Abstract
Purpose
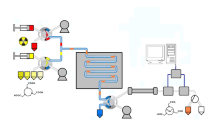
The increasing demand for radiopharmaceuticals to be provided reproducibly and flexibly with high frequency for clinical application and animal imaging would be better met by improved or even new strategies for automated tracer production. Radiosynthesis in microfluidic systems, i.e. narrow tubing with a diameter of approximately 50–500 μm, holds promise for providing the means for repetitive multidose and multitracer production. In this study, the performance of a conceptually simple microfluidic device integrated into a fully automated synthesis procedure for in-capillary radiosynthesis (ICR) of clinical grade [18F]FDG was evaluated.
Materials and methods
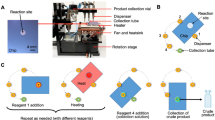
The instrumental set-up consisted of pumps for reagent and solvent delivery into small mixing chambers, μ-fluidic capillaries, in-process radioactivity monitoring, solid-phase extraction and on-column deprotection of the 18F-labelled intermediate followed by on-line formulation of [18F]FDG.
Results
In-capillary18F-fluorination of 2.1 μmol 1,3,4,6-tetra-O-acetyl-2-O-trifluoromethanesulphonyl-beta-d-mannopyranose (TATM; precursor for [18F]FDG) in acetonitrile (MeCN) at a flow rate of 0.3 ml/min within 40 s and subsequent on-line hydrolysis of the intermediate by treatment with 0.3 M NaOH for 1 min at 40°C resulted in a radiochemical yield of 88 ± 4% within <7 min. Reproducibility, robustness and suitability as a fast and efficient radiopharmaceutical research tool for 18F-fluorination was demonstrated by eight independent, sequentially performed ICRs which provided identical tracer quality (radiochemical purity >97%, MeCN <5 μg/ml) and similar absolute yields (approximately 1.4 GBq).
Conclusion
The described ICR process is a simple and efficient alternative to classic radiotracer production systems and provides a comparatively cheap instrumental methodology for the repetitive production of [18F]FDG with remarkably high efficiency and high yield under fully automated conditions. Although the results concerning the levels of activity need to be confirmed after installation of the equipment in a suitable GMP hot-cell environment, we expect the instrumental design to allow up-scaling without major difficulties or fundamental restrictions. Furthermore, we are convinced that similar or nearly identical procedures, and thus instrumentation, will allow ICR of other 18F-labelled radiopharmaceuticals.


Similar content being viewed by others
References
Townsend DW. Positron emission tomography/computed tomography. Semin Nucl Med 2008;38:152–66.
Judenhofer MS, Wehrl HF, Newport DF, Catana C, Siegel SB, Becker M, et al. Simultaneous PET-MRI: a new approach for functional and morphological imaging. Nat Med 2008;14:459–65.
Wester HJ. Nuclear imaging probes: from bench to bedside. Clin Cancer Res 2007;13:3470–81.
Schwaiger M, Ziegler S, Nekolla SG. PET/CT: challenge for nuclear cardiology. J Nucl Med 2005;46:1664–78.
Herholz K, Heiss WD. Positron emission tomography in clinical neurology. Mol Imaging Biol 2004;6:239–69.
Drzezga A. Basic pathologies of neurodegenerative dementias and their relevance for state-of-the-art molecular imaging studies. Eur J Nucl Med Mol Imaging 2008;35(Suppl 1):S4–11.
Hamacher K, Blessing G, Nebling B. Computer-aided synthesis (CAS) of no-carrier-added 2-[18F]fluoro-2-deoxy-D-glucose: an efficient automated system for the aminopolyether supported nucleophilic fluorination. Appl Radiat Isot 1990;41:49–55.
Service RF. Microchip arrays put DNA on the spot. Science 1998;282:396–99.
Ramsay G. DNA chips: state-of-the art. Nat Biotechnol 1998;16:40–4.
Spisak S, Tulassay Z, Molnar B, Guttman A. Protein microchips in biomedicine and biomarker discovery. Electrophoresis 2007;28:4261–73.
Brivio M, Verboom W, Reinhoudt DN. Miniaturized continuous flow reaction vessels: influence on chemical reactions. Lab Chip 2006;6:329–44.
Lee CC, Sui G, Elizarov A, Shu CJ, Shin YS, Dooley AN, et al. Multistep synthesis of a radiolabeled imaging probe using integrated microfluidics. Science 2005;310:1793–96.
Steel CJ, O’Brien AT, Luthra SK, Brady F. Automated PET radiosyntheses using microfluidic devices. J Labelled Compds Radiopharm 2007;50:308–11.
Gillies JM, Prenant C, Chimon GN, Smethurst GJ, Dekker BA, Zweit J. Microfluidic technology for PET radiochemistry. Appl Radiat Isot 2006;64:333–6.
Giamis A, Smith A, Yu M, Matteo J. Multiple biomarker labeling, hydrolysis, and purification from a single drying of potassium [18F]fluoride using microfluidics (abstract). J Nucl Med 2007;48(Suppl 2):137P.
Wilson AA, Garcia A, Jin L, Houle S. Radiotracer synthesis from [(11)C]iodomethane: a remarkably simple captive solvent method. Nucl Med Biol 2000;27:529–32.
McCarron JA, Turton DR, Pike VW, Poole KG. Remotely controlled production of the 5-HT1A receptor radioligand, [carbonyl-11C]WAY-100635, via 11C-carboxylation of an immobilized Grignard reagent. J Labelled Compds Radiopharm 1996;38:941–53.
Soloviev D, Tamburella C. Captive solvent [11C]acetate synthesis in GMP conditions. Appl Radiat Isot 2006;64:995–1000.
Watkins GL, Jewett DM, Mulholland GK, Kilbourn MR, Toorongian SA. A captive solvent method for rapid N-[11C]methylation of secondary amides: application to the benzodiazepine, 4′-chlorodiazepam (RO5-4864). Int J Rad Appl Instrum [A] 1988;39:441–4.
Meyer GJ, Matzke KH, Hamacher K, Füchtner F, Steinbach J, Notohamiprodjo G, et al. The stability of 2-[18F]fluoro-deoxy-D-glucose towards epimerisation under alkaline conditions. Appl Radiat Isot 1999;51:37–41.
Hamacher K, Coenen HH, Stöcklin G. Efficient stereospecific synthesis of no-carrier-added 2-[18F]-fluoro-2-deoxy-D-glucose using aminopolyether supported nucleophilic substitution. J Nucl Med 1986;27:235–8.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hans-Jürgen Wester and Gjermund Henriksen contributed equally to the study reported here.
Rights and permissions
About this article
Cite this article
Wester, HJ., Schoultz, B.W., Hultsch, C. et al. Fast and repetitive in-capillary production of [18F]FDG. Eur J Nucl Med Mol Imaging 36, 653–658 (2009). https://doi.org/10.1007/s00259-008-0985-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0985-9