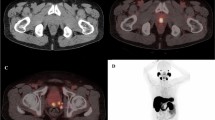
Abstract
Purpose
68Ga-PSMA PET/CT has a high detection rate in prostate cancer patients with biochemical recurrence (BCR). However, few studies have reported other imaging methods for BCR with negative 68Ga-PSMA PET/CT findings. We investigated the value of 18F-FDG compared with 68Ga-PSMA and identified BCR patients with 68Ga-PSMA-negative findings who are most likely to benefit from 18F-FDG PET/CT.
Methods
Seventy-two BCR patients with negative 68Ga-PSMA PET/CT findings were retrospectively identified from 510 patients who underwent concomitant 68Ga-PSMA and 18F-FDG PET/CT between June 2018 and August 2020. Patients were categorised into groups with positive or negative 18F-FDG PET/CT findings. Differences in patients’ characteristics between these two groups and predictors of positive 18F-FDG findings were analysed.
Results
The detection rate of 18F-FDG PET/CT was 16.7% (12/72) in BCR patients with 68Ga-PSMA-negative findings. PSA and Gleason score were significantly higher in the 18F-FDG-positive group than in the 18F-FDG-negative group (P < 0.001 and P < 0.001, respectively). A multivariate analysis indicated that PSA (PSA ≥2.3 ng/mL) and Gleason score (Gleason score ≥ 8) correlated with 18F-FDG-positive findings (P < 0.001 and P = 0.015, respectively). The probabilities of 18F-FDG-positive findings in the low-potential (PSA <2.3 ng/mL and Gleason score <8), moderate-potential (PSA <2.3 ng/mL and Gleason score ≥ 8 or PSA ≥2.3 ng/mL and Gleason score <8), and high-potential (PSA ≥2.3 ng/mL and Gleason score ≥ 8) groups were 0%, 11.5%, and 90.0%, respectively (P < 0.001).
Conclusion
PSA level and Gleason score are independent predictors of 18F-FDG-positive findings, and BCR patients with 68Ga-PSMA-negative findings with high PSA and Gleason score are most likely to benefit from 18F-FDG PET/CT.




Similar content being viewed by others
Data availability
The data in this study can obtain from the corresponding author upon request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30. https://doi.org/10.3322/caac.21590.
Boorjian SA, Eastham JA, Graefen M, Guillonneau B, Karnes RJ, Moul JW, et al. A critical analysis of the long-term impact of radical prostatectomy on cancer control and function outcomes. Eur Urol. 2012;61:664–75. https://doi.org/10.1016/j.eururo.2011.11.053.
Briganti A, Abdollah F, Nini A, Suardi N, Gallina A, Capitanio U, et al. Performance characteristics of computed tomography in detecting lymph node metastases in contemporary patients with prostate cancer treated with extended pelvic lymph node dissection. Eur Urol. 2012;61:1132–8. https://doi.org/10.1016/j.eururo.2011.11.008.
Perera M, Papa N, Roberts M, Williams M, Udovicich C, Vela I, et al. Gallium-68 prostate-specific membrane antigen positron emission tomography in advanced prostate Cancer-updated diagnostic utility, sensitivity, specificity, and distribution of prostate-specific membrane antigen-avid lesions: a systematic review and meta-analysis. Eur Urol. 2020;77:403–17. https://doi.org/10.1016/j.eururo.2019.01.049.
Sprute K, Kramer V, Koerber S, Meneses M, Fernandez R, Soza-Ried C, et al. Diagnostic accuracy of (18)F-PSMA-1007-PET/CT imaging for lymph node staging of prostate carcinoma in primary and biochemical recurrence. J Nucl Med. 2020. https://doi.org/10.2967/jnumed.120.246363.
Tan N, Oyoyo U, Bavadian N, Ferguson N, Mukkamala A, Calais J, et al. PSMA-targeted radiotracers versus (18)F Fluciclovine for the detection of prostate cancer biochemical recurrence after definitive therapy: a systematic review and meta-analysis. Radiology. 2020;296:44–55. https://doi.org/10.1148/radiol.2020191689.
Fendler WP, Ferdinandus J, Czernin J, Eiber M, Flavell RR, Behr SC, et al. Impact of (68)Ga-PSMA-11 PET on the management of recurrent prostate cancer in a prospective single-arm clinical trial. J Nucl Med. 2020. https://doi.org/10.2967/jnumed.120.242180.
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014;41:11–20. https://doi.org/10.1007/s00259-013-2525-5.
Calais J, Ceci F, Eiber M, Hope TA, Hofman MS, Rischpler C, et al. (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: a prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol. 2019;20:1286–94. https://doi.org/10.1016/S1470-2045(19)30415-2.
Crocerossa F, Marchioni M, Novara G, Carbonara U, Ferro M, Russo GI, et al. Detection rate of prostate-specific membrane antigen tracers for positron emission tomography/computed tomography in prostate cancer biochemical recurrence: a systematic review and network meta-analysis. J Urol. 2020:101097JU0000000000001369. https://doi.org/10.1097/JU.0000000000001369.
Paschalis A, Sheehan B, Riisnaes R, Rodrigues DN, Gurel B, Bertan C, et al. Prostate-specific membrane antigen heterogeneity and DNA repair defects in prostate cancer. Eur Urol. 2019;76:469–78. https://doi.org/10.1016/j.eururo.2019.06.030.
Ferraro DA, Ruschoff JH, Muehlematter UJ, Kranzbuhler B, Muller J, Messerli M, et al. Immunohistochemical PSMA expression patterns of primary prostate cancer tissue are associated with the detection rate of biochemical recurrence with (68)Ga-PSMA-11-PET. Theranostics. 2020;10:6082–94. https://doi.org/10.7150/thno.44584.
Celli M, De Giorgi U, Caroli P, Di Iorio V, Fantini L, Rossetti V, et al. Clinical value of negative (68)Ga-PSMA PET/CT in the management of biochemical recurrent prostate cancer patients. Eur J Nucl Med Mol Imaging. 2020. https://doi.org/10.1007/s00259-020-04914-8.
Jadvar H. Imaging evaluation of prostate cancer with 18F-fluorodeoxyglucose PET/CT: utility and limitations. Eur J Nucl Med Mol Imaging. 2013;40(Suppl 1):S5–10. https://doi.org/10.1007/s00259-013-2361-7.
Perez PM, Hope TA, Behr SC, van Zante A, Small EJ, Flavell RR. Intertumoral heterogeneity of 18F-FDG and 68Ga-PSMA uptake in prostate cancer pulmonary metastases. Clin Nucl Med. 2019;44:e28–32. https://doi.org/10.1097/RLU.0000000000002367.
Parida GK, Tripathy S, Datta Gupta S, Singhal A, Kumar R, Bal C, et al. Adenocarcinoma prostate with neuroendocrine differentiation: potential utility of 18F-FDG PET/CT and 68Ga-DOTANOC PET/CT over 68Ga-PSMA PET/CT. Clin Nucl Med. 2018;43:248–9. https://doi.org/10.1097/RLU.0000000000002013.
Richter JA, Rodriguez M, Rioja J, Penuelas I, Marti-Climent J, Garrastachu P, et al. Dual tracer 11C-choline and FDG-PET in the diagnosis of biochemical prostate cancer relapse after radical treatment. Mol Imaging Biol. 2010;12:210–7. https://doi.org/10.1007/s11307-009-0243-y.
Eiber M, Herrmann K, Calais J, Hadaschik B, Giesel FL, Hartenbach M, et al. Prostate cancer molecular imaging standardized evaluation (PROMISE): proposed miTNM classification for the interpretation of PSMA-ligand PET/CT. J Nucl Med. 2018;59:469–78. https://doi.org/10.2967/jnumed.117.198119.
Hofman MS, Hicks RJ, Maurer T, Eiber M. Prostate-specific membrane antigen PET: clinical utility in prostate cancer, normal patterns, pearls, and pitfalls. Radiographics. 2018;38:200–17. https://doi.org/10.1148/rg.2018170108.
Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, et al. (68)Ga-PSMA PET/CT: Joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44:1014–24. https://doi.org/10.1007/s00259-017-3670-z.
Clark MS, Packard AT, Johnson DR, Johnson GB. Pitfalls of a mixed metabolic response at PET/CT. Radiographics. 2019;39:1461–75. https://doi.org/10.1148/rg.2019180093.
Ozturk H, Karapolat I. (18)F-fluorodeoxyglucose PET/CT for detection of disease in patients with prostate-specific antigen relapse following radical treatment of a local-stage prostate cancer. Oncol Lett. 2016;11:316–22. https://doi.org/10.3892/ol.2015.3903.
Bakht MK, Lovnicki JM, Tubman J, Stringer KF, Chiaramonte J, Reynolds MR, et al. Differential expression of glucose transporters and hexokinases in prostate cancer with a neuroendocrine gene signature: a mechanistic perspective for (18)F-FDG imaging of PSMA-suppressed tumors. J Nucl Med. 2020;61:904–10. https://doi.org/10.2967/jnumed.119.231068.
Wang HT, Yao YH, Li BG, Tang Y, Chang JW, Zhang J. Neuroendocrine Prostate Cancer (NEPC) progressing from conventional prostatic adenocarcinoma: factors associated with time to development of NEPC and survival from NEPC diagnosis-a systematic review and pooled analysis. J Clin Oncol. 2014;32:3383–90. https://doi.org/10.1200/JCO.2013.54.3553.
Michaud L, Touijer KA, Mauguen A, Zelefsky MJ, Morris MJ, Lyashschenko SK, et al. (11)C-choline PET/CT in recurrent prostate cancer: retrospective analysis in a large U.S. patient series. J Nucl Med. 2020;61:827–33. https://doi.org/10.2967/jnumed.119.233098.
Funding
The study was supported by the National Natural Science Foundation of China (no. 81701724).
Author information
Authors and Affiliations
Contributions
Ruohua Chen, Jianjun Liu, and Gang Huang designed and wrote the experiments. Ruohua Chen, Yining Wang, Yiping Shi, and Lian Xu collected and analysed the data. Yinjie Zhu revised the manuscript.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
The study involving human participants was in line with the principles of the ethics committee of Renji Hospital and the Declaration of Helsinki in 1964. This study does not include animal research. Informed consent was not obtained.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - Genitourinary
Rights and permissions
About this article
Cite this article
Chen, R., Wang, Y., Shi, Y. et al. Diagnostic value of 18F-FDG PET/CT in patients with biochemical recurrent prostate cancer and negative 68Ga-PSMA PET/CT. Eur J Nucl Med Mol Imaging 48, 2970–2977 (2021). https://doi.org/10.1007/s00259-021-05221-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-021-05221-6