Abstract
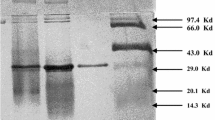
A novel raw starch degrading cyclomaltodextrin glucanotransferase (CGTase; E.C. 2.4.1.19), produced by Bacillus firmus, was purified to homogeneity by ultrafiltration, affinity and gel filtration chromatography. The molecular weight of the pure protein was estimated to be 78 000 and 82 000 Da, by SDS-PAGE and gel filtration, respectively. The pure enzyme had a pH optimum in the range 5.5–8.5. It was stable over the pH range 7–11 at 10 °C, and at pH 7.0 at 60 °C. The optimum temperature for enzyme activity was 65 °C. In the absence of substrate, the enzyme rapidly lost its activity above 30 °C. K m and k cat for the pure enzyme were 1.21 mg/ml and 145.17 μM/mg per minute respectively, with soluble starch as the substrate. For cyclodextrin production, tapioca starch was the best substrate used when gelatinized, while wheat starch was the best substrate used when raw. This CGTase could degrade raw wheat starch very efficiently; up to 50% conversion to cyclodextrins was obtained from 150 g/l starch without using any additives. The enzyme produced α-, β- and γ-cyclodextrins in the ratio of 0.2:9.2:0.6 and 0.2:8.6:1.2 from gelatinized tapioca starch and raw wheat starch with 150 g/l concentration respectively, after 18 h incubation.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 25 September 1998 / Received revision: 15 December 1998 / Accepted: 21 December 1998
Rights and permissions
About this article
Cite this article
Gawande, B., Goel, A., Patkar, A. et al. Purification and properties of a novel raw starch degrading cyclomaltodextrin glucanotransferase from Bacillus firmus . Appl Microbiol Biotechnol 51, 504–509 (1999). https://doi.org/10.1007/s002530051424
Issue Date:
DOI: https://doi.org/10.1007/s002530051424