Abstract
Objective
To establish baseline T2* values in healthy knee joint cartilage at 3 T.
Materials and Methods
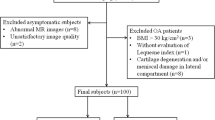
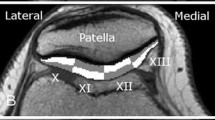
Thirty-four volunteers (mean age: 24.6 ± 2.7 years) with no history or clinical findings indicative of any knee joint disease were enrolled. The protocol included a double-echo steady-state (DESS) sequence for morphological cartilage evaluation and a gradient-echo multi-echo sequence for T2* assessment. Bulk and zonal T2* values were assessed in eight regions: posterior lateral femoral condyle; central lateral femoral condyle; trochlea; patella; lateral tibial plateau; posterior medial femoral condyle; central medial femoral condyle; and medial tibial plateau. Statistical evaluation comprised a two-tailed t test and a one-way analysis of variance to identify zonal and regional differences.
Results
T2* mapping revealed higher T2* values in the superficial zone in all regions (P values ≤ 0.001) except for the posterior medial femur condyle (P = 0.087), and substantial regional differences demonstrating superior values in trochlear cartilage, intermediate values in patellar and central femoral condylar cartilage, and low T2* values in posterior femoral condylar cartilage and tibial plateau cartilage.
Conclusion
Substantial regional differences in T2* measures should be taken into consideration when conducting T2* mapping of knee joint cartilage.







Similar content being viewed by others
References
Altman RD. The syndrome of osteoarthritis. J Rheumatol. 1997;24(4):766–7.
Burstein D, Gray M, Mosher T, Dardzinski B. Measures of molecular composition and structure in osteoarthritis. Radiol Clin N Am. 2009;47(4):675–86.
Crema MD, Roemer FW, Marra MD, Burstein D, Gold GE, Eckstein F, et al. Articular cartilage in the knee: current MR imaging techniques and applications in clinical practice and research. Radiographics 2011;31(1):37–61.
Mosher TJ, Dardzinski BJ. Cartilage MRI T2 relaxation time mapping: overview and applications. Semin Musculoskelet Radiol. 2004;8(4):355–68.
Mamisch TC, Hughes T, Mosher TJ, Mueller C, Trattnig S, Boesch C, et al. T2 star relaxation times for assessment of articular cartilage at 3 T: a feasibility study. Skeletal Radiol. 2012;41(3):287–92.
Welsch GH, Mamisch TC, Hughes T, Zilkens C, Quirbach S, Scheffler K, et al. In vivo biochemical 7.0 Tesla magnetic resonance: preliminary results of dGEMRIC, zonal T2, and T2* mapping of articular cartilage. Invest Radiol. 2008;43(9):619–26.
Welsch GH, Trattnig S, Hughes T, Quirbach S, Olk A, Blanke M, et al. T2 and T2* mapping in patients after matrix-associated autologous chondrocyte transplantation: initial results on clinical use with 3.0-Tesla MRI. Eur Radiol. 2010;20(6):1515–23.
Bittersohl B, Hosalkar HS, Hughes T, Kim YJ, Werlen S, Siebenrock KA, et al. Feasibility of T2* mapping for the evaluation of hip joint cartilage at 1.5 T using a three-dimensional (3D), gradient-echo (GRE) sequence: a prospective study. Magnetic resonance in medicine : official journal of the Society of Magnetic Resonance in Medicine/Society of. Magn Reson Med. 2009;62(4):896–901.
Bittersohl B, Miese FR, Dekkers C, Senyurt H, Kircher J, Wittsack HJ, et al. T2* mapping and delayed gadolinium-enhanced magnetic resonance imaging in cartilage (dGEMRIC) of glenohumeral cartilage in asymptomatic volunteers at 3 T. Eur Radiol. 2013;23(5):1367–74.
Bittersohl B, Miese FR, Hosalkar HS, Herten M, Antoch G, Krauspe R, et al. T2* mapping of hip joint cartilage in various histological grades of degeneration. Osteoarthritis Cartilage. 2012;20(7):653–60.
Bittersohl B, Miese FR, Hosalkar HS, Mamisch TC, Antoch G, Krauspe R, et al. T2* mapping of acetabular and femoral hip joint cartilage at 3 T: a prospective controlled study. Invest Radiol. 2012;47(7):392–7.
Buchbender C, Scherer A, Kropil P, Korbl B, Quentin M, Reichelt D, et al. Cartilage quality in rheumatoid arthritis: comparison of T2* mapping, native T1 mapping, dGEMRIC, DeltaR1 and value of pre-contrast imaging. Skelet Radiol. 2012;41(6):685–92.
Marik W, Apprich S, Welsch GH, Mamisch TC, Trattnig S. Biochemical evaluation of articular cartilage in patients with osteochondrosis dissecans by means of quantitative T2- and T2-mapping at 3 T MRI: a feasibility study. Eur J Radiol. 2012;81(5):923–7.
Miese FR, Zilkens C, Holstein A, Bittersohl B, Kropil P, Mamisch TC, et al. Assessment of early cartilage degeneration after slipped capital femoral epiphysis using T2 and T2* mapping. Acta Radiol. 2011;52(1):106–10.
Newbould RD, Miller SR, Toms LD, Swann P, Tielbeek JA, Gold GE, et al. T2* measurement of the knee articular cartilage in osteoarthritis at 3 T. J Magn Reson Imaging. 2012;35(6):1422–9.
Pai A, Li X, Majumdar S. A comparative study at 3 T of sequence dependence of T2 quantitation in the knee. Magn Reson Imaging. 2008;26(9):1215–20.
Williams A, Qian Y, Chu CR. UTE-T2 * mapping of human articular cartilage in vivo: a repeatability assessment. Osteoarthritis Cartilage. 2011;19(1):84–8.
Mosher TJ, Dardzinski BJ, Smith MB. Human articular cartilage: influence of aging and early symptomatic degeneration on the spatial variation of T2–preliminary findings at 3 T. Radiology. 2000;214(1):259–66.
Waterton JC, Solloway S, Foster JE, Keen MC, Gandy S, Middleton BJ, et al. Diurnal variation in the femoral articular cartilage of the knee in young adult humans. Magn Reson Med. 2000;43(1):126–32.
Liess C, Lusse S, Karger N, Heller M, Gluer CC. Detection of changes in cartilage water content using MRI T2-mapping in vivo. Osteoarthritis Cartilage. 2002;10(12):907–13.
Crema MD, Roemer FW, Marra MD, Guermazi A. Magnetic resonance imaging assessment of subchondral bone and soft tissues in knee osteoarthritis. Rheum Dis Clin N Am. 2009;35(3):557–77.
Peterfy CG, Schneider E, Nevitt M. The osteoarthritis initiative: report on the design rationale for the magnetic resonance imaging protocol for the knee. Osteoarthritis Cartilage. 2008;16(12):1433–41.
Stehling C, Lane NE, Nevitt MC, Lynch J, McCulloch CE, Link TM. Subjects with higher physical activity levels have more severe focal knee lesions diagnosed with 3 T MRI: analysis of a non-symptomatic cohort of the osteoarthritis initiative. Osteoarthritis Cartilage. 2010;18(6):776–86.
Stehling C, Liebl H, Krug R, Lane NE, Nevitt MC, Lynch J, et al. Patellar cartilage: T2 values and morphologic abnormalities at 3.0-T MR imaging in relation to physical activity in asymptomatic subjects from the osteoarthritis initiative. Radiology. 2010;254(2):509–20.
Kellgren JH, Lawrence JS. Radiological assessment of osteo-arthrosis. Ann Rheum Dis. 1957;16(4):494–502.
Rubenstein JD, Kim JK, Henkelman RM. Effects of compression and recovery on bovine articular cartilage: appearance on MR images. Radiology. 1996;201(3):843–50.
Xia Y. Magic-angle effect in magnetic resonance imaging of articular cartilage: a review. Invest Radiol. 2000;35(10):602–21.
Clark JM. Variation of collagen fiber alignment in a joint surface: a scanning electron microscope study of the tibial plateau in dog, rabbit, and man. J Orthop Res. 1991;9(2):246–57.
Goodwin DW, Wadghiri YZ, Zhu H, Vinton CJ, Smith ED, Dunn JF. Macroscopic structure of articular cartilage of the tibial plateau: influence of a characteristic matrix architecture on MRI appearance. AJR Am J Roentgenol. 2004;182(2):311–8.
Shiomi T, Nishii T, Nakata K, Tamura S, Tanaka H, Yamazaki Y, et al. Three-dimensional topographical variation of femoral cartilage T2 in healthy volunteer knees. Skelet Radiol. 2013;42(3):363–70.
Acknowledgements
This study was funded by a research grant from the “German Research Foundation” (Deutsche Forschungsgemeinschaft). The authors have full control of all primary data.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bittersohl, B., Hosalkar, H.S., Sondern, M. et al. Spectrum of T2* values in knee joint cartilage at 3 T: a cross-sectional analysis in asymptomatic young adult volunteers. Skeletal Radiol 43, 443–452 (2014). https://doi.org/10.1007/s00256-013-1806-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-013-1806-1