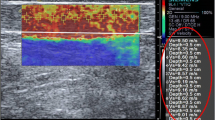
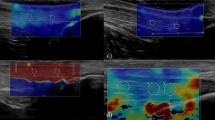
Abstract
Objective
To determine the reproducibility of compression elastography (CE) when measuring strain data, a measure of stiffness of the human Achilles tendon in vivo, over consecutive measures, consecutive days and when using different foot positions.
Materials and methods
Eight participants (4 males, 4 females; mean age 25.5 ± 2.51 years, range 21–30 years; height 173.6 ± 11.7 cm, range 156–189 cm) had five consecutive CE measurements taken on one day and a further five CE measures taken, one per day, at the same time of day, every day for a consecutive 5-day period. These 80 measurements were used to assess both the repeatability and reproducibility of the technique. Means, standard deviations, coefficient of variation (CV), Pearson correlation analysis (R) and intra-class correlation coefficients (ICC) were calculated.
Results
For CE data, all CVs were above 53%, R values indicated no-to-weak correlations between measures at best (range 0.01–0.25), and ICC values were all classified in the poor category (range 0.00–0.11). CVs for length and diameter measures were acceptably low indicating a high level of reliability.
Conclusions
Given the wide variation obtained in the CE results, it was concluded that CE using this specific system has a low level of reproducibility for measuring the stiffness of the human Achilles tendon in vivo over consecutive days, consecutive measures and in different foot positions.




Similar content being viewed by others
References
Milgrom C, Finestone A, Zin D, Mandel D, Novack V. Cold weather training: a risk factor for Achilles paratendinitis among recruits. Foot Ankle Int. 2003;24:398–401.
Wren TAL, Yerby SA, Beaupré GS, Carter DR. Mechanical properties of the human Achilles tendon. Clin Biomech. 2001;16:245–51.
Murtaugh B, Ihm JM. Eccentric training for the treatment of tendinopathies. Curr Sports Med Rep. 2013;12:175–82.
Ophir J, Cespedes I, Ponnekanti H, Yazdi Y, Li X. Elastography: a quantitative method for imaging the elasticity of biological tissues. Ultrason Imaging. 1991;13:111–34.
Horton L. Correlation of B-mode sonography and compression elastography for diagnosis of tendon degeneration: a pictorial review. Ultrasound. 2013;21:176–80.
Sconfienza LM, Silvestri E, Orlandi D, et al. Real-time sonoelastography of the plantar fascia: comparison between patients with plantar fasciitis and healthy control subjects. Radiology. 2013;267:195–200.
Sarvazyan A, Hall TJ, Urban MW, Fatemi M, Aglyamov SR, Garra BS. An overview of elastography—an emerging branch of medical imaging. Curr Med Imaging Rev. 2011;7:255–82.
Itoigawa Y, Sperling JW, Steinmann SP, et al. Feasibility assessment of shear wave elastography to rotator cuff muscle. Clin Anat. 2015;28:213–8.
Ahn K-S, Kang CH, Hong S-J, Jeong W-K. Ultrasound elastography of lateral epicondylosis: clinical feasibility of quantitative elastographic measurements. AJR Am J Roentgenol. 2014;202:1094–9.
Porta F, Damjanov N, Galluccio F, Iagnocco A, Matucci-Cerinic M. Ultrasound elastography is a reproducible and feasible tool for the evaluation of the patellar tendon in healthy subjects. Int J Rheum Dis. 2014;17:760–4.
Ooi CC, Malliaras P, Schneider ME, Connell DA. “Soft, hard, or just right?” Applications and limitations of axial-strain sonoelastography and shear-wave elastography in the assessment of tendon injuries. Skelet Radiol. 2013;43:1–12.
Yamamoto Y, Yamaguchi S, Sasho T, et al. Quantitative ultrasound elastography with an acoustic coupler for Achilles tendon elasticity: measurement repeatability and normative values. J Ultrasound Med. 2015;35:159–66.
De Zordo T, Fink C, Feuchtner GM, Smekal V, Reindl M, Klauser AS. Real-time sonoelastography findings in healthy Achilles tendons. AJR Am J Roentgenol. 2009;193:W134–8.
Drakonaki EE, Allen GM, Wilson DJ. Real-time ultrasound elastography of the normal Achilles tendon: reproducibility and pattern description. Clin Radiol. 2009;64:1196–202.
Klauser AS, Miyamoto H, Tamegger M, et al. Achilles tendon assessed with sonoelastography: histologic agreement. Radiology. 2013;267:837–42.
Palle L, Reddy MB, Reddy KJ, Kumari MV. Technical note: real-time sonoelastography evaluation of Achilles tendon. Indian J Radiol Imaging. 2011;21:267–9.
Tan S, Kudaş S, Özcan AS, et al. Real-time sonoelastography of the Achilles tendon: pattern description in healthy subjects and patients with surgically repaired complete ruptures. Skelet Radiol. 2012;41:1067–72.
Schneebeli A, Del Grande F, Vincenzo G, et al. Real-time sonoelastography using an external reference material: test-retest reliability of healthy Achilles tendons. Skelet Radiol. 2016;45:1045–52.
Boesen MI, Koenig MJ, Torp-Pedersen S, Bliddal H, Langberg H. Tendinopathy and Doppler activity: the vascular response of the achilles tendon to exercise. Scand J Med Sci Sport. 2006;16:463–9.
Rosengarten SD, Cook JL, Bryant AL, Cordy JT, Daffy J, Docking SI. Australian football players’ Achilles tendons respond to game loads within 2 days: an ultrasound tissue characterisation (UTC) study. Br J Sports. 2015;49:183–7.
Wu CH, Chang KV, Mio S, Chen WS, Wang TG. Sonoelastography of the plantar fascia. Radiology. 2011;259:502–7.
Chino K, Akagi R, Dohi M, Fukashiro S, Takahashi H. Reliability and validity of quantifying absolute muscle hardness using ultrasound elastography. PLoS One. 2012;7, e45764.
Arda K, Ciledag N, Aktas E, Aribas BK, Köse K. Quantitative assessment of normal soft-tissue elasticity using shear-wave ultrasound elastography. AJR Am J Roentgenol. 2011;197:532–6.
Treece G, Lindop J, Chen L, Housden J, Prager R, Gee A. Real-time quasi-static ultrasound elastography. Interface Focus. 2011;1:540–52.
Iversen JV, Bartels EM, Langberg H. The Victorian Institute of Sports Assessment—Achilles questionnaire (VISA-A)—a reliable tool for measuring Achilles tendinopathy. Int J Sports Phys Ther. 2012;7:76–84.
Brandenburg JE, Eby SF, Song P, et al. Ultrasound elastography: the new frontier in direct measurement of muscle stiffness. Arch Phys Med Rehabil. 2014;95:2207–19.
Calvete AC, Rodríguez JM, de Dios Berná-Mestre J, Rios A, Abellán-Rivero D, Reus M. Interobserver agreement for thyroid elastography value of the quality factor. J Ultrasound Med. 2013;32:495–504.
Dancey C, Reidy J. Statistics without maths for psychology using SPSS for windows. 3rd ed. New Jersey: Pearson Prentice Hall; 2004.
Giraudeau B. Negative values of the intraclass correlation coefficient are not theoretically possible. J Clin Epidemiol. 1996;49:1205–6.
Ying M, Yeung E, Li B, Li W, Lui M, Tsoi C. Sonographic evaluation of the size of Achilles tendon: the effect of exercise and dominance of the ankle. Ultrasound Med Biol. 2003;29:637–42.
Chen XM, Cui LG, He P, Shen WW, Qian YJ, Wang JR. Shear wave elastographic characterization of normal and torn achilles tendons a pilot study. J Ultrasound Med. 2013;32:449–55.
Peltz CD, Haladik JA, Divine G, Siegal D, van Holsbeeck M, Bey MJ. ShearWave elastography: repeatability for measurement of tendon stiffness. Skelet Radiol. 2013;42:1151–6.
Eby SF, Song P, Chen S, Chen Q, Greenleaf JF, An K-N. Validation of shear wave elastography in skeletal muscle. J Biomech. 2013;46:2381–7.
Klauser AS, Miyamoto H, Bellmann-Weiler R, Feuchtner GM, Wick MC, Jaschke WR. Sonoelastography: musculoskeletal applications. Radiology. 2014;272:622–33.
Sconfienza LM, Silvestri E, Cimmino MA. Sonoelastography in the evaluation of painful Achilles tendon in amateur athletes. Clin Exp Rheumatol. 2010;28:373–8.
Obuchowski N, Lieber M. Statistics and methodology. Skelet Radiol. 2008;37:393–6.
Hoskins PR. Principles of ultrasound elastography. Ultrasound. 2012;20:8–15.
Drakonaki EE, Allen GM, Wilson DJ. Ultrasound elastography for musculoskeletal applications. Br J Radiol. 2012;85:1435–45.
van Schie HTM, Bakker EM, Jonker AM, van Weeren PR. Computerized ultrasonographic tissue characterization of equine superficial digital flexor tendons by means of stability quantification of echo patterns in contiguous transverse ultrasonographic images. Am J Vet Res. 2003;64:366–75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Payne, C., Webborn, N., Watt, P. et al. Poor reproducibility of compression elastography in the Achilles tendon: same day and consecutive day measurements. Skeletal Radiol 46, 889–895 (2017). https://doi.org/10.1007/s00256-017-2629-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-017-2629-2