Abstract
Purpose
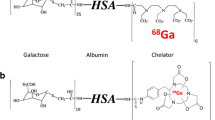
The hepatic asialoglycoprotein receptor is responsible for degradation of desialylated glycoproteins through receptor-mediated endocytosis. It has been shown that imaging of the receptor density using [99mTc]diethylenetriamine pentaacetic acid (DTPA) galactosyl human serum albumin ([99mTc]GSA) allows non-invasive determination of functional hepatocellular mass. Here we present the synthesis and evaluation of [68Ga]GSA for the potential use with positron emission tomography (PET).
Methods
Labelling of GSA with 68Ga was carried out using a fractionated elution protocol. For quality control thin-layer chromatography (TLC), high-performance liquid chromatography (HPLC) and size exclusion chromatography (SEC) techniques were evaluated. Stability of [68Ga]GSA was studied in phosphate-buffered saline (PBS) and human serum. For in vivo evaluation [68Ga]GSA distribution in Lewis rats was compared with [99mTc]GSA by using a dual isotope protocol. PET and planar imaging studies were performed using the same scaled molar dose of [68Ga]GSA and [99mTc]GSA. Time-activity curves (TAC) for heart and liver were generated and corresponding parameters calculated (t50, t90).
Results
[68Ga]GSA can be produced with high radiochemical purity. The best TLC methods for determining potential free 68Ga include 0.1 M sodium citrate as eluent. None of the TLC methods tested were able to determine potential colloids. This can be achieved by SEC. HPLC confirmed high radiochemical purity (>98 %). Stability after 120 min incubation at 37 °C was high in PBS (>95 % intact tracer) and low in human serum (∼27 % intact tracer). Biodistribution studies simultaneously injecting both tracers showed comparable liver uptake, whereas activity concentration in blood was higher for [68Ga]GSA compared to [99mTc]GSA. The [99mTc]GSA TACs exhibited a small degree of hepatic metabolism compared to the [68Ga]GSA curves. The mean [68Ga]GSA t90 was higher than the mean t90 for [99mTc]GSA. The mean [68Ga]GSA t50 was not significantly different from the mean t50 for [99mTc]GSA.
Conclusion
This study provides a promising new 68Ga-labelled compound based on a commercially used kit for imaging the functional hepatocellular mass.







Similar content being viewed by others
References
de Graaf W, Bennink RJ, Veteläinen R, van Gulik TM. Nuclear imaging techniques for the assessment of hepatic function in liver surgery and transplantation. J Nucl Med 2010;51:742–52.
Hoekstra LT, de Graaf W, Nibourg GA, Heger M, Bennink RJ, Stieger B, et al. Physiological and biochemical basis of clinical liver function tests: a review. Ann Surg 2013;257:27–36.
Kaibori M, Ha-Kawa SK, Maehara M, Ishizaki M, Matsui K, Sawada S, et al. Usefulness of Tc-99m-GSA scintigraphy for liver surgery. Ann Nucl Med 2011;25:593–602.
Virgolini I, Müller C, Klepetko W, Angelberger P, Bergmann H, O’Grady J, et al. Decreased hepatic function in patients with hepatoma or liver metastasis monitored by a hepatocyte specific galactosylated radioligand. Br J Cancer 1990;61:937–41.
Kurtaran A, Li SR, Raderer M, Leimer M, Müller C, Pidlich J, et al. Technetium-99m-galactosyl-neoglycoalbumin combined with iodine-123-Tyr-(A14)-insulin visualizes human hepatocellular carcinomas. J Nucl Med 1995;36:1875–81.
Virgolini I, Kornek G, Höbart J, Li SR, Raolerer M, Bergmann H, et al. Scintigraphic evaluation of functional hepatic mass in patients with advanced breast cancer. Br J Cancer 1993;68:549–54.
Bennink RJ, Tulchinsky M, de Graaf W, Kadry Z, van Gulik TM. Liver function testing with nuclear medicine techniques is coming of age. Semin Nucl Med 2012;42:124–37.
Virgolini I, Müller C, Angelberger P, Höbart J, Bergmann H, Sinzinger H. Functional liver imaging with 99Tcm-galactosyl-neoglycoalbumin (NGA) in alcoholic liver cirrhosis and liver fibrosis. Nucl Med Commun 1991;12:507–17.
Mansi L, Virgolini I. Diagnosis and therapy are walking together on radiopeptides’ avenue. Eur J Nucl Med Mol Imaging 2011;38:605–12.
D’Arienzo M, Chiaramida P, Chiacchiararelli L, Coniglio A, Cianni R, Salvatori R, et al. 90Y PET-based dosimetry after selective internal radiotherapy treatments. Nucl Med Commun 2012;33:633–40.
Vera DR, Krohn KA, Stadalnik RC, Scheibe PO. Tc-99m-galactosyl-neoglycoalbumin: in vivo characterization of receptor-mediated binding to hepatocytes. Radiology 1984;151:191–6.
Vera DR, Krohn KA, Stadalnik RC, Scheibe PO. Tc-99m galactosyl-neoglycoalbumin: in vitro characterization of receptor-mediated binding. J Nucl Med 1984;25:779–87.
Virgolini I, Angelberger P, Müller C, O’Grady J, Sinzinger H. 99mTc-neoglycoalbumin (NGA)-binding to human hepatic binding protein (HBP) in vitro. Br J Clin Pharmacol 1990;29:207–14.
Kokudo N, Vera DR, Makuuchi M. Clinical application of TcGSA. Nucl Med Biol 2003;30:845–9.
Marshall JS, Green AM, Pensky J, Williams S, Zinn A, Carlson DM. Measurement of circulating desialylated glycoproteins and correlation with hepatocellular damage. J Clin Invest 1974;54:555–62.
Vera DR, Stadalnik RC, Trudeau WL, Scheibe PO, Krohn KA. Measurement of receptor concentration and forward-binding rate constant via radiopharmacokinetic modeling of technetium-99m-galactosyl-neoglycoalbumin. J Nucl Med 1991;32:1169–76.
Kudo M, Todo A, Ikekubo K, Yamamoto K, Vera DR, Stadalnik RC. Quantitative assessment of hepatocellular function through in vivo radioreceptor imaging with technetium 99m galactosyl human serum albumin. Hepatology 1993;17:814–9.
Vera DR, Stadalnik RC, Metz CE, Pimstone NR. Diagnostic performance of a receptor-binding radiopharmacokinetic model. J Nucl Med 1996;37:160–4.
Decristoforo C. Gallium-68—a new opportunity for PET available from a long shelf-life generator—automation and applications. Curr Radiopharm 2012;5:212–20.
Putzer D, Gabriel M, Henninger B, Kendler D, Uprimny C, Dobrozemsky G, et al. Bone metastases in patients with neuroendocrine tumor: 68Ga-DOTA-Tyr3-octreotide PET in comparison to CT and bone scintigraphy. J Nucl Med 2009;50:1214–21.
Kudo M, Washino K, Yamamichi Y, Ikekubo K. Synthesis and radiolabeling of galactosyl human serum albumin. Methods Enzymol 1994;247:383–94.
Breeman WA, de Jong M, de Blois E, Bernard BF, Konijnenberg M, Krenning EP. Radiolabelling DOTA-peptides with 68Ga. Eur J Nucl Med Mol Imaging 2005;32:478–85.
Kudo M, Vera DR, Trudeau WL, Stadalnik RC. Hepatic uptake of [99mTc]galactosyl-neoglycoalbumin is sensitive to receptor quantity. Int J Rad Appl Instrum B 1991;18:663–6.
Miki K, Kubota K, Inoue Y, Vera DR, Makuuchi M. Receptor measurements via Tc-GSA kinetic modeling are proportional to functional hepatocellular mass. J Nucl Med 2001;42:733–7.
Stadalnik RC, Vera DR. The evolution of (99m)Tc-NGA as a clinically useful receptor-binding radiopharmaceutical. Nucl Med Biol 2001;28:499–503.
Reichert D, Lewis J, Anderson C. Metal complexes as diagnostic tools. Coord Chem Rev 1999;184:3–66.
Anderson WT, Strand M. Stability, targeting, and biodistribution of scandium-46- and gallium-67-labeled monoclonal antibody in erythroleukemic mice. Cancer Res 1985;45:2154–8.
Koop B, Reske SN, Neumaier B. Labelling of a monoclonal antibody with 68Ga using three DTPA-based bifunctional ligands and their in vitro evaluation for application in radioimmunotherapy. Radiochim Acta 2007;95:39–42.
Miki K, Kubota K, Kokudo N, Inoue Y, Bandai Y, Makuuchi M. Asialoglycoprotein receptor and hepatic blood flow using technetium-99m-DTPA-galactosyl human serum albumin. J Nucl Med 1997;38:1798–807.
Vera DR. Gallium-labeled deferoxamine-galactosyl-neoglycoalbumin: a radiopharmaceutical for regional measurement of hepatic receptor biochemistry. J Nucl Med 1992;33:1160–6.
Knetsch PA, Petrik M, Griessinger CM, Rangger C, Fani M, Kesenheimer C, et al. [68Ga]NODAGA-RGD for imaging alphavbeta3 integrin expression. Eur J Nucl Med Mol Imaging 2011;38:1303–12.
Acknowledgment
Nihon Medi-Physics Co., Ltd. (Tokyo, Japan) is acknowledged for providing the GSA kits.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haubner, R., Vera, D.R., Farshchi-Heydari, S. et al. Development of 68Ga-labelled DTPA galactosyl human serum albumin for liver function imaging. Eur J Nucl Med Mol Imaging 40, 1245–1255 (2013). https://doi.org/10.1007/s00259-013-2397-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2397-8