Abstract.
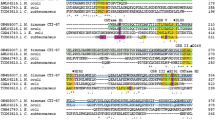
A region downstream of the gene for pullulan-hydrolyzing α-amylase, TVA II, of Thermoactinomyces vulgaris R-47 was sequenced, and an open reading frame encoding an enzyme homologous to glucoamylase was found. The nucleotide sequence of this enzyme, designated TGA, consists of 1,953 base pairs corresponding to a protein of 651 amino acid residues. The TGA gene was subcloned and expressed in Escherichia coli. Enzymatic analyses showed that, like other glucoamylases, TGA produced β-D-glucose from its substrate. However, TGA hydrolyzed maltooligosaccharides such as maltotetraose and maltose more efficiently than starch, while fungal glucoamylases preferred starch to maltooligosaccharides. The primary structure of TGA resembled a putative glucoamylase from the hyperthermophilic archaeon Methanococcus jannaschii (MGA), while homologies between TGA and the fungal glucoamylases were low. The enzymatic properties of recombinant MGA produced in E. coli cells were similar to those of TGA. These findings indicate that TGA and MGA are novel glucoamylase-type enzymes with oligosaccaharide-metabolizing activity.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received revision: 25 December 2000
Electronic Publication
Rights and permissions
About this article
Cite this article
Uotsu-Tomita, .R., Tonozuka, .T., Sakai, .H. et al. Novel glucoamylase-type enzymes from Thermoactinomyces vulgaris and Methanococcus jannaschii whose genes are found in the flanking region of the α-amylase genes. Appl Microbiol Biotechnol 56, 465–473 (2001). https://doi.org/10.1007/s002530100609
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002530100609