Abstract
Purpose
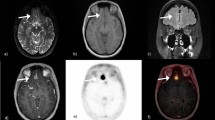
PET imaging with somatostatin receptor ligands, such as 68Ga-DOTATOC, is a well-established method for detection and target volume definition of meningiomas prior to radiotherapy. Since DOTATOC PET delivers a higher contrast between meningiomas and surrounding tissues than MRI, we conducted a retrospective analysis to compare the diagnostic accuracy of contrast-enhanced MRI (CE-MRI) with 68Ga-DOTATOC PET/CT in patients with cranial meningiomas prior to radiotherapy.
Methods
Over a period of 6 years, 134 patients (20–82 years of age, 107 women and 27 men) underwent cranial CE-MRI and 68Ga-DOTATOC PET/CT. To compare the two methods, the lesions considered typical of meningiomas visually were counted and analysed with respect to their location and SUVmax.
Results
In the 134 patients investigated by both modalities, 190 meningiomas were detected by 68Ga-DOTATOC PET/CT and 171 by CE-MRI. With knowledge of the PET/CT data, the MRI scans were reinvestigated, which led to the detection of 4 of the 19 incidental meningiomas, resulting in an overall detection rate of 92 % of the meningioma lesions that were found by PET/CT.
Conclusion
Ga-DOTATOC PET/CT demonstrated an improved sensitivity in meningioma detection when compared to CE-MRI. Tumours adjacent to the falx cerebri, located at the skull base or obscured by imaging artefacts or calcification are particularly difficult to detect by MRI. Therefore 68Ga-DOTATOC PET/CT may provide additional information in patients with uncertain or equivocal results on MRI or could help to confirm a diagnosis of meningioma based on MRI or could help to confirm MRI-based diagnosis of meningiomas in cases of biopsy limitations. It is possible that not only radiotherapy and surgical planning, but also follow-up strategies would benefit from this imaging modality.





Similar content being viewed by others
References
Black P. Meningiomas. In: Black P, Loeffler JS, editors. Cancer of the nervous system. Oxford, UK: Blackwell Science; 1997. p. 349–62.
Mirimanoff RO, Dosoretz DE, Linggood RM, Ojemann RG, Martuza RL. Meningioma: analysis of recurrence and progression following neurosurgical resection. J Neurosurg. 1985;62:18–24.
De Jesús O, Sekhar LN, Parikh HK, Wright DC, Wagner DP. Long-term follow-up of patients with meningioma involving the cavernous sinus. Recurrence, progression and quality of life. Neurosurgery. 1996;39:915–9.
Mathiesen T, Lindquist C, Kihlstrom L, Karlsson B. Recurrence of cranial base meningiomas. Neurosurgery. 1996;39:2–7.
Stafford SL, Perry A, Suman VJ, Meyer FB, Scheithauer BW, Lohse CM, et al. Primarily resected meningiomas: outcome and prognostic factors in 581 Mayo Clinic patients, 1978 through 1988. Mayo Clin Proc. 1998;73:936–42.
Henze M, Schuhmacher J, Hipp P, Kowalski J, Becker DW, Doll J, et al. PET imaging of somatostatin receptors using [68GA]DOTA-D-Phe1-Tyr3-octreotide: first results in patients with meningiomas. J Nucl Med. 2001;42:1053–6.
Henze M, Dimitrakopoulou-Strauss A, Milker-Zabel S, Schuhmacher J, Strauss LG, Doll J, et al. Characterization of 68Ga-DOTA-D-Phe1-Tyr3-octreotide kinetics in patients with meningiomas. J Nucl Med. 2005;46:763–9.
Milker-Zabel S, Zabel-du Bois A, Henze M, Huber P, Schulz-Ertner D, Hoess A, et al. Improved target volume definition for fractionated stereotactic radiotherapy in patients with intracranial meningiomas by correlation of CT, MRI, and [68Ga]-DOTATOC-PET. Int J Radiat Oncol Biol Phys. 2006;65(1):222–7.
Buchmann I, Henze M, Engelbrecht S, Eisenhut M, Runz A, Schafer M, et al. Comparison of (68)Ga-DOTATOC PET and (111)In-DTPAOC (Octreoscan) SPECT in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2007;34:1617–26.
Otte A, Jermann E, Behe M, Goetze M, Bucher HC, Roser HW, et al. DOTATOC: a powerful new tool for receptor-mediated radionuclide therapy. Eur J Nucl Med. 1997;24:792–5.
Gehler B, Paulsen F, Oksüz MO, Hauser TK, Eschmann SM, Bares R, et al. [68Ga]-DOTATOC-PET/CT for meningioma IMRT treatment planning. Radiat Oncol. 2009;4:56.
Brix G, Bellemann ME, Hauser H, Doll J. Recovery coefficients for the quantification of the arterial input functions from dynamic PET measurements: experimental and theoretical determination. Nuklearmedizin. 2002;41:184–90.
Rostomily RC, Elias M, Deng M, Elias P, Born DE, Muballe D, et al. Clinical utility of somatostatin receptor scintigraphic imaging (octreoscan) in esthesioneuroblastoma: a case study and survey of somatostatin receptor subtype expression. Head Neck. 2006;28:305–12.
Pasquali D, Notaro A, Bonavolonta’ G, Vassallo P, Bellastella A, Sinisi AA. Somatostatin receptor genes are expressed in lymphocytes from retroorbital tissues in Graves’ disease. J Clin Endocrinol Metab. 2002;87:5125–9.
Scheidhauer K, Hildebrandt G, Luyken C, Schomäcker K, Klug N, Schicha H. Somatostatin receptor scintigraphy in brain tumors and pituitary tumors: first experiences. Horm Metab Res Suppl. 1993;27:59–62.
Chen CC, Czerwiec FS, Feuillan PP. Visualization of fibrous dysplasia during somatostatin receptor scintigraphy. J Nucl Med. 1998;39:238–40.
Kang S, Mishkin FS. Visualization of Paget’s disease during somatostatin receptor scintigraphy. Clin Nucl Med. 1999;24:900–2.
Elgeti F, Amthauer H, Denecke T, Steffen I, Heuck F, Stelter L, et al. Incidental detection of breast cancer by 68Ga-DOTATOC-PET/CT in women suffering from neuroendocrine tumours. Nuklearmedizin. 2008;47:261–5.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Afshar-Oromieh, A., Giesel, F.L., Linhart, H.G. et al. Detection of cranial meningiomas: comparison of 68Ga-DOTATOC PET/CT and contrast-enhanced MRI. Eur J Nucl Med Mol Imaging 39, 1409–1415 (2012). https://doi.org/10.1007/s00259-012-2155-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-012-2155-3