Abstract
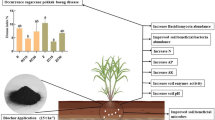
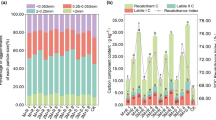
The continuous obstacles of cropping cause severe economic loss, which seriously threaten agricultural sustainable development. In addition, managing excess waste, such as potato peel and mineral waste residues, is a vital burden for industry and agriculture. Therefore, we explored the feasibility of reductive soil disinfestation (RSD) with potato peel and amendment with iron mineral waste residues for the production of Fritillaria thunbergii, which is vulnerable to continuous obstacles. In this study, the influences of iron mineral, RSD with different organic maters, as well as the combined effects of iron mineral and RSD on Fritillaria rhizosphere soil physicochemical properties, microbial communities, and Fritillaria production were investigated. The results revealed that the RSD treatments with potato peel significantly reduced the soil salinity and increased the soil pH, microbial activity, organic matter, and the contents of K and Ca. RSD with potato peel also significantly thrived of the beneficial microbes (Bacillus, Azotobacter, Microvirga, and Chaetomium), and down-regulated potential plant pathogens. RSD with potato peel significantly promoted F. thunbergii yield and quality. Moreover, the combined effects of RSD and iron mineral amendment further enhanced soil health, improved microbial community composition, and increased the yield and peimisine content of F. thunbergii by 24.2% and 49.3%, respectively. Overall, our results demonstrated that RSD with potato peel and amendment with iron mineral waste residues can efficiently improve soil fertility, modify the microbial community, and benefit for both the sustainable production of F. thunbergii and the management of waste.
Key points
• RSD increases soil pH, organic matter, microbial activity, and mineral content
• RSD with potato peel enriches beneficial microbes and decreases plant pathogens
• PP + Fe treatment increases Fritillaria yield by 24.2% and peimisine content by 49.3%






Similar content being viewed by others
Data availability
The original sequences have been deposited in the NCBI Sequence Read Archive (SRA) under BioProject ID number PRJNA916813.
References
Ab Rahman SFS, Singh E, Pieterse CMJ, Schenk PM (2018) Emerging microbial biocontrol strategies for plant pathogens. Plant Sci 267:102–111. https://doi.org/10.1016/j.plantsci.2017.11.012
Antonkiewicz J, Pełka R, Bik-Małodzińska M, Żukowska G, Gleń-Karolczyk K (2018) The effect of cellulose production waste and municipal sewage sludge on biomass and heavy metal uptake by a plant mixture. Environ Sci Pollut Res 25:31101–31112. https://doi.org/10.1007/s11356-018-3109-5
Chen C, Hall SJ, Coward E, Thompson A (2020) Iron-mediated organic matter decomposition in humid soils can counteract protection. Nat Commun 11:2255. https://doi.org/10.1038/s41467-020-16071-5
Desjardins AE, Manandhar HK, Plattner RD, Manandhar GG, Poling SM, Maragos CM (2000) Fusarium species from nepalese rice and production of mycotoxins and gibberellic acid by selected species. Appl Environ Microbiol 66(3):1020–1025. https://doi.org/10.1128/AEM.66.3.1020-1025.2000
Gu S, Wei Z, Shao Z, Friman VP, Cao K, Yang T, Kramer J, Wang X, Li M, Mei X, Xu Y, Shen Q, Kümmerli R, Jousset A (2020) Competition for iron drives phytopathogen control by natural rhizosphere microbiomes. Nat Microbiol 5(8):1002–1010. https://doi.org/10.1038/s41564-020-0719-8
Hakulinen N, Turunen O, Jnis J, Leisola M, Rouvinen J (2010) Three-dimensional structures of thermophilic beta-1,4-xylanases from Chaetomium thermophilum and Nonomuraea flexuosa. Comparison of twelve xylanases in relation to their thermal stability. Eur J Biochem 270(7):1399–1412. https://doi.org/10.1046/j.1432-1033.2003.03496.x
Haro R, Benito B (2019) The role of soil fungi in K+ plant nutrition. Int J Mol Sci 20(13):3169. https://doi.org/10.3390/ijms20133169
Huang XQ, Wen T, Zhang JB, Meng L, Zhu TB, Liu LL, Cai ZC (2015) Control of soil-borne pathogen Fusarium oxysporum by biological soil disinfestation with incorporation of various organic matters. Eur J Plant Pathol 143(2):223–235. https://doi.org/10.1007/s10658-015-0676-x
Huang XQ, Liu L, Wen T, Zhang J, Wang F, Cai Z (2016a) Changes in the soil microbial community after reductive soil disinfestation and cucumber seedling cultivation. Appl Microbiol Biot 100(12):5581–5593. https://doi.org/10.1007/s00253-016-7362-6
Huang XQ, Liu LL, Wen T, Zhang JB, Shen QR, Cai ZC (2016b) Reductive soil disinfestations combined or not with Trichoderma for the treatment of a degraded and Rhizoctonia solani infested greenhouse soil. Sci Hortic 206:51–61. https://doi.org/10.1007/s00253-016-7362-6
Huang XQ, Zhao J, Zhou X, Zhang J, Cai Z (2019) Differential responses of soil bacterial community and functional diversity to reductive soil disinfestation and chemical soil disinfestation. Geoderma 348:124–134. https://doi.org/10.1016/j.geoderma.2019.04.027
Ji C, Ye R, Yin Y, Sun X, Ma H, Gao R (2022) Reductive soil disinfestation with biochar amendment modified microbial community composition in soils under plastic greenhouse vegetable production. Soil Till Res 218. https://doi.org/10.1016/j.still.2022.105323
Jiménez-Gómez A, Saati-Santamaría Z, Igual JM, Rivas R, Mateos PF, García-Fraile P (2019) Genome insights into the novel species Microvirga brassicacearum, a rapeseed endophyte with biotechnological potential. Microorganisms 7(9). https://doi.org/10.3390/microorganisms7090354
Joong-Hyeop P, Ja CG, Soo JK, Kyoung LH, Tae KH, Yun CK, Jin-Cheol K (2005) Antifungal activity against plant pathogenic fungi of chaetoviridins isolated from Chaetomium globosum. Fems Microbiol Lett (2):309–313. https://doi.org/10.1016/j.femsle.2005.09.013
Kato A, Miyake T, Nishigata K, Tateishi H, Teraoka T, Arie T (2012) Use of fluorescent proteins to visualize interactions between the Bakanae disease pathogen Gibberella fujikuroi and the biocontrol agent Talaromyces sp. KNB-422. J Gen Plant Pathol 78(1):54–61. https://doi.org/10.1007/s10327-011-0343-9
Lahlali R, Peng G (2014) Suppression of clubroot by Clonostachys rosea via antibiosis and induced host resistance. Plant Pathol 63(2):447–455. https://doi.org/10.1111/ppa.12112
Lemaire G, Sinclair T, Sadras V, Bélanger G (2019) Allometric approach to crop nutrition and implications for crop diagnosis and phenotyping. A review. Agron Sustain Dev 39(2):27. https://doi.org/10.1007/s13593-019-0570-6
Li H, Yuan G, Zhu C, Zhao T, Zhang R, Wang X, Yang J, Ma J, Zhang Y, Zhang X (2019) Soil fumigation with ammonium bicarbonate or metam sodium under high temperature alleviates continuous cropping-induced Fusarium wilt in watermelon. Sci Hortic 246:979–986. https://doi.org/10.1016/j.scienta.2018.11.090
Li J, Wen S, Zhang Y, Jin L, Zhao W (2022) Identification of pathogenic fungi species of gray mold disease of Fritillaria thunbergii. Plant Prot 48(2):151–156
Liao H, Yunxia LI, Shao J, Fang F, Weidong GUO, Chen W (2011) Impacts of continuous cropping on Fritillaria thunbergii Miq. growth and rhizosphere soil properties. Chin J Ecol 30(10):2203–2208
Liu J, Wang S, Xin G, Li H, Li P (2010) Determination of Peimisine in Fritillaria taipaiensis and Fritillaria unibracteata by HPLC-ELSD. J Chin Pharm Sci 45(13):1032–1034
Liu H, Khan MY, Carvalhais LC, Delgado-Baquerizo M, Yan L, Crawford M, Dennis PG, Singh B, Schenk PM (2019) Soil amendments with ethylene precursor alleviate negative impacts of salinity on soil microbial properties and productivity. Sci Rep 9(1):6892. https://doi.org/10.1038/s41598-019-43305-4
Liu N, Shao C, Sun H, Liu Z, Guan Y, Wu L, Zhang L, Pan X, Zhang Z, Zhang Y, Zhang B (2020) Arbuscular mycorrhizal fungi biofertilizer improves American ginseng (Panax quinquefolius L.) growth under the continuous cropping regime. Geoderma 363:114155. https://doi.org/10.1016/j.geoderma.2019.114155
Liu Y, Bei K, Zheng W, Yu G, Sun C (2022) Assessment of health risks associated with pesticide and heavy metal contents in Fritillaria thunbergii Miq. (Zhe Beimu). Environ Sci Pollut Res. https://doi.org/10.1007/s11356-022-23995-6
Lopes DV, Sillanpää M, Wolkersdorfer C (2020) Nitrate reduction of the Siilinjärvi/Finland mine water with zero-valent iron and iron waste as alternative iron sources. Mine Water Environ 39(2):280–290. https://doi.org/10.1007/s10230-020-00668-9
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol Rev 37(5):634–663. https://doi.org/10.1111/1574-6976.12028
Meng T, Ren G, Wang G, Ma Y (2019) Impacts on soil microbial characteristics and their restorability with different soil disinfestation approaches in intensively cropped greenhouse soils. Appl Microbiol Biotechnol 103(15):6369–6383. https://doi.org/10.1007/s00253-019-09964-z
Miretzky P, Cirelli AF (2010) Remediation of arsenic-contaminated soils by iron amendments: a review. Crit Rev Environ Sci Technol 40(2):93–115. https://doi.org/10.1080/10643380802202059
Momma N, Kobara Y, Uematsu S, Kita N, Shinmura A (2013) Development of biological soil disinfestations in Japan. Appl Microbiol Biotechnol 97:3801–3809. https://doi.org/10.1007/s00253-013-4826-9
National Pharmacopoeia Committee. (2020) Pharmacopoeia of the People’s Republic of China. 2020 edition Part 1. Appendix 304
Nehra V, Choudhary M (2015) A review on plant growth promoting rhizobacteria acting as bioinoculants and their biological approach towards the production of sustainable agriculture. J Appl Nat Sci 7(1):540–556. https://doi.org/10.31018/jans.v7i1.642
Nelson DW, Sommers LE (1982) Total carbon, organic carbon and organic matter, in: Methods of soil analysis part 2. Chem Microbial Prop. https://doi.org/10.2134/agronmonogr9.2.2ed.c29
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling JS, Kennedy PG (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248. https://doi.org/10.1016/j.funeco.2015.06.006
Nile SH, Su J, Wu D, Wang L, Hu J, Sieniawska E, Kai G (2021) Fritillaria thunbergii Miq. (Zhe Beimu): a review on its traditional uses, phytochemical profile and pharmacological properties. Food Chem Toxicol 153:112289. https://doi.org/10.1016/j.fct.2021.112289
Nilsson RH, Larsson KH, Taylor AFS, Bengtsson-Palme J, Jeppesen TS, Schigel D, Kennedy P, Picard K, Glöckner FO, Tedersoo L, Saar I, Kõljalg U, Abarenkov K (2019) The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res 47(D1):D259-d264. https://doi.org/10.1093/nar/gky1022
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin P, O’Hara RB, Simpson G, Solymos P, Stevens MHN, Wagner H (2012) Package ‘vegan’—Community Ecology Package, version 2.0–4. https://CRAN.R-project.org/package=vegan
Ondrasek G, Bakić Begić H, Zovko M, Filipović L, Meriño-Gergichevich C, Savić R, Rengel Z (2019) Biogeochemistry of soil organic matter in agroecosystems and environmental implications. Sci Total Environ 658:1559–1573. https://doi.org/10.1016/j.scitotenv.2018.12.243
Philippot L, Raaijmakers JM, Lemanceau P, van der Putten WH (2013) Going back to the roots: the microbial ecology of the rhizosphere. Nat Rev Microbiol 11(11):789–799. https://doi.org/10.1038/nrmicro3109
Read JJ, Reddy KR, Jenkins JN (2006) Yield and fiber quality of Upland cotton as influenced by nitrogen and potassium nutrition. Eur J Agron 24(3):282–290. https://doi.org/10.1016/j.eja.2005.10.004
Sattar A, Naveed M, Ali M, Zahir ZA, Nadeem SM, Yaseen M, Meena VS, Farooq M, Singh R, Rahman M, Meena HN (2019) Perspectives of potassium solubilizing microbes in sustainable food production system: a review. Appl Soil Ecol 133:146–159. https://doi.org/10.1016/j.apsoil.2018.09.012
Schnürer J, Rosswall T (1982) Fluorescein diacetate hydrolysis as a measure of total microbial activity in soil and litter. Appl Environ Microbiol 43(6):1256–1261. https://doi.org/10.1128/aem.43.6.1256-1261.1982
Schoch CL, Sung GH, López-Giráldez F, Townsend JP, Spatafora JW (2009) The Ascomycota tree of life: a phylum-wide phylogeny clarifies the origin and evolution of fundamental reproductive and ecological traits. Syst Biol 58(2):224–239. https://doi.org/10.1093/sysbio/syp020
Shafi J, Tian H, Ji M (2017) Bacillus species as versatile weapons for plant pathogens: a review. Biotechnol Biotechnol Equip 31(3):446–459. https://doi.org/10.1080/13102818.2017.1286950
Sterkenburg E, Bahr A, Brandstrom Durling M, Clemmensen KE, Lindahl BD (2015) Changes in fungal communities along a boreal forest soil fertility gradient. New Phytol 207(4):1145–1158. https://doi.org/10.1111/nph.13426
Su L, Deng H, Niu Y-C (2017) Phylogenetic analysis of Plectosphaerella species based on multi-locus DNA sequences and description of P. sinensis sp. nov. Mycol Prog 16(8):823–829. https://doi.org/10.1007/s11557-017-1319-8
Turner S, Pryer KM, Miao VP, Palmer JD (1999) Investigating deep phylogenetic relationships among cyanobacteria and plastids by small submit rRNA sequence analysis. J Eukaryot Microbiol 46:327–338. https://doi.org/10.1111/j.1550-7408.1999.tb04612.x
Walters W, Hyde ER, Berg-Lyons D, Ackermann G, Humphrey G, Parada A, Gilbert JA, Jansson JK, Caporaso JG, Fuhrman JA, Apprill A, Knight R (2016) Improved bacterial 16S rRNA gene (V4 and V4–5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. mSystems 1(1). https://doi.org/10.1128/mSystems.00009-15
Wang F, Wang F, Zhang H, Qin F, Xiang W, Wu C, Yan C, Zhu Z, Chen J, Ge T (2022) Deciphering differences in microbial community composition and multifunctionality between healthy and Alternaria solani-infected potato rhizosphere soils. Plant Soil. https://doi.org/10.1007/s11104-022-05797-4
Wu F, Tian M, Sun Y, Wu C, Liu X (2022) Efficacy, chemical composition, and pharmacological effects of herbal drugs derived from Fritillaria cirrhosa D. Don and Fritillaria thunbergii Miq. Front Pharmacol 13. https://doi.org/10.3389/fphar.2022.985935
Xiang L, Harindintwali JD, Wang F, Redmile-Gordon M, Chang SX, Fu Y, He C, Muhoza B, Brahushi F, Bolan N, Jiang X, Ok YS, Rinklebe J, Schaeffer A, Zhu YG, James MT, Xing B (2022) Integrating biochar, bacteria, and plants for sustainable remediation of soils contaminated with organic pollutants. Environ Sci Technol. https://doi.org/10.1021/acs.est.2c02976
Xiong W, Li R, Ren Y, Liu C, Zhao Q, Wu H, Jousset A, Shen Q (2017) Distinct roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Boil Biochem 107:198–207. https://doi.org/10.1016/j.soilbio.2017.01.010
Yang W, Zhang D, Cai X, Xia L, Luo Y, Cheng X, An S (2019) Significant alterations in soil fungal communities along a chronosequence of Spartina alterniflora invasion in a Chinese Yellow Sea coastal wetland. Sci Total Environ 693:133548. https://doi.org/10.1016/j.scitotenv.2019.07.354
Yang G, Xu C, Varjani S, Zhou Y, Wc Wong J, Duan G (2022) Metagenomic insights into improving mechanisms of Fe(0) nanoparticles on volatile fatty acids production from potato peel waste anaerobic fermentation. Bioresour Technol 361:127703. https://doi.org/10.1016/j.biortech.2022.127703
You J, Liu X, Zhang B, Xie Z, Hou Z, Yang Z (2015) Seasonal changes in soil acidity and related properties in ginseng artificial bed soils under a plastic shade. J Ginseng Res 39(1):81–88. https://doi.org/10.1016/j.jgr.2014.08.002
Zhao J, Zhou X, Jiang A, Fan J, Lan T, Zhang J, Cai Z (2018) Distinct impacts of reductive soil disinfestation and chemical soil disinfestation on soil fungal communities and memberships. Appl Microbiol Biotechnol 102(17):7623–7634. https://doi.org/10.1007/s00253-018-9107-1
Funding
This study was supported by Pan’an County Chinese Medicine Industry Project (grant no.PZYF202103) and the National Natural Science Foundation of China (grant no. 41991332).
Author information
Authors and Affiliations
Contributions
Y.Y., J.Z., P.W., G.L.D, and Y.G.Z. conceived and designed the investigation. Y.Y., P.W., and H.M.D. conducted the experiments. Y.Y. analyzed data and wrote the manuscript. J.Z., S.M.Y., Y.Y.Z., J.W.W., G.L.D., and Y.G.Z reviewed and edited the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, Y., Zhao, J., Wang, P. et al. Reductive soil disinfestation and Fe amendment improve soil microbial composition and Fritillaria production. Appl Microbiol Biotechnol 107, 6703–6716 (2023). https://doi.org/10.1007/s00253-023-12766-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12766-z