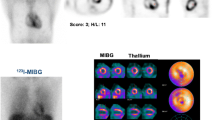
Abstract
Purpose
Cardiac sympathetic denervation may be detectable in patients with Anderson-Fabry disease (AFD), suggesting its usefulness for early detection of the disease. However, the relationship between sympathetic neuronal damage measured by 123I–metaiodobenzylguanidine (MIBG) imaging with myocardial fibrosis on cardiac magnetic resonance (CMR) is still unclear.
Methods
Cardiac sympathetic innervation was assessed by 123I–MIBG single-photon emission computed tomography (SPECT) in 25 patients with genetically proved AFD. Within one month from MIBG imaging, all patients underwent contrast-enhanced CMR. MIBG defect size and fibrosis size on CMR were measured for the left ventricle (LV) and expressed as %LV.
Results
Patients were divided into three groups according to MIBG and CMR findings: (1) matched normal, without MIBG defects and without fibrosis on CMR (n = 10); (2) unmatched, with MIBG defect but without fibrosis (n = 5); and (3) matched abnormal, with MIBG defect and fibrosis (n = 10). The three groups did not differ with respect to age, gender, α-galactosidase, proteinuria, glomerular filtration rate, and troponin I, while New York Heart Association class (p = 0.008), LV hypertrophy (p = 0.05), and enzyme replacement therapy (p = 0.02) were different among groups. Although in patients with matched abnormal findings, there was a significant correlation between MIBG defect size and area of fibrosis at CMR (r2 = 0.98, p < 0.001), MIBG defect size was larger than fibrosis size (26 ± 23 vs. 18 ± 13%LV, p = 0.02).
Conclusion
Sympathetic neuronal damage is frequent in AFD patients, and it may precede myocardial damage, such as fibrosis. Thus, 123I–MIBG imaging can be considered a challenging technique for early detection of cardiac involvement in AFD.




Similar content being viewed by others
References
Desnick RJ, Ioannou YA, Eng CM. α-Galactosidase A deficiency: Fabry disease. In: Scriver CR, Beaudet AL, Sly WS, Valle D, editors. The metabolic and molecular bases of inherited disease. 8th ed. New York: McGraw Hill; 2001. p. 3733–74.
Linhart A, Kampmann C, Zamorano JL, Sunder-Plassmann G, Beck M, Mehta A, et al. European FOS investigators. Cardiac manifestations of Anderson-Fabry disease: results from the international Fabry outcome survey. Eur Heart J. 2007;28:1228–35.
Schiffmann R. Fabry disease. Handb Clin Neurol. 2015;132:231–48.
Schiffmann R, Warnock DG, Banikazemi M, Bultas J, Linthorst GE, Packman S, et al. Fabry disease: progression of nephropathy, and prevalence of cardiac and cerebrovascular events before enzyme replacement therapy. Nephrol Dial Transplant. 2009;24:2102–11.
Moon JC, Sachdev B, Elkington AG, McKenna WJ, Mehta A, Pennell DJ, et al. Gadolinium enhanced cardiovascular magnetic resonance in Anderson-Fabry disease. Evidence for a disease specific abnormality of the myocardial interstitium. Eur Heart J. 2003;24:2151–5.
Weidemann F, Sanchez-Niño MD, Politei J, Oliveira JP, Wanner C, Warnock DG, et al. Fibrosis: a key feature of Fabry disease with potential therapeutic implications. Orphanet J Rare Dis. 2013;8:116.
Mewton N, Liu CY, Croisille P, Bluemke D, Lima JA. Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol. 2011;57:891–903.
Wu KC, Weiss RG, Thiemann DR, Kitagawa K, Schmidt A, Dalal D, et al. Late gadolinium enhancement by cardiovascular magnetic resonance heralds an adverse prognosis in non ischemic cardiomyopathy. J Am Coll Cardiol. 2008;51:2414–21.
Stirrat J, White JA. The prognostic role of late gadolinium enhancement magnetic resonance imaging in patients with cardiomyopathy. Can J Cardiol. 2013;29:329–36.
Oh-Ici D, Ridgway JP, Kuehne T, Berger F, Plein S, Sivananthan M, et al. Cardiovascular magnetic resonance of myocardial edema using a short inversion time inversion recovery (STIR) black-blood technique: diagnostic accuracy of visual and semi-quantitative assessment. J Cardiovasc Magn Reson. 2012;14:22.
Kline RC, Swanson DP, Wieland DM, Thrall JH, Gross MD, Pit B, et al. Myocardial imaging in man with I-123 metaiodobenzylguanidine. J Nucl Med. 1981;22:129–32.
Wieland DM, Brown LE, LesRogers W, Worthington KC, Wu JI, Clinthorne NH, et al. Myocardial imaging with a radioionated norepinephrine storage analog. J Nucl Med. 1981;22:22–31.
Wellman HN, Zipes DP. Cardiac sympathetic imaging with radioionated metaiodobenzylguanidine (MIBG). In: Zipes DP, Rowlands DJ, editors. Progress in cardiology 3/1. Philadelphia/London: Lea & Febiger; 1990. p. 161–74.
Barber MJ, Mueller TM, Henry DP, Felten SY, Zipes DP. Transmural myocardial infarction in the dog produces sympathectomy in non infarcted myocardium. Circulation. 1983;67:787–96.
Dae MW, Herre JM, O’Connell JW, Botvinick EH, Newman D, Munoz L. Scintigraphic assessment of sympathetic innervation after transmural versus non transmural myocardial infarction. J Am Coll Cardiol. 1991;17:1416–23.
Stanton MS, Tuli MM, Radtke NL, Heger J, Miles WM, Mock BH, et al. Regional sympathetic denervation after myocardial infarction in humans detected non-invasively using I-123-metaiodobenzylguaninidine. J Am Coll Cardiol. 1989;14:1519–26.
Glowniak JV, Turner FE, Gray LL, Palac RT, Lagunas-Solar MC, Woodward WR. Iodine-123 metaiodobenzylguanidine imaging of the heart in idiopathic congestive cardiomyopathy and cardiac transplants. J Nucl Med. 1989;30:1182–91.
Schofer J, Spielmann R, Schuchert A, Weber K, Schluter M. Iodine-123-meta-iodobenzylguanidine scintigraphy: a noninvasive method to demonstrate myocardial adrenergic nervous system disintegrity in patients with idiopathic dilated cardiomyopathy. J Am Coll Cardiol. 1988;12:1252–8.
Henderson EB, Kahn JK, Corbett JR, Jansen DE, Pippin J, Kulkarni P, et al. Abnormal I-123 metaiodobenzylguanidine myocardial washout and distribution may reflect myocardial adrenergic derangement in patients with congestive cardiomyopathy. Circulation. 1988;78:1192–9.
Nakajima K, Bunko H, Taki J, Shimizu M, Muramori A, Hisada K. Quantitative analysis of l23 I-metaiodobenzylguanidine (MIBG) uptake in hypertrophic cardiomyopathy. Am Heart J. 1990;119:1329–37.
Stark RP, McGinn AL, Wilson RF. Chest pain in cardiac-transplant recipients. N Engl J Med. 1991;324:1791–4.
Spinelli L, Pellegrino T, Pisani A, Giudice CA, Riccio E, Imbriaco M, et al. Relationship between left ventricular diastolic function and myocardial sympathetic denervation measured by (123) I-meta-iodobenzylguanidine imaging in Anderson-Fabry disease. Eur J Nucl Med Mol Imaging. 2016;43:729–39.
Yamamoto S, Suzuki H, Sugimura K, Tatebe S, Aoki T, Miura M, et al. Focal reduction in cardiac (123) I-meta-iodobenzylguanidine uptake in patients with Anderson-Fabry disease. Circ J. 2016;25(80):2550–1.
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of diet in renal disease study group. Ann Intern Med. 1999;130:461–70.
Flotats A, Carrió I, Agostini D, Le Guludec D, Marcassa C, Schäfers M, et al. Proposal for standardization of 123I metaiodobenzylguanidine (MIBG) cardiac sympathetic imaging by the EANM cardiovascular committee and the European Council of Nuclear Cardiology. Eur J Nucl Med Mol Imaging. 2010;37:1802–12.
Pellegrino T, Petretta M, De Luca S, Paolillo S, Boemio A, Carotenuto R, et al. Observer reproducibility of results from a low-dose 123I-metaiodobenzylguanidine cardiac imaging protocol in patients with heart failure. Eur J Nucl Med Mol Imaging. 2013;40:1549–57.
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK, et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the cardiac imaging Committee of the Council on clinical cardiology of the American Heart Association. Circulation. 2002;105:539–42.
Ponsiglione A, Puglia M, Morisco C, Barbuto L, Rapacciuolo A, Santoro M, et al. A unique association of arrhythmogenic right ventricular dysplasia and acute myocarditis, as assessed by cardiac MRI: a case report. BMC Cardiovasc Disord. 2016;21:16–230.
Kim RJ, Wu E, Rafael A, Chen E, Parker MA, Simonetti O, et al. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. N Engl J Med. 2000;343:1445–53.
Moon JC, Sheppard M, Reed E, Lee P, Elliot PM, Pennell DJ. The histological basis of late gadolinium enhancement cardiovascular magnetic resonance in a patient with Anderson-Fabry disease. J Cardiovasc Magn Reson. 2006;8:479–82.
Kozor R, Grieve SM, Tchan MC, Callaghan F, Hamilton-Craig C, Denaro C, et al. Cardiac involvement in genotype-positive Fabry disease patients assessed by cardiovascular MR. Heart. 2016;102:298–302.
De Cobelli F, Esposito A, Belloni E, Pieroni M, Perseghin G, Chimenti C, et al. Delayed-enhanced cardiac MRI for differentiation of Fabry’s disease from symmetric hypertrophic cardiomyopathy. Am J Roentgenol. 2009;192:W97–102.
Niemann M, Herrmann S, Hu K, Breunig F, Strotmann J, Beer M, et al. Differences in Fabry cardiomyopathy between female and male patients: consequences for diagnostic assessment. JACC Cardiovasc Imaging. 2011;4:592–601.
Pica S, Sado DM, Maestrini V, Fontana M, White SK, Treibel T, et al. Reproducibility of native myocardial T1 mapping in the assessment of Fabry disease and its role in early detection of cardiac involvement by cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2014;5:16–99.
Sado DM, White SK, Piechnik SK, Banypersad SM, Treibel T, Captur G, et al. Identification and assessment of Anderson-Fabry disease by cardiovascular magnetic resonance noncontrast myocardial T1 mapping. Circ Cardiovasc Imaging. 2013;6:392–8.
Krämer J, Niemann M, Liu D, Hu K, MacHann W, Beer M, et al. Two-dimensional speckle tracking as a non-invasive tool for identification of myocardial fibrosis in Fabry disease. Eur Heart J. 2013;34:1587–96.
Nappi C, Altiero M, Imbriaco M, Nicolai E, Giudice CA, Aiello M, et al. First experience of simultaneous PET/MRI for the early detection of cardiac involvement in patients with Anderson-Fabry disease. Eur J Nucl Med Mol Imaging. 2015;42:1025–31.
Verschure DO, Lutter R, van Eck-Smit BL, Somsen GA, Verberne HJ. Myocardial (123)I–MIBG scintigraphy in relation to markers of inflammation and long-term clinical outcome in patients with stable chronic heart failure. J Nucl Cardiol. 2016. doi:10.1007/s12350-016-0697-7
Bertelsen AK, Tøndel C, Krohn J, Bull N, Aarseth J, Houge G, et al. Small fibre neuropathy in Fabry disease. J Neurol. 2013;260:917–9.
Alamartine E, Sury A, Roche F, Pichot V, Barthelemy JC. Autonomic nervous system activity in patients with Fabry disease. Open J Intern Med. 2012;2:116–22.
Namdar M, Steffel J, Vidovic M, Brunckhorst CB, Holzmeister J, Luscher TF, et al. Electrocardiographic changes in early recognition of Fabry disease. Heart. 2011;97:485–90.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Imbriaco, M., Pellegrino, T., Piscopo, V. et al. Cardiac sympathetic neuronal damage precedes myocardial fibrosis in patients with Anderson-Fabry disease. Eur J Nucl Med Mol Imaging 44, 2266–2273 (2017). https://doi.org/10.1007/s00259-017-3778-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3778-1