Abstract
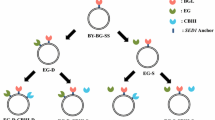
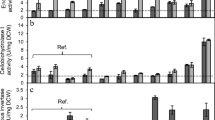
Consolidated bioprocessing (CBP) remains an attractive option for the production of commodity products from pretreated lignocellulose if a process-suitable organism can be engineered. The yeast Saccharomyces cerevisiae requires engineered cellulolytic activity to enable its use in CBP production of second-generation (2G) bioethanol. A promising strategy for heterologous cellulase production in yeast entails displaying enzymes on the cell surface by means of glycosylphosphatidylinositol (GPI) anchors. While strains producing a core set of cell-adhered cellulases that enabled crystalline cellulose hydrolysis have been created, secreted levels of enzyme were insufficient for complete cellulose hydrolysis. In fact, all reported recombinant yeast CBP candidates must overcome the drawback of generally low secretion titers. Rational strain engineering can be applied to enhance the secretion phenotype. This study aimed to improve the amount of cell-adhered cellulase activities of recombinant S. cerevisiae strains expressing a core set of four cellulases, through overexpression of genes that were previously shown to enhance cellulase secretion. Results showed significant increases in cellulolytic activity for all cell-adhered cellulase enzyme types. Cell-adhered cellobiohydrolase activity was improved by up to 101%, β-glucosidase activity by up to 99%, and endoglucanase activity by up to 231%. Improved hydrolysis of crystalline cellulose of up to 186% and improved ethanol yields from this substrate of 40–50% in different strain backgrounds were also observed. In addition, improvement in resistance to fermentation stressors was noted in some strains. These strains represent a step towards more efficient organisms for use in 2G biofuel production.
Key points
• Cell-surface–adhered cellulase activity was improved in strains engineered for CBP.
• Levels of improvement of activity were strain and enzyme dependent.
• Crystalline cellulose conversion to ethanol could be improved up to 50%.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Bai FW, Zhao XQ (2012) High gravity ethanol fermentations and yeast tolerance. In: Liu Z (eds) Microbial stress tolerance for biofuels. Microbiology Monographs, vol 22. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-21467-7_5
Bull VH, Thiede B (2012) Proteome analysis of tunicamycin-induced ER stress. Electrophor 33(12):1814–1823. https://doi.org/10.1002/elps.201100565
Carvalho-Netto OV, Carazzolle MF, Mofatto LS, Teixeira PJ, Noronha MF, Calderón LA, Mieczkowski PA, Argueso JL, Pereira GA (2015) Saccharomyces cerevisiae transcriptional reprograming due to bacterial contamination during industrial scale bioethanol production. Microb Cell Fact 14:13. https://doi.org/10.1186/s12934-015-0196-6
Chandel AK, Albarelli JQ, Santos DT, Chundawat SP, Puri M, Meireles MAA (2019) Comparative analysis of key technologies for cellulosic ethanol production from Brazilian sugarcane bagasse at a commercial scale. Biofuels, Bioprod Biorefin 13(4):994–1014. https://doi.org/10.1002/bbb.1990
Claes A, Deparis Q, Foulquié-Moreno MR, Thevelein JM (2020) Simultaneous secretion of seven lignocellulolytic enzymes by an industrial second-generation yeast strain enables efficient ethanol production from multiple polymeric substrates. Metab Eng 59:131–141. https://doi.org/10.1016/j.ymben.2020.02.004
Davison SA, Den Haan R, Van Zyl WH (2016) Heterologous expression of cellulase genes in natural Saccharomyces cerevisiae strains. Appl Microbiol Biotechnol 100(18):8241–8254. https://doi.org/10.1007/s00253-016-7735-x
Davison SA, Den Haan R, van Zyl WH (2019) Identification of superior cellulase secretion phenotypes in haploids derived from natural Saccharomyces cerevisiae isolates. FEMS Yeast Res 19(2):foy117. https://doi.org/10.1093/femsyr/foy117
Davison SA, Den Haan R, van Zyl WH (2020) Exploiting strain diversity and rational engineering strategies to enhance recombinant cellulase secretion by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 104(12):5163–5184. https://doi.org/10.1007/s00253-020-10602-2
De Ruijter JC, Koskela EV, Frey AD (2016) Enhancing antibody folding and secretion by tailoring the Saccharomyces cerevisiae endoplasmic reticulum. Microb Cell Fact 15:87. https://doi.org/10.1186/s12934-016-0488-5
Den Haan R, Rose SH, Lynd LR, Van Zyl WH (2007) Hydrolysis and fermentation of amorphous cellulose by recombinant Saccharomyces cerevisiae. Metab Eng 9(1):87–94. https://doi.org/10.1016/j.ymben.2006.08.005
Den Haan R, Kroukamp H, van Zyl JH, Van Zyl WH (2013) Cellobiohydrolase secretion by yeast: Current state and prospects for improvement. Process Biochem 48(1):1–12. https://doi.org/10.1016/j.procbio.2012.11.015
Den Haan R, van Zyl JM, Harms TM, Van Zyl WH (2013) Modeling the minimum enzymatic requirements for optimal cellulose conversion. Environ Res Lett 8:025013. https://doi.org/10.1088/1748-9326/8/2/025013
Den Haan R, van Rensburg E, Rose SH, Gorgens JF, Van Zyl WH (2015) Progress and challenges in the engineering of non-cellulolytic microorganisms for consolidated bioprocessing. Curr Opin Biotechnol 33:32–38. https://doi.org/10.1016/j.copbio.2014.10.003
Den Haan R, Rose SH, Cripwell RA, Trollope KM, Myburgh MW, Viljoen-Bloom M, van Zyl WH (2021) Heterologous production of cellulose- and starch-degrading hydrolases to expand Saccharomyces cerevisiae substrate utilization: lessons learnt. Biotechnol Adv 53:107859. https://doi.org/10.1016/j.biotechadv.2021.107859
Deparis Q, Claes A, Foulquie-Moreno MR, Thevelein JM (2017) Engineering tolerance to industrially relevant stress factors in yeast cell factories. FEMS Yeast Res 17(4):fox036. https://doi.org/10.1093/femsyr/fox036
Favaro L, Jansen T, Van Zyl WH (2019) Exploring industrial and natural Saccharomyces cerevisiae strains for the bio-based economy from biomass: the case of bioethanol. Crit Rev Biotechnol 39(6):800–816. https://doi.org/10.1080/07388551.2019.1619157
Hannula I, Reiner D (2019) Near-term potential of biofuels, electrofuels, and battery electric vehicles in decarbonizing road transport. Joule 3:2390–2402. https://doi.org/10.1016/j.joule.2019.08.013
Hasunuma T, Kondo A (2012) Development of yeast cell factories for consolidated bioprocessing of lignocellulose to bioethanol through cell surface engineering. Biotechnol Adv 30(6):1207–1218. https://doi.org/10.1016/j.biotechadv.2011.10.011
Hill J, Ian KA, Donald G, Griffiths DE (1991) DMSO-enhanced whole cell yeast transformation. Nucleic Acids Res 19:5791–5791. https://doi.org/10.1093/nar/19.20.5791
Idiris A, Tohda H, Kumagai H, Takegawa K (2010) Engineering of protein secretion in yeast: strategies and impact on protein production. Appl Microbiol Biotechnol 86(2):403–417. https://doi.org/10.1007/s00253-010-2447-0
Ilmén M, Den Haan R, Brevnova E, McBride J, Wiswall E, Froehlich A, Koivula A, Voutilainen SP, Siika-aho M, Lagrange DC, Thorngren N, Ahlgren S, Mellon M, Deleault K, Rajgarhia V, Van Zyl WH, Penttila M (2011) High level secretion of cellobiohydrolases by Saccharomyces cerevisiae. Biotechnol Biofuels 4(1):30. https://doi.org/10.1186/1754-6834-4-30
Inokuma K, Hasunuma T, Kondo A (2014) Efficient yeast cell-surface display of exo- and endo-cellulase using the SED1 anchoring region and its original promoter. Biotechnol Biofuels 7(1):8. https://doi.org/10.1186/1754-6834-7-8
Klinke HB, Thomsen AB, Ahring BK (2004) Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl Microbiol Biotechnol 66(1):10–26. https://doi.org/10.1007/s00253-004-1642-2
Kroukamp H, Den Haan R, van Wyk N, Van Zyl WH (2013) Overexpression of native PSE1 and SOD1 in Saccharomyces cerevisiae improved heterologous cellulase secretion. Appl Energ 102:150–156. https://doi.org/10.1016/j.apenergy.2012.05.062
Kroukamp H, Den Haan R, van Zyl J-H, van Zyl WH (2018) Rational strain engineering interventions to enhance cellulase secretion by Saccharomyces cerevisiae. Biofuels, Bioprod Biorefin 12(1):108–124. https://doi.org/10.1002/bbb.1824
Kroukamp H, Den Haan R, la Grange DC, Sibanda N, Foulquie-Moreno MR, Thevelein JM, van Zyl WH (2017) Strain breeding enhanced heterologous cellobiohydrolase secretion by Saccharomyces cerevisiae in a protein specific manner. Biotechnol J 12(10). https://doi.org/10.1002/biot.201700346
Li M, Xu J, Xie H, Wang Y (2018) Transport biofuels technological paradigm based conversion approaches towards a bio-electric energy framework. Energy Convers Manag 172:554–566. https://doi.org/10.1016/jenconman201807049
Liu Z, Liu L, Österlund T, Hou J, Huang M, Fagerberg L, Petranovic D, Uhlén M, Nielsen J (2014) Improved production of a heterologous amylase in Saccharomyces cerevisiae by inverse metabolic engineering. Appl Environ Microbiol 80(17):5542–5550. https://doi.org/10.1128/aem.00712-14
Liu Z, Inokuma K, Ho SH, Den Haan R, Hasunuma T, Van Zyl WH, Kondo A (2015) Combined cell-surface display- and secretion-based strategies for production of cellulosic ethanol with Saccharomyces cerevisiae. Biotechnol Biofuels 8:162. https://doi.org/10.1186/s13068-015-0344-6
Liu Z, Ho SH, Hasunuma T, Chang JS, Ren NQ, Kondo A (2016) Recent advances in yeast cell-surface display technologies for waste biorefineries. Biores Technol 15:324–333. https://doi.org/10.1016/j.biortech.2016.03.132
Liu Z, Ho SH, Sasaki K, Den Haan R, Inokuma K, Ogino C, van Zyl WH, Hasunuma T, Kondo A (2016) Engineering of a novel cellulose-adherent cellulolytic Saccharomyces cerevisiae for cellulosic biofuel production. Sci Rep 6:24550. https://doi.org/10.1038/srep24550
Liu Z, Inokuma K, Ho SH, Den Haan R, van Zyl WH, Hasunuma T, Kondo A (2017) Improvement of ethanol production from crystalline cellulose via optimizing cellulase ratios in cellulolytic Saccharomyces cerevisiae. Biotechnol Bioeng 114(6):1201–1207. https://doi.org/10.1002/bit.26252
Liu H, Sun J, Chang JS, Shukla P (2018) Engineering microbes for direct fermentation of cellulose to bioethanol. Crit Rev Biotechnol 38(7):1089–1105. https://doi.org/10.1080/07388551.2018.1452891
Lynd LR, Weimer PJ, Van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577. https://doi.org/10.1128/MMBR.66.3.506-577.2002
Menon VA, Rao M (2012) Trends in bioconversion of lignocellulose: Biofuels, platform chemicals & biorefinery concept. Prog Energy Combust Sci 38:522–550. https://doi.org/10.1016/J.PECS.2012.02.002
Mukherjee V, Steensels J, Lievens B, Voorde I, Verplaetse A, Aerts G (2014) Phenotypic evaluation of natural and industrial Saccharomyces yeasts for different traits desirable in industrial bioethanol production. Appl Microbiol Biotechnol 98:9483–9498. https://doi.org/10.1007/s00253-014-6090-z
Phienluphon A, Mhuantong W, Boonyapakron K, Deenarn P, Champreda V, Wichadakul D, Suwannarangsee S (2019) Identification and evaluation of novel anchoring proteins for cell surface display on Saccharomyces cerevisiae. Appl Microbiol Biotechnol 103(7):3085–3097. https://doi.org/10.1007/s00253-019-09667-5
Saini JK, Saini R, Tewari L (2015) Lignocellulosic agriculture wastes as biomass feedstocks for second-generation bioethanol production: concepts and recent developments. 3 Biotech 5(4):337–353. https://doi.org/10.1007/s13205-014-0246-5
Sakwa L, Cripwell RA, Rose SH, Viljoen-Bloom M (2018) Consolidated bioprocessing of raw starch with Saccharomyces cerevisiae strains expressing fungal alpha-amylase and glucoamylase combinations. FEMS Yeast Res 18(7):foy085. https://doi.org/10.1093/femsyr/foy085
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2 edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y
Stanley D, Bandara A, Fraser S, Chambers PJ, Stanley GA (2010) The ethanol stress response and ethanol tolerance of Saccharomyces cerevisiae. J Appl Microbiol 109(1):13–24. https://doi.org/10.1111/j.1365-2672.2009.04657.x
Van Zyl WH, Bloom M, Viktor MJ (2012) Engineering yeasts for raw starch conversion. Appl Microbiol Biotechnol 95(6):1377–1388. https://doi.org/10.1007/s00253-012-4248-0
Van Zyl JH, Den Haan R, Van Zyl WH (2014) Over-expression of native Saccharomyces cerevisiae exocytic SNARE genes increased heterologous cellulase secretion. Appl Microbiol Biotechnol 98(12):5567–5578. https://doi.org/10.1007/s00253-014-5647-1
Van Zyl JH, Den Haan R, Van Zyl WH (2016) Overexpression of native Saccharomyces cerevisiae ER-to-Golgi SNARE genes increased heterologous cellulase secretion. Appl Microbiol Biotechnol 100:505–518. https://doi.org/10.1007/s00253-015-7022-2
van Rensburg E, den Haan R, Smith J, van Zyl WH, Görgens JF (2012) The metabolic burden of cellulase expression by recombinant Saccharomyces cerevisiae Y294 in aerobic batch culture. Appl Microbiol Biotechnol 96:197–209. https://doi.org/10.1007/s00253-012-4037-9
Wu Y, Sun X, Xue X, Luo H, Yao B, Xie X, Su X (2017) Overexpressing key component genes of the secretion pathway for enhanced secretion of an Aspergillus niger glucose oxidase in Trichoderma reesei. Enzyme Microb Technol 106:83–87. https://doi.org/10.1016/j.enzmictec.2017.07.007
Xia J, Yang Y, Liu CG, Yang S, Bai FW (2019) Engineering Zymomonas mobilis for robust cellulosic ethanol production. Trends Biotechnol 37(9):960–972. https://doi.org/10.1016/j.tibtech.2019.02.002
Xu L, Shen Y, Hou J, Peng B, Tang H, Bao X (2014) Secretory pathway engineering enhances secretion of cellobiohydrolase I from Trichoderma reesei in Saccharomyces cerevisiae. J Biosci Bioeng 117(1):45–52. https://doi.org/10.1016/j.jbiosc.2013.06.017
Xu Q, Alahuhta M, Wei H, Knoshaug EP, Wang W, Baker JO, Vander Wall T, Himmel ME, Zhang M (2018) Expression of an endoglucanase–cellobiohydrolase fusion protein in Saccharomyces cerevisiae, Yarrowia lipolytica, and Lipomyces starkeyi. Biotechnol Biofuels 11(1):322. https://doi.org/10.1186/s13068-018-1301-y
Yamada R, Taniguchi N, Tanaka T, Ogino C, Fukuda H, Kondo A (2010) Cocktail delta-integration: a novel method to construct cellulolytic enzyme expression ratio-optimized yeast strains. Microb Cell Fact 9:32. https://doi.org/10.1186/1475-2859-9-32
Acknowledgements
This project was funded by the National Research Foundation (South Africa) grant nr. 118894 and grant nr. 92798. In addition, the work was supported by the Japan Society for the Promotion of Science (JSPS) under the JSPS—NRF Joint Research Program (Grant Number JPJSBP120196503).
Author information
Authors and Affiliations
Contributions
RDH, WHVZ, KI, and TH conceived and designed research. BJC and RDH conducted experiments. KI and TH contributed yeast strains and analytical procedures. BJC and RDH analyzed data and wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with animals or human participants performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chetty, B.J., Inokuma, K., Hasunuma, T. et al. Improvement of cell-tethered cellulase activity in recombinant strains of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 106, 6347–6361 (2022). https://doi.org/10.1007/s00253-022-12114-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12114-7