Abstract
Purpose
Angiogenesis is a key event in the progression of glioblastomas (GBM). Our goal was to measure different anatomical and physiological parameters of GBM vessels using steady-state contrast-enhanced magnetic resonance imaging (SSCE-MRI), together with the assessment of biochemical parameters on GBM proliferation and angiogenesis using [11C]methyl-L-methionine (MET) and 3′-deoxy-3′-[18F]fluorothymidine (FLT) and positron emission tomography (PET). We focused on how these anatomical and biochemical read-outs correlate with one another and with immunohistochemistry.
Methods
SSCE-MRI together with 11C-MET and 18F-FLT PET were performed 3 weeks after intracranial implantation of human GBM spheroids in nude rats (n = 8). Total cerebral blood volume (tCBV), blood volume present in microvessels (μCBV), vessel density and size were calculated. Rats were treated with bevacizumab (n = 4) or vehicle (n = 4) for 3 weeks. Imaging was repeated at week 6, and thereafter immunohistochemistry was performed.
Results
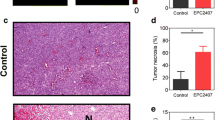
Three weeks after implantation, MRI showed an increase of vessel density and μCBV in the tumour compared to the contralateral brain. At week 6, non-treated rats showed a pronounced increase of 11C-MET and 18F-FLT tumour uptake. Between weeks 3 and 6, tCBV and vessel size increased, whereas vessel density and μCBV decreased. In rats treated with bevacizumab μCBV values were significantly smaller at week 6 than in non-treated rats, whereas the mean vessel size was higher. Accumulation of both radiotracers was lower for the treated versus the non-treated group. Most importantly, non-invasive measurement of tumour vessel characteristics and tumour proliferation correlated to immunohistochemistry findings.
Conclusion
Our study demonstrates that SSCE-MRI enables non-invasive assessment of the anatomy and physiology of the vasculature of experimental gliomas. Combined SSCE-MRI and 11C-MET/18F-FLT PET for monitoring biochemical markers of angiogenesis and proliferation in addition to vessel anatomy could be useful to improve our understanding of therapy response of gliomas.






Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin 2009;59:225–49.
Vredenburgh JJ, Desjardins A, Herndon 2nd JE, Dowell JM, Reardon DA, Quinn JA, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 2007;13:1253–9.
Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol 2009;27:4733–40.
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS, et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 1993;362:841–4.
Verhoeff JJ, van Tellingen O, Claes A, Stalpers LJ, van Linde ME, Richel DJ, et al. Concerns about anti-angiogenic treatment in patients with glioblastoma multiforme. BMC Cancer 2009;9:444–53.
Dhermain FG, Hau P, Lanfermann H, Jacobs AH, van den Bent MJ. Advanced MRI and PET imaging for assessment of treatment response in patients with gliomas. Lancet Neurol 2010;9:906–20.
Waerzeggers Y, Monfared P, Viel T, Winkeler A, Jacobs AH. Mouse models in neurological disorders: applications of non-invasive imaging. Biochim Biophys Acta 2010;1802:819–39.
Hylton N. Dynamic contrast-enhanced magnetic resonance imaging as an imaging biomarker. J Clin Oncol 2006;24:3293–8.
Tofts PS, Brix G, Buckley DL, Evelhoch JL, Henderson E, Knopp MV, et al. Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging 1999;10:223–32.
Koh TS, Zeman V, Darko J, Lee TY, Milosevic MF, Haider M, et al. The inclusion of capillary distribution in the adiabatic tissue homogeneity model of blood flow. Phys Med Biol 2001;46:1519–38.
Dennie J, Mandeville JB, Boxerman JL, Packard SD, Rosen BR, Weisskoff RM. NMR imaging of changes in vascular morphology due to tumour angiogenesis. Magn Reson Med 1998;40:793–9.
Jensen JH, Chandra R. MR imaging of microvasculature. Magn Reson Med 2000;44:224–30.
Wu EX, Tang H, Jensen JH. High-resolution MR imaging of mouse brain microvasculature using the relaxation rate shift index Q. NMR Biomed 2004;17:507–12.
Pannetier N, Lemasson B, Christen T, Tachrount M, Troprès I, Farion R, et al. Vessel size index measurements in a rat model of glioma: comparison of the dynamic (Gd) and steady-state (iron-oxide) susceptibility contrast MRI approaches. NMR Biomed 2012;25:218–26.
Ullrich RT, Jikeli JF, Diedenhofen M, Böhm-Sturm P, Unruh M, Vollmar S, et al. In-vivo visualization of tumour microvessel density and response to anti-angiogenic treatment by high resolution MRI in mice. PLoS One 2011;6:e19592.
Viel T, Talasila KM, Monfared P, Wang J, Jikeli JF, Waerzeggers Y, et al. Analysis of the growth dynamics of angiogenesis-dependent and -independent experimental glioblastomas by multi-modal small-animal PET and MRI. J Nucl Med 2012;53:1135–45.
Jacobs AH, Thomas A, Kracht LW, Li H, Dittmar C, Garlip G, et al. 18F-fluoro-L-thymidine and 11C-methylmethionine as markers of increased transport and proliferation in brain tumors. J Nucl Med 2005;46:1948–58.
Machulla HJ, Blocher A, Kuntzsch M, Piert M, Wei R, Grierson JR. Simplified labeling approach for synthesizing 3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT). J Radioanal Nucl Chem 2000;243:843–6.
Miller AJ, Joseph PM. The use of power images to perform quantitative analysis on low SNR MR images. Magn Reson Imaging 1993;11:1051–6.
Kiselev VG. Spin echo amplitude in biological tissue with implications for vessel size imaging. Joint Annual Meeting ISMRM-ESMRMB, Stockholm. 2010;01–07052010(1):1792.
Hoehn-Berlage M, Eis M, Schmitz B. Regional and directional anisotropy of apparent diffusion coefficient in rat brain. NMR Biomed 1999;12:45–50.
Vollmar SCJ, Sue M, Klein J, Jacobs AH, Herholz K. VINCI - volume imaging in neurological research, co-registration and ROIs included. In: Kremer K, Macho V, editors. Research and Scientific Computing 2003. Göttingen: Gesellschaft für wissenschaftliche Datenverarbeitung; 2004. p. 115–31.
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nature Methods 2012;9:676–82.
Sakariassen PØ, Prestegarden L, Wang J, Skaftnesmo KO, Mahesparan R, Molthoff C, et al. Angiogenesis-independent tumour growth mediated by stem-like cancer cells. Proc Natl Acad Sci U S A 2006;103:16466–71.
Keunen O, Johansson M, Oudin A, Sanzey M, Rahim SA, Fack F, et al. Anti-VEGF treatment reduces blood supply and increases tumor cell invasion in glioblastoma. Proc Natl Acad Sci U S A 2011;108:3749–54.
Beer AJ, Schwaiger M. Imaging of integrin alphavbeta3 expression. Cancer Metastasis Rev 2008;27:631–44.
Jain RK, di Tomaso E, Duda DG, Loeffler JS, Sorensen AG, Batchelor TT. Angiogenesis in brain tumours. Nat Rev Neurosci 2007;8:610–22.
Klein B, Kuschinsky W, Schröck H, Vetterlein F. Interdependency of local capillary density, blood flow, and metabolism in rat brains. Am J Physiol 1986;251:H1333–40.
Schor AM, Pendleton N, Pazouki S, Smither RL, Morris J, Lessan K, et al. Assessment of vascularity in histological sections: effects of methodology and value as an index of angiogenesis in breast tumours. Histochem J 1998;30:849–56.
von Baumgarten L, Brucker D, Tirniceru A, Kienast Y, Grau S, Burgold S, et al. Bevacizumab has differential and dose-dependent effects on glioma blood vessels and tumor cells. Clin Cancer Res 2011;17:6192–205.
Kiselev VG, Posse S. Analytical model of susceptibility-induced MR signal dephasing: effect of diffusion in a microvascular network. Magn Reson Med 1999;41:499–509.
Drevs J, Müller-Driver R, Wittig C, Fuxius S, Esser N, Hugenschmidt H, et al. PTK787/ZK 222584, a specific vascular endothelial growth factor-receptor tyrosine kinase inhibitor, affects the anatomy of the tumor vascular bed and the functional vascular properties as detected by dynamic enhanced magnetic resonance imaging. Cancer Res 2002;62:4015–22.
Miletic H, Niclou SP, Johansson M, Bjerkvig R. Anti-VEGF therapies for malignant glioma: treatment effects and escape mechanisms. Expert Opin Ther Targets 2009;13:455–68.
Okubo S, Zhen HN, Kawai N, Nishiyama Y, Haba R, Tamiya T. Correlation of L-methyl-11C-methionine (MET) uptake with L-type amino acid transporter 1 in human gliomas. J Neurooncol 2010;99:217–25.
Kracht LW, Friese M, Herholz K, Schroeder R, Bauer B, Jacobs A, et al. Methyl-[11C]-l-methionine uptake as measured by positron emission tomography correlates to microvessel density in patients with glioma. Eur J Nucl Med Mol Imaging 2003;30:868–73.
Chen W, Delaloye S, Silverman DH, Geist C, Czernin J, Sayre J, et al. Predicting treatment response of malignant gliomas to bevacizumab and irinotecan by imaging proliferation with [18F]fluorothymidine positron emission tomography: a pilot study. J Clin Oncol 2007;25:4714–21.
Schwarzenberg J, Czernin J, Cloughesy TF, Ellingson BM, Pope WB, Geist C, et al. 3'-deoxy-3'-18F-fluorothymidine PET and MRI for early survival predictions in patients with recurrent malignant glioma treated with bevacizumab. J Nucl Med 2012;53:29–36.
Acknowledgments
The authors thank Irmgard Hoppe and Christa Möllmann (EIMI, Münster) for the technical assistance with immunohistochemistry. This work is supported in part by BMBF (Bundesministerium für Bildung und Forschung) grant MoBiMed and the European Union Framework Program 7 project HEALTH-F5-2008-201842 (ENCITE).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Thomas Viel and Philipp Boehm-Sturm contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 924 kb)
Rights and permissions
About this article
Cite this article
Viel, T., Boehm-Sturm, P., Rapic, S. et al. Non-invasive imaging of glioma vessel size and densities in correlation with tumour cell proliferation by small animal PET and MRI. Eur J Nucl Med Mol Imaging 40, 1595–1606 (2013). https://doi.org/10.1007/s00259-013-2464-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2464-1