Abstract
Purpose
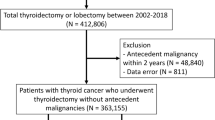
Risk of second primary malignancy (SPM) after radioiodine (RAI) therapy has been continuously debated. The aim of this study is to identify the risk of SPM in thyroid cancer (TC) patients with RAI compared with TC patients without RAI from matched cohort.
Methods
Retrospective propensity-matched cohorts were constructed across 4 hospitals in South Korea via the Observational Health Data Science and Informatics (OHDSI), and electrical health records were converted to data of common data model. TC patients who received RAI therapy constituted the target group, whereas TC patients without RAI therapy constituted the comparative group with 1:1 propensity score matching. Hazard ratio (HR) by Cox proportional hazard model was used to estimate the risk of SPM, and meta-analysis was performed to pool the HRs.
Results
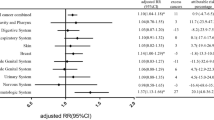
Among a total of 24,318 patients, 5,374 patients from each group were analyzed (mean age 48.9 and 49.2, women 79.4% and 79.5% for target and comparative group, respectively). All hazard ratios of SPM in TC patients with RAI therapy were ≤ 1 based on 95% confidence interval(CI) from full or subgroup analyses according to thyroid cancer stage, time-at-risk period, SPM subtype (hematologic or non-hematologic), and initial age (< 30 years or ≥ 30 years). The HR within the target group was not significantly higher (< 1) in patients who received over 3.7 GBq of I-131 compared with patients who received less than 3.7 GBq of I-131 based on 95% CI.
Conclusion
There was no significant difference of the SPM risk between TC patients treated with I-131 and propensity-matched TC patients without I-131 therapy.



Similar content being viewed by others
Data access, responsibility, and analysis
Ho-Young Lee and Sooyoung Yoo had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
References
Becker DV, Sawin CT. Radioiodine and thyroid disease: the beginning. Semin Nucl Med. 1996;26:155–64. https://doi.org/10.1016/s0001-2998(96)80020-1.
Jonklaas J, Sarlis NJ, Litofsky D, Ain KB, Bigos ST, Brierley JD, et al. Outcomes of patients with differentiated thyroid carcinoma following initial therapy. Thyroid. 2006;16:1229–42. https://doi.org/10.1089/thy.2006.16.1229.
Kazaure HS, Roman SA, Sosa JA. Aggressive variants of papillary thyroid cancer: incidence, characteristics and predictors of survival among 43,738 patients. Ann Surg Oncol. 2012;19:1874–80. https://doi.org/10.1245/s10434-011-2129-x.
Kazaure HS, Roman SA, Sosa JA. Insular thyroid cancer: a population-level analysis of patient characteristics and predictors of survival. Cancer. 2012;118:3260–7. https://doi.org/10.1002/cncr.26638.
Podnos YD, Smith DD, Wagman LD, Ellenhorn JD. Survival in patients with papillary thyroid cancer is not affected by the use of radioactive isotope. J Surg Oncol. 2007;96:3–7. https://doi.org/10.1002/jso.20656.
Ruel E, Thomas S, Dinan M, Perkins JM, Roman SA, Sosa JA. Adjuvant radioactive iodine therapy is associated with improved survival for patients with intermediate-risk papillary thyroid cancer. J Clin Endocrinol Metab. 2015;100:1529–36. https://doi.org/10.1210/jc.2014-4332.
Filetti S, Durante C, Hartl D, Leboulleux S, Locati LD, Newbold K, et al. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann Oncol. 2019;30:1856–83. https://doi.org/10.1093/annonc/mdz400.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26:1–133. https://doi.org/10.1089/thy.2015.0020.
Yu CY, Saeed O, Goldberg AS, Farooq S, Fazelzad R, Goldstein DP, et al. A systematic review and meta-analysis of subsequent malignant neoplasm risk after radioactive iodine treatment of thyroid cancer. Thyroid. 2018;28:1662–73. https://doi.org/10.1089/thy.2018.0244.
Fallahi B, Adabi K, Majidi M, Fard-Esfahani A, Heshmat R, Larijani B, et al. Incidence of second primary malignancies during a long-term surveillance of patients with differentiated thyroid carcinoma in relation to radioiodine treatment. Clin Nucl Med. 2011;36:277–82. https://doi.org/10.1097/RLU.0b013e31820a9fe3.
Khang AR, Cho SW, Choi HS, Ahn HY, Yoo WS, Kim KW, et al. The risk of second primary malignancy is increased in differentiated thyroid cancer patients with a cumulative (131)I dose over 37 GBq. Clin Endocrinol (Oxf). 2015;83:117–23. https://doi.org/10.1111/cen.12581.
Lang BH, Wong IO, Wong KP, Cowling BJ, Wan KY. Risk of second primary malignancy in differentiated thyroid carcinoma treated with radioactive iodine therapy. Surgery. 2012;151:844–50. https://doi.org/10.1016/j.surg.2011.12.019.
Souza MC, Momesso DP, Vaisman F, Vieira Neto L, Martins RA, Corbo R, et al. Is radioactive iodine- 131 treatment related to the occurrence of non-synchronous second primary malignancy in patients with differentiated thyroid cancer? Arch Endocrinol Metab. 2016;60:9–15. https://doi.org/10.1590/2359-3997000000078.
Kim C, Bi X, Pan D, Chen Y, Carling T, Ma S, et al. The risk of second cancers after diagnosis of primary thyroid cancer is elevated in thyroid microcarcinomas. Thyroid. 2013;23:575–82. https://doi.org/10.1089/thy.2011.0406.
Lin CY, Lin CL, Huang WS, Kao CH. Risk of breast cancer in patients with thyroid cancer receiving or not receiving 131I treatment: a nationwide population-based cohort study. J Nucl Med. 2016;57:685–90. https://doi.org/10.2967/jnumed.115.164830.
Molenaar RJ, Pleyer C, Radivoyevitch T, Sidana S, Godley A, Advani AS, et al. Risk of developing chronic myeloid neoplasms in well-differentiated thyroid cancer patients treated with radioactive iodine. Leukemia. 2018;32:952–9. https://doi.org/10.1038/leu.2017.323.
Molenaar RJ, Sidana S, Radivoyevitch T, Advani AS, Gerds AT, Carraway HE, et al. Risk of hematologic malignancies after radioiodine treatment of well-differentiated thyroid cancer. J Clin Oncol. 2018;36:1831–9. https://doi.org/10.1200/jco.2017.75.0232.
Seo GH, Cho YY, Chung JH, Kim SW. Increased risk of leukemia after radioactive iodine therapy in patients with thyroid cancer: a nationwide population-based study in Korea. Thyroid. 2015;25:927–34. https://doi.org/10.1089/thy.2014.0557.
Ko KY, Kao CH, Lin CL, Huang WS, Yen RF. (131)I treatment for thyroid cancer and the risk of developing salivary and lacrimal gland dysfunction and a second primary malignancy: a nationwide population-based cohort study. Eur J Nucl Med Mol Imaging. 2015;42:1172–8. https://doi.org/10.1007/s00259-015-3055-0.
Overhage JM, Ryan PB, Reich CG, Hartzema AG, Stang PE. Validation of a common data model for active safety surveillance research. J Am Med Inform Assoc. 2012;19:54–60. https://doi.org/10.1136/amiajnl-2011-000376.
Hripcsak G, Duke JD, Shah NH, Reich CG, Huser V, Schuemie MJ, et al. Observational Health Data Sciences and Informatics (OHDSI): Opportunities for Observational Researchers. Stud Health Technol Inform. 2015;216:574–8.
AJCC cancer staging manual, 7th edition.: Springer; 2010.
Hong CM, Shin JY, Kim BI, Song HC, Yoon JK, Won KS, et al. Incidence rate and factors associated with the development of secondary cancers after radioiodine therapy in differentiated thyroid cancer: a multicenter retrospective study. Eur J Nucl Med Mol Imaging. 2021. https://doi.org/10.1007/s00259-021-05608-5.
Laha D, Nilubol N, Boufraqech M. New therapies for advanced thyroid cancer. Front Endocrinol. 2020;11. https://doi.org/10.3389/fendo.2020.00082.
Wood ME, Vogel V, Ng A, Foxhall L, Goodwin P, Travis LB. Second malignant neoplasms: assessment and strategies for risk reduction. J Clin Oncol. 2012;30:3734–45. https://doi.org/10.1200/jco.2012.41.8681.
Funding
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI) and was funded by the Ministry of Health & Welfare, Republic of Korea (grand number: HI19C0378).
Author information
Authors and Affiliations
Contributions
Seok Kim was involved in the data curation; formal analysis; methodology; writing—original draft; writing—review and editing.
Ji-In Bang contributed to the formal analysis; writing—original draft; writing—review and editing.
Dachung Boo contributed to the data curation; formal analysis; methodology; writing—review and editing.
Borham Kim helped in the data curation; formal analysis; writing—review and editing.
In Young Choi contributed to the data curation; formal analysis; methodology; writing—review and editing.
SooJeong Ko was involved in the data curation; formal analysis; methodology; writing—review and editing.
Ie Ryung Yoo was involved in the data curation; writing—review and editing.
Kwangsoo Kim contributed to the methodology; formal analysis; writing—review and editing.
Junmo Kim helped in the methodology; formal analysis; writing—review and editing.
YoungHwan Joo contributed to the methodology; formal analysis; writing—review and editing.
Hyun Gee Ryoo contributed to the data curation; writing—review and editing.
Jin Chul Paeng was involved in the data curation; writing—review and editing.
Jung Mi Park helped in the data curation; writing—review and editing.
Woncheol Jang was involved in the methodology; formal analysis; writing—review and editing.
Byungwon Kim contributed to the formal analysis; writing—review and editing.
Yangha Chung contributed to the formal analysis; writing—review and editing.
Dongyoon Yang was involved in the formal analysis; writing—review and editing.
Sooyoung Yoo was involved in the conceptualization; investigation; supervision; writing—review and editing.
Ho-Young Lee helped in the conceptualization; investigation; supervision; writing—review and editing.
Corresponding authors
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was waived by the institutional review boards of Seoul National University Bundang Hospital, Seoul National University Hospital, Seoul St. Mary’s Hospital of Catholic University, and Soonchunhyang University Hospital Bucheon in the retrospective nature of the study, and all the procedures being performed were part of the routine care.
Consent to publish
Not applicable.
Competing interest
Seok Kim, Ji-In Bang, Dachung Boo, Borham Kim, In Young Choi, SooJeong Ko, Ie Ryung Yoo, Kwangsoo Kim, Junmo Kim, YoungHwan Joo, Hyun Gee Ryoo, Jin Chul Paeng, Jung Mi Park, Woncheol Jang, Byungwon Kim, Yangha Chung, Dongyoon Yank, Sooyoung Yoo, and Ho-Young Lee declare that there are no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - Head and Neck
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, S., Bang, JI., Boo, D. et al. Second primary malignancy risk in thyroid cancer and matched patients with and without radioiodine therapy analysis from the observational health data sciences and informatics. Eur J Nucl Med Mol Imaging 49, 3547–3556 (2022). https://doi.org/10.1007/s00259-022-05779-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05779-9