Abstract.
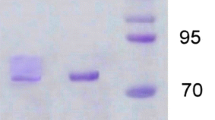
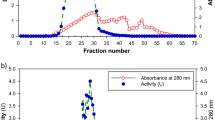
Ganoderma lucidum, a medicinal white-rot basidiomycete, produces many laccase isozymes in liquid culture. Three laccase isozymes (GaLc 1, 2, 3) have been purified 32.4-fold from the crude enzyme protein through anion exchange chromatography, preparative gel electrophoresis, and electroelution. Their estimated molecular weights are 65–68 kDa, and they contain 7–10% N-linked carbohydrates. The three isozymes have identical N-terminal amino acid sequences: G-I-G-P-T. The optimum pH and temperature both for each isozyme singly and the isozyme mixture are pH 3.5 and 20°C, respectively. One isozyme (GaLc 3) is quite stable at pH 4.0–10.0, and shows good stability when incubated at temperatures lower than 40°C. The K m values of GaLc 3 for o-tolidine and 2,2'-azino-bis-(3-ethylthiazoline-6-sulfonate) (ABTS) are 401.6 µM and 3.7 µM respectively, and the V max of GaLc 3 for these substrates is 0.0198 OD min–1unit–1 and 0.0142 OD min–1unit–1, respectively.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received revision: 18 April 2001
Electronic Publication
Rights and permissions
About this article
Cite this article
Ko, .EM., Leem, .YE. & Choi, .H. Purification and characterization of laccase isozymes from the white-rot basidiomycete Ganoderma lucidum . Appl Microbiol Biotechnol 57, 98–102 (2001). https://doi.org/10.1007/s002530100727
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002530100727