Abstract
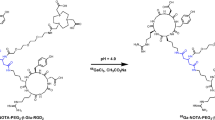
We have previously labeled cyclic RGD peptide c(RGDyK) with fluorine-18 through conjugation labeling via a prosthetic 4-[18F]fluorobenzoyl moiety and applied this [18F]FB-RGD radiotracer for αv-integrin expression imaging in different preclinical tumor models with good tumor-to-background contrast. However, the unfavorable hepatobiliary excretion and rapid tumor washout rate of this tracer limit its potential clinical applications. The aims of this study were to modify the [18F]FB-RGD tracer by inserting a heterobifunctional poly(ethylene glycol) (PEG, M.W. =3,400) between the 18F radiolabel and the RGD moiety and to test this [18F]FB-PEG-RGD tracer for brain tumor targeting and in vivo kinetics. [18F]FB-PEG-RGD was prepared by coupling the RGD-PEG conjugate with N-succinimidyl 4-[18F]fluorobenzoate ([18F]SFB) under slightly basic conditions (pH=8.5). The radiochemical yield was about 20–30% based on the active ester [18F]SFB, and specific activity was over 100 GBq/μmol. This tracer had fast blood clearance, rapid and high tumor uptake in the subcutaneous U87MG glioblastoma model (5.2±0.5%ID/g at 30 min p.i.). Moderately rapid tumor washout was observed, with the activity accumulation decreased to 2.2±0.4%ID/g at 4 h p.i. MicroPET and autoradiography imaging showed a very high tumor-to-background ratio and limited activity accumulation in the liver, kidneys and intestinal tracts. U87MG tumor implanted into the mouse forebrain was well visualized with [18F]FB-PEG-RGD. Although uptake in the orthotopic tumor was significantly lower (P<0.01) than in the subcutaneous tumor, the maximum tumor-to-brain ratio still reached 5.0±0.6 due to low normal brain background. The results of H&E staining post mortem agreed with the anatomical information obtained from non-invasive microPET imaging. In conclusion, PEGylation suitably modifies the physiological behavior of the RGD peptide. [18F]FB-PEG-RGD gave improved tumor retention and in vivo kinetics compared with [18F]FB-RGD.






Similar content being viewed by others
References
Aldape KD, Okcu MF, Bondy ML, Wrensch M. Molecular epidemiology of glioblastoma. Cancer J 2003; 9:99–106.
Castro MG, Cowen R, Williamson IK, David A, Jimenez-Dalmaroni MJ, Yuan X, Bigliari A, Williams JC, Hu J, Lowenstein PR. Current and future strategies for the treatment of malignant brain tumors. Pharmacol Ther 2003; 98:71–108.
Armstrong TS, Gilbert MR. Glial tumors—new approaches in chemotherapy. J Neurosci Nurs 2002; 34:326–330.
Wong TZ, van der Westhuizen GJ, Coleman RE. Positron emission tomography imaging of brain tumors. Neuroimaging Clin Am 2002; 12:615–626.
Shields AF, Grierson JR, Dohmen BM, Machulla HJ, Stayanoff JC, Lawhorn-Crews JM, Obradovich JE, Muzik O, Mangner TJ. Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 1998; 4:1334–1336.
Conti PS, Alauddin MM, Fissekis JR, Schmall B, Watanabe KA. Synthesis of 2’-fluoro-5-[11C]-methyl-1-β-d-arabinofuranosyluracil ([11C]-FMAU): a potential nucleoside analog for in vivo study of cellular proliferation with PET. Nucl Med Biol 1995; 22:783–789.
Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol 2002; 29:15–18.
Longo R, Sarmiento R, Fanelli M, Capaccetti B, Gattuso D, Gasparini G. Anti-angiogenic therapy: rationale, challenges and clinical studies. Angiogenesis 2003; 5:237–256.
Lopes MB. Angiogenesis in brain tumors. Microsc Res Tech 2003; 60:225–230.
Fisher MJ, Adamson PC. Anti-angiogenic agents for the treatment of brain tumors. Neuroimaging Clin Am 2002; 12:477–499.
Mischel PS, Cloughesy TF. Targeted molecular therapy of GBM. Brain Pathol 2003; 13:52–61.
Mikkelsen T. Cytostatic agents in the management of malignant gliomas. Cancer Control 1998; 5:150–162.
Taga T, Suzuki A, Gonzalez-Gomez I, Gilles FH, Stins M, Shimada H, Barsky L, Weinberg KI, Laug WE. αv-Integrin antagonist EMD 121974 induces apoptosis in brain tumor cells growing on vitronectin and tenascin. Int J Cancer 2002; 98:690–697.
MacDonald TJ, Taga T, Shimada H, Tabrizi P, Zlokovic BV, Cheresh DA, Laug WE. Preferential susceptibility of brain tumors to the antiangiogenic effects of an αv-integrin antagonist. Neurosurgery 2001; 48:151–157.
Sipkins DA, Cheresh DA, Kazemi MR, Nevin LM, Bednarski MD, Li KC. Detection of tumor angiogenesis in vivo by αvβ3-targeted magnetic resonance imaging. Nat Med 1998; 4:623–626.
Haubner R, Wester HJ, Reuning U, Senekowitsch-Schmidtke R, Diefenbach B, Kessler H, Stocklin G, Schwaiger M. Radiolabeled αvβ3 integrin antagonists: a new class of tracers for tumor targeting. J Nucl Med 1999; 40:1061–1071.
Haubner R, Wester HJ, Burkhart F, Senekowitsch-Schmidtke R, Weber W, Goodman SL, Kessler H, Schwaiger M. Glycosylated RGD-containing peptides: tracer for tumor targeting and angiogenesis imaging with improved biokinetics. J Nucl Med 2001; 42:326–336.
Haubner R, Wester HJ, Weber WA, Mang C, Ziegler SI, Goodman SL, Senekowitsch-Schmidtke R, Kessler H, Schwaiger M. Noninvasive imaging of αvβ3 integrin expression using18F-labeled RGD-containing glycopeptide and positron emission tomography. Cancer Res 2001; 61:1781–1785.
Ogawa M, Hatano K, Oishi S, Kawasumi Y, Fujii N, Kawaguchi M, Doi R, Imamura M, Yamamoto M, Ajito K, Mukai T, Saji H, Ito K. Direct electrophilic radiofluorination of a cyclic RGD peptide for in vivo αvβ3 integrin related tumor imaging. Nucl Med Biol 2003; 30:1–9.
Janssen M, Oyen WJ, Massuger LF, Frielink C, Dijkgraaf I, Edwards DS, Radjopadhye M, Corstens FH, Boerman OC. Comparison of a monomeric and dimeric radiolabeled RGD-peptide for tumor targeting. Cancer Biother Radiopharm 2002; 17:641–646.
Janssen ML, Oyen WJ, Dijkgraaf I, Massuger LF, Frielink C, Edwards DS, Rajopadhye M, Boonstra H, Corstens FH, Boerman OC. Tumor targeting with radiolabeled αvβ3 integrin binding peptides in a nude mouse model. Cancer Res 2002; 62:6146–6151.
Van Hagen PM, Breeman WA, Bernard HF, Schaar M, Mooij CM, Srinivasan A, Schmidt MA, Krenning EP, de Jong M. Evaluation of a radiolabelled cyclic DTPA-RGD analogue for tumour imaging and radionuclide therapy. Int J Cancer 2000; 90:186–198.
Su ZF, Liu G, Gupta S, Zhu Z, Rusckowski M, Hnatowich DJ. In vitro and in vivo evaluation of a technetium-99m-labeled cyclic RGD peptide as a specific marker of αvβ3 integrin for tumor imaging. Bioconjug Chem 2002; 13:561–570.
Chen X, Park R, Shahinian AH, Tohme M, Khankaldyyan V, Bozorgzadeh MH, Bading JR, Moats R, Laug WE, Conti PS.18F-labeled RGD peptide: initial evaluation for imaging brain tumor angiogenesis. Nucl Med Biol 2004; 31:179–189
Chen X, Park R, Shahinian AH, Bading JR, Conti PS. Pharmacokinetics and tumor retention of125I-labeled RGD peptide are improved by PEGylation. Nucl Med Biol 2004; 31:11–19.
Harris JM, Chess RB. Effect of pegylation on pharmaceuticals. Nat Rev Drug Discov 2003; 2:214–221.
Harris JM, Martin NE, Modi M. Pegylation: a novel process for modifying pharmacokinetics. Clin Pharmacokinet 2001; 40:539–551.
Roberts MJ, Bentley MD, Harris JM. Chemistry for peptide and protein PEGylation. Adv Drug Deliv Rev 2002; 54:459–476.
Wester HJ, Hamacher K, Stocklin G. A comparative study of N.C.A. fluorine-18 labeling of proteins via acylation and photochemical conjugation. Nucl Med Biol 1996; 23:365–372.
Kilbourn MR, Dence CS, Welch MJ, Mathias CJ. Fluorine-18 labeling of proteins. J Nucl Med 1987; 28:462–470.
Lang L, Eckelman WC. Labeling proteins at high specific activity using N-succinimidyl 4-[18F](fluoromethyl) benzoate. Appl Radiat Isot 1997; 48:169–173.
Bailon P, Berthold W. Polyethylene glycol-conjugated pharmaceutical proteins. Pharm Sci Technol Today 1998; 1:353–356.
Delgado C, Francis GE, Fisher D. The uses and properties of PEG-linked proteins. Crit Rev Ther Drug Carrier Syst 1992; 9:249–304.
Brenner B, Rector F Jr. Brenner and Rector’s: the kidney, 5th edn. Philadelphia: Saunders, 1996.
Ruoslahti E, Pierschbacher MD. New perspectives in cell adhesion: RGD and integrins. Science 1987; 238:491–497.
Dechantsreiter MA, Planker E, Mathae B, Lohof E, Hoelzemann G, Jonczyk A, Goodman SL, Kessler H. N-methylated cyclic RGD peptide as highly active and selective αvβ3 integrin antagonists. J Med Chem 1999; 42:3033–3040.
Acknowledgements
This work was carried out in part with contributions from ACS grant ACS-IRG-580007-42, the Wright Foundation, NIBIB grant R21 EB001785 and the DOD BCRP Concept Award DAMD17-03-1-0752 (to X.C.), by the T.J. Martell Foundation and R01 Grant CA82989 (to W.E.L.), and by NIH grant P20 CA86532 (to P.S.C.). The USC cyclotron team, particularly Joseph Cook and Luis Pedroza, are acknowledged for radionuclide production.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, X., Park, R., Hou, Y. et al. MicroPET imaging of brain tumor angiogenesis with 18F-labeled PEGylated RGD peptide. Eur J Nucl Med Mol Imaging 31, 1081–1089 (2004). https://doi.org/10.1007/s00259-003-1452-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-003-1452-2