viewed by others
References
Ekstrom AM, Hansson LE, Signorello LB et al. (2000) Decreasing incidence of both major histologic subtypes of gastric adenocarcinoma—a population-based study in Sweden. Br J Cancer 83:391–396
Jemal A, Siegel R, Ward E et al. (2007) Cancer statistics, 2007. CA Cancer J Clin 57:43–66
Parkin DM, Bray F, Ferlay J et al. (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Roukos DH (2000) Current status and future perspectives in gastric cancer management. Cancer Treat Rev 26:243–255
Gore RM (1997) Gastric cancer. Clinical and pathologic features. Radiol Clin North Am 35:295–310
Kim JP (1999) Surgical results in gastric cancer. Semin Surg Oncol 17:132–138
Cunningham D, Allum WH, Stenning SP et al. (2006) Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 355:11–20
Therasse P, Arbuck SG, Eisenhauer EA et al. (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Therasse P, Eisenhauer EA, Verweij J (2006) RECIST revisited: a review of validation studies on tumour assessment. Eur J Cancer 42:1031–1039
Hayes TG, Falchook GF, Varadhachary GR et al. (2006) Phase I trial of oral talactoferrin alfa in refractory solid tumors. Invest New Drugs 24:233–240
Husband JE, Schwartz LH, Spencer J et al. (2004) Evaluation of the response to treatment of solid tumours—a consensus statement of the International Cancer Imaging Society. Br J Cancer 90:2256–2260
Beer AJ, Wieder HA, Lordick F et al. (2006) Adenocarcinomas of esophagogastric junction: multi-detector row CT to evaluate early response to neoadjuvant chemotherapy. Radiology 239:472–480
Kim SH, Lee JM, Han JK et al. (2005) Effect of adjusted positioning on gastric distention and fluid distribution during CT gastrography. AJR Am J Roentgenol 185:1180–1184
Fleming ID, Cooper JS, Henson DE et al. (1997) American Joint Committee on Cancer. Manual for staging of cancer, 5th ed., Philadelphia, Lippincott, pp 71–76
Dworak O, Keilholz L, Hoffmann A (1997) Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 12:19–23
Mandard AM, Dalibard F, Mandard JC et al. (1994) Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 73:2680–2686
Kumano S, Murakami T, Kim T et al. (2005) T staging of gastric cancer: role of multi-detector row CT. Radiology 237:961–966
Kim HJ, Kim AY, Oh ST et al. (2005) Gastric cancer staging at multi-detector row CT gastrography: comparison of transverse and volumetric CT scanning. Radiology 236:879–885
Kim AY, Kim HJ, Ha HK (2005) Gastric cancer by multidetector row CT: preoperative staging. Abdom Imaging 30:465–472
Hur J, Park MS, Lee JH et al. (2006) Diagnostic accuracy of multidetector row computed tomography in T- and N staging of gastric cancer with histopathologic correlation. J Comput Assist Tomogr 30:372–377
Chen CY, Hsu JS, Wu DC et al. (2007) Gastric cancer: preoperative local staging with 3D multi-detector row CT–correlation with surgical and histopathologic results. Radiology 242:472–482
Yang DM, Kim HC, Jin W et al. (2007) 64 multidetector-row computed tomography for preoperative evaluation of gastric cancer: histological correlation. J Comput Assist Tomogr 31:98–103
Ott K, Fink U, Becker K et al. (2003) Prediction of response to preoperative chemotherapy in gastric carcinoma by metabolic imaging: results of a prospective trial. J Clin Oncol 21:4604–4610
Shreve PD, Anzai Y, Wahl RL (1999) Pitfalls in oncologic diagnosis with FDG PET imaging: physiologic and benign variants. Radiographics 19:61–77; quiz 150–151
Stahl A, Ott K, Weber WA et al. (2003) FDG PET imaging of locally advanced gastric carcinomas: correlation with endoscopic and histopathological findings. Eur J Nucl Med Mol Imaging 30:288–295
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, S.M., Kim, S.H., Lee, J.M. et al. Usefulness of CT volumetry for primary gastric lesions in predicting pathologic response to neoadjuvant chemotherapy in advanced gastric cancer. Abdom Imaging 34, 430–440 (2009). https://doi.org/10.1007/s00261-008-9420-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-008-9420-8