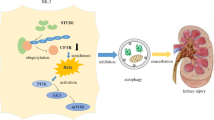
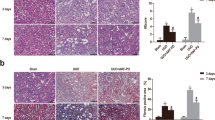
Abstract
Oxalate-induced crystalline kidney injury is one of the most common types of crystalline nephropathy. Unfortunately, there is no effective treatment to reduce the deposition of calcium oxalate crystals and alleviate kidney damage. Thus, proactive therapeutic is urgently needed to alleviate the suffering it causes to patient. Here, we investigated whether IL-22 exerted nephroprotective effects to sodium oxalate-mediated kidney damage and its potential mechanism. Crystalline kidney injury models were developed in vitro and in vivo that was often observed in clinic. We provided evidence that IL-22 could effectively decrease the accumulation of ROS and mitochondrial damage in cell and animal models and reduce the death of TECs. Moreover, IL-22 decreased the expression of the NLRP3 inflammasome and mature IL-1β in renal tissue induced by sodium oxalate. Further studies confirmed that IL-22 could play an anti-inflammatory role by reducing the levels of cytokines such as IL-1β, IL-18, and TNF-α in serum. In conclusion, our study confirmed that IL-22 has protective effects on sodium oxalate-induced crystalline kidney injury by reducing the production of ROS, protecting mitochondrial membrane potential, and inhibiting the inflammatory response. Therefore, IL-22 may play a potential preventive role in sodium oxalate-induced acute renal injury.
Key points
• IL-22 could reduce sodium oxalate-mediated cytotoxicity and ameliorate renal injury.
• IL-22 could alleviate oxidative stress and mitochondrial dysfunction induced by sodium oxalate.
• IL-22 could inhibit inflammatory response of renal injury caused by sodium oxalate.






Similar content being viewed by others
References
Anders HJ, Suarez-Alvarez B, Grigorescu M, Foresto-Neto O, Steiger S, Desai J, Marschner JA, Honarpisheh M, Shi C, Jordan J, Müller L, Burzlaff N, Bäuerle T, Mulay SR (2018) The macrophage phenotype and inflammasome component NLRP3 contributes to nephrocalcinosis-related chronic kidney disease independent from IL-1-mediated tissue injury. Kidney Int 93(3):656–669. https://doi.org/10.1016/j.kint.2017.09.022
Baker RG, Hayden MS, Ghosh S (2011) NF-κB, inflammation, and metabolic disease. Cell Metab 13(1):11–22. https://doi.org/10.1016/j.cmet.2010.12.008
Borghi M, De Luca A, Puccetti M, Jaeger M, Mencacci A, Oikonomou V, Pariano M, Garlanda C, Moretti S, Bartoli A, Sobel J, van de Veerdonk FL, Dinarello CA, Netea MG, Romani L (2015) Pathogenic NLRP3 inflammasome activity during candida infection is negatively regulated by IL-22 via activation of NLRC4 and IL-1Ra. Cell Host Microbe 18(2):198–209. https://doi.org/10.1016/j.chom.2015.07.004
Chaiyarit S, Thongboonkerd V (2012) Changes in mitochondrial proteome of renal tubular cells induced by calcium oxalate monohydrate crystal adhesion and internalization are related to mitochondrial dysfunction. J Proteome Res 11(6):3269–3280. https://doi.org/10.1021/pr300018c
Che Y, Tian Y, Chen R, Xia L, Liu F, Su Z (2021) IL-22 ameliorated cardiomyocyte apoptosis in cardiac ischemia/reperfusion injury by blocking mitochondrial membrane potential decrease, inhibiting ROS and cytochrome C. Biochim Biophys Acta Mol basis Dis 1867(9):166171 doi:https://doi.org/10.1016/j.bbadis.2021.166171
Chen W, Zhang X, Fan J, Zai W, Luan J, Li Y, Wang S, Chen Q, Wang Y, Liang Y, Ju D (2017) Tethering interleukin-22 to apolipoprotein A-I ameliorates mice from acetaminophen-induced liver injury. Theranostics 7(17):4135–4148. https://doi.org/10.7150/thno.20955
Chen W, Luan J, Wei G, Zhang X, Fan J, Zai W, Wang S, Wang Y, Liang Y, Nan Y, Yin C, Li Y, Liu ML, Ju D (2018a) In vivo hepatocellular expression of interleukin-22 using penetratin-based hybrid nanoparticles as potential anti-hepatitis therapeutics. Biomaterials 187:66–80. https://doi.org/10.1016/j.biomaterials.2018.09.046
Chen Y, Jin S, Teng X, Hu Z, Zhang Z, Qiu X, Tian D, Wu Y (2018b) Hydrogen sulfide attenuates LPS-induced acute kidney injury by inhibiting inflammation and oxidative stress. Oxidative Med Cell Longev 2018:6717212. https://doi.org/10.1155/2018/6717212
Chen W, Shen Y, Fan J, Zeng X, Zhang X, Luan J, Wang Y, Zhang J, Fang S, Mei X, Zhao Z, Ju D (2021) IL-22-mediated renal metabolic reprogramming via PFKFB3 to treat kidney injury. Clin Transl Med 11(2):e324. https://doi.org/10.1002/ctm2.324
Cho KA, Suh JW, Lee KH, Kang JL, Woo SY (2012) IL-17 and IL-22 enhance skin inflammation by stimulating the secretion of IL-1β by keratinocytes via the ROS-NLRP3-caspase-1 pathway. Int Immunol 24(3):147–158. https://doi.org/10.1093/intimm/dxr110
Convento MB, Pessoa EA, Cruz E, da Glória MA, Schor N, Borges FT (2017) Calcium oxalate crystals and oxalate induce an epithelial-to-mesenchymal transition in the proximal tubular epithelial cells: contribution to oxalate kidney injury. Sci Rep 7:45740. https://doi.org/10.1038/srep45740
Dela Peña A, Leclercq I, Field J, George J, Jones B, Farrell G (2005) NF-kappaB activation, rather than TNF, mediates hepatic inflammation in a murine dietary model of steatohepatitis. Gastroenterology 129(5):1663–1674. https://doi.org/10.1053/j.gastro.2005.09.004
Diao C, Chen Z, Qiu T, Liu H, Yang Y, Liu X, Wu J, Wang L (2019) Inhibition of PRMT5 attenuates oxidative stress-induced pyroptosis via activation of the Nrf2/HO-1 signal pathway in a mouse model of renal ischemia-reperfusion injury. Oxidative Med Cell Longev 2019:2345658. https://doi.org/10.1155/2019/2345658
Duan X, Kong Z, Mai X, Lan Y, Liu Y, Yang Z, Zhao Z, Deng T, Zeng T, Cai C, Li S, Zhong W, Wu W, Zeng G (2018) Autophagy inhibition attenuates hyperoxaluria-induced renal tubular oxidative injury and calcium oxalate crystal depositions in the rat kidney. Redox Biol 16:414–425. https://doi.org/10.1016/j.redox.2018.03.019
Duewell P, Kono H, Rayner KJ, Sirois CM, Vladimer G, Bauernfeind FG, Abela GS, Franchi L, Nuñez G, Schnurr M, Espevik T, Lien E, Fitzgerald KA, Rock KL, Moore KJ, Wright SD, Hornung V, Latz E (2010) NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464(7293):1357–1361. https://doi.org/10.1038/nature08938
Evan AP, Worcester EM, Coe FL, Williams J, Jr., Lingeman JE (2015) Mechanisms of human kidney stone formation. Urolithiasis 43 Suppl 1(0 1):19-32 doi:https://doi.org/10.1007/s00240-014-0701-0
Eyerich S, Eyerich K, Cavani A, Schmidt-Weber C (2010) IL-17 and IL-22: siblings, not twins. Trends Immunol 31(9):354–361. https://doi.org/10.1016/j.it.2010.06.004
Gan QZ, Sun XY, Bhadja P, Yao XQ, Ouyang JM (2016) Reinjury risk of nano-calcium oxalate monohydrate and calcium oxalate dihydrate crystals on injured renal epithelial cells: aggravation of crystal adhesion and aggregation. Int J Nanomedicine 11:2839–2854. https://doi.org/10.2147/ijn.S104505
Geng H, Bu HF, Liu F, Wu L, Pfeifer K, Chou PM, Wang X, Sun J, Lu L, Pandey A, Bartolomei MS, De Plaen IG, Wang P, Yu J, Qian J, Tan XD (2018) In Inflamed intestinal tissues and epithelial cells, interleukin 22 signaling increases expression of H19 long noncoding RNA, which promotes mucosal regeneration. Gastroenterology 155(1):144–155. https://doi.org/10.1053/j.gastro.2018.03.058
Halasi M, Wang M, Chavan TS, Gaponenko V, Hay N, Gartel AL (2013) ROS inhibitor N-acetyl-L-cysteine antagonizes the activity of proteasome inhibitors. Biochem J 454(2):201–208. https://doi.org/10.1042/bj20130282
Hasegawa M, Yada S, Liu MZ, Kamada N, Muñoz-Planillo R, Do N, Núñez G, Inohara N (2014) Interleukin-22 regulates the complement system to promote resistance against pathobionts after pathogen-induced intestinal damage. Immunity 41(4):620–632. https://doi.org/10.1016/j.immuni.2014.09.010
Hirose M, Yasui T, Okada A, Hamamoto S, Shimizu H, Itoh Y, Tozawa K, Kohri K (2010) Renal tubular epithelial cell injury and oxidative stress induce calcium oxalate crystal formation in mouse kidney. Int J Urol 17(1):83–92. https://doi.org/10.1111/j.1442-2042.2009.02410.x
Keir M, Yi Y, Lu T, Ghilardi N (2020) The role of IL-22 in intestinal health and disease. J Exp Med 217(3):e20192195. https://doi.org/10.1084/jem.20192195
Kelley N, Jeltema D, Duan Y, He Y (2019) The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 20(13). https://doi.org/10.3390/ijms20133328
Khan SR (2014) Reactive oxygen species, inflammation and calcium oxalate nephrolithiasis. Transl Androl Urol 3(3):256–276. https://doi.org/10.3978/j.issn.2223-4683.2014.06.04
Kim YG, Kim SM, Kim KP, Lee SH, Moon JY (2019) The role of inflammasome-dependent and inflammasome-independent NLRP3 in the kidney. Cells 8(11). https://doi.org/10.3390/cells8111389
Lindahl H, Olsson T (2021) Interleukin-22 influences the Th1/Th17 axis. Front Immunol 12:618110. https://doi.org/10.3389/fimmu.2021.618110
Lindemans CA, Calafiore M, Mertelsmann AM, O'Connor MH, Dudakov JA, Jenq RR, Velardi E, Young LF, Smith OM, Lawrence G, Ivanov JA, Fu YY, Takashima S, Hua G, Martin ML, O'Rourke KP, Lo YH, Mokry M, Romera-Hernandez M et al (2015) Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature 528(7583):560–564. https://doi.org/10.1038/nature16460
Liu YD, Yu SL, Wang R, Liu JN, Jin YS, Li YF, An RH (2019) Rosiglitazone suppresses calcium oxalate crystal binding and oxalate-induced oxidative stress in renal epithelial cells by promoting PPAR-γ activation and subsequent regulation of TGF-β1 and HGF expression. Oxidative Med Cell Longev 2019:4826525. https://doi.org/10.1155/2019/4826525
Liu H, Yang X, Tang K, Ye T, Duan C, Lv P, Yan L, Wu X, Chen Z, Liu J, Deng Y, Zeng G, Xing J, Ye Z, Xu H (2020a) Sulforaphane elicts dual therapeutic effects on renal inflammatory injury and crystal deposition in calcium oxalate nephrocalcinosis. Theranostics 10(16):7319–7334. https://doi.org/10.7150/thno.44054
Liu J, Yang K, Jin Y, Liu Y, Chen Y, Zhang X, Yu S, Song E, Chen S, Zhang J, Jing G, An R (2020b) H3 relaxin protects against calcium oxalate crystal-induced renal inflammatory pyroptosis. Cell Prolif 53(10):e12902. https://doi.org/10.1111/cpr.12902
Lu DH, Guo XY, Qin SY, Luo W, Huang XL, Chen M, Wang JX, Ma SJ, Yang XW, Jiang HX (2015) Interleukin-22 ameliorates liver fibrogenesis by attenuating hepatic stellate cell activation and downregulating the levels of inflammatory cytokines. World J Gastroenterol 21(5):1531–1545. https://doi.org/10.3748/wjg.v21.i5.1531
Lu MC, Zhao J, Liu YT, Liu T, Tao MM, You QD, Jiang ZY (2019) CPUY192018, a potent inhibitor of the Keap1-Nrf2 protein-protein interaction, alleviates renal inflammation in mice by restricting oxidative stress and NF-κB activation. Redox Biol 26:101266. https://doi.org/10.1016/j.redox.2019.101266
Lu CL, Teng TY, Liao MT, Ma MC (2021) TRPV1 hyperfunction contributes to renal inflammation in oxalate nephropathy. Int J Mol Sci 22(12). https://doi.org/10.3390/ijms22126204
Ludwig-Portugall I, Bartok E, Dhana E, Evers BD, Primiano MJ, Hall JP, Franklin BS, Knolle PA, Hornung V, Hartmann G, Boor P, Latz E, Kurts C (2016) An NLRP3-specific inflammasome inhibitor attenuates crystal-induced kidney fibrosis in mice. Kidney Int 90(3):525–539. https://doi.org/10.1016/j.kint.2016.03.035
Lv P, Liu H, Ye T, Yang X, Duan C, Yao X, Li B, Tang K, Chen Z, Liu J, Deng Y, Wang T, Xing J, Liang C, Xu H, Ye Z (2021) XIST inhibition attenuates calcium oxalate nephrocalcinosis-induced renal inflammation and oxidative injury via the miR-223/NLRP3 pathway. Oxidative Med Cell Longev 2021:1676152. https://doi.org/10.1155/2021/1676152
Mulay SR, Anders HJ (2016) Crystallopathies. N Engl J Med 374(25):2465–2476. https://doi.org/10.1056/NEJMra1601611
Mulay SR, Anders HJ (2017) Crystal nephropathies: mechanisms of crystal-induced kidney injury. Nat Rev Nephrol 13(4):226–240. https://doi.org/10.1038/nrneph.2017.10
Mulay SR, Kulkarni OP, Rupanagudi KV, Migliorini A, Darisipudi MN, Vilaysane A, Muruve D, Shi Y, Munro F, Liapis H, Anders HJ (2013) Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1beta secretion. J Clin Invest 123(1):236–246. https://doi.org/10.1172/JCI63679
Mulay SR, Shi C, Ma X, Anders HJ (2018) Novel insights into crystal-induced kidney injury. Kidney Dis (Basel) 4(2):49–57. https://doi.org/10.1159/000487671
Mulay SR, Honarpisheh MM, Foresto-Neto O, Shi C, Desai J, Zhao ZB, Marschner JA, Popper B, Buhl EM, Boor P, Linkermann A, Liapis H, Bilyy R, Herrmann M, Romagnani P, Belevich I, Jokitalo E, Becker JU, Anders HJ (2019) Mitochondria permeability transition versus necroptosis in oxalate-induced AKI. J Am Soc Nephrol 30(10):1857–1869. https://doi.org/10.1681/ASN.2018121218
Nikoopour E, Bellemore SM, Singh B (2015) IL-22, cell regeneration and autoimmunity. Cytokine 74(1):35–42. https://doi.org/10.1016/j.cyto.2014.09.007
Pan H, Hong F, Radaeva S, Gao B (2004) Hydrodynamic gene delivery of interleukin-22 protects the mouse liver from concanavalin A-, carbon tetrachloride-, and Fas ligand-induced injury via activation of STAT3. Cell Mol Immunol 1(1):43–49
Rutz S, Eidenschenk C, Ouyang W (2013) IL-22, not simply a Th17 cytokine. Immunol Rev 252(1):116–132. https://doi.org/10.1111/imr.12027
Sacks D, Baxter B, Campbell BCV, Carpenter JS, Cognard C, Dippel D, Eesa M, Fischer U, Hausegger K, Hirsch JA, Shazam Hussain M, Jansen O, Jayaraman MV, Khalessi AA, Kluck BW, Lavine S, Meyers PM, Ramee S, Rüfenacht DA et al (2018) Multisociety consensus quality improvement revised consensus statement for endovascular therapy of acute ischemic stroke. Int J Stroke 13(6):612–632. https://doi.org/10.1177/1747493018778713
Schulz TJ, Zarse K, Voigt A, Urban N, Birringer M, Ristow M (2007) Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab 6(4):280–293. https://doi.org/10.1016/j.cmet.2007.08.011
Sena LA, Chandel NS (2012) Physiological roles of mitochondrial reactive oxygen species. Mol Cell 48(2):158–167. https://doi.org/10.1016/j.molcel.2012.09.025
Shen Y, Miao NJ, Xu JL, Gan XX, Xu D, Zhou L, Xue H, Zhang W, Lu LM (2016) N-acetylcysteine alleviates angiotensin II-mediated renal fibrosis in mouse obstructed kidneys. Acta Pharmacol Sin 37(5):637–644. https://doi.org/10.1038/aps.2016.12
Shen Y, Jin X, Chen W, Gao C, Bian Q, Fan J, Luan J, Cao Z, Guo Z, Gu Y, Liu H, Ju D, Mei X (2020) Interleukin-22 ameliorated acetaminophen-induced kidney injury by inhibiting mitochondrial dysfunction and inflammatory responses. Appl Microbiol Biotechnol 104(13):5889–5898. https://doi.org/10.1007/s00253-020-10638-4
Sun Y, Liu Y, Guan X, Kang J, Wang X, Liu Q, Li D, Xu H, Tao Z, Deng Y (2020a) Atorvastatin inhibits renal inflammatory response induced by calcium oxalate crystals via inhibiting the activation of TLR4/NF-κB and NLRP3 inflammasome. IUBMB Life 72(5):1065–1074. https://doi.org/10.1002/iub.2250
Sun Y, Kang J, Tao Z, Wang X, Liu Q, Li D, Guan X, Xu H, Liu Y, Deng Y (2020b) Effect of endoplasmic reticulum stress-mediated excessive autophagy on apoptosis and formation of kidney stones. Life Sci 244:117232. https://doi.org/10.1016/j.lfs.2019.117232
Tang TT, Lv LL, Pan MM, Wen Y, Wang B, Li ZL, Wu M, Wang FM, Crowley SD, Liu BC (2018) Hydroxychloroquine attenuates renal ischemia/reperfusion injury by inhibiting cathepsin mediated NLRP3 inflammasome activation. Cell Death Dis 9(3):351. https://doi.org/10.1038/s41419-018-0378-3
Thamilselvan V, Menon M, Thamilselvan S (2014) Oxalate at physiological urine concentrations induces oxidative injury in renal epithelial cells: effect of alpha-tocopherol and ascorbic acid. BJU Int 114(1):140–150. https://doi.org/10.1111/bju.12642
Tsujihata M (2008) Mechanism of calcium oxalate renal stone formation and renal tubular cell injury. Int J Urol 15(2):115–120. https://doi.org/10.1111/j.1442-2042.2007.01953.x
Wang L, Hauenstein AV (2020) The NLRP3 inflammasome: mechanism of action, role in disease and therapies. Mol Asp Med 76:100889. https://doi.org/10.1016/j.mam.2020.100889
Wang S, Li Y, Fan J, Zhang X, Luan J, Bian Q, Ding T, Wang Y, Wang Z, Song P, Cui D, Mei X, Ju D (2017) Interleukin-22 ameliorated renal injury and fibrosis in diabetic nephropathy through inhibition of NLRP3 inflammasome activation. Cell Death Dis 8(7):e2937. https://doi.org/10.1038/cddis.2017.292
Wang S, Fan J, Mei X, Luan J, Li Y, Zhang X, Chen W, Wang Y, Meng G, Ju D (2019) Interleukin-22 attenuated renal tubular injury in aristolochic acid nephropathy via suppressing activation of NLRP3 inflammasome. Front Immunol 10:2277. https://doi.org/10.3389/fimmu.2019.02277
Weidenbusch M, Rodler S, Anders HJ (2015) Interleukin-22 in kidney injury and regeneration. Am J Physiol Ren Physiol 308(10):F1041–F1046. https://doi.org/10.1152/ajprenal.00005.2015
Wilson GJ, Gois PHF, Zhang A, Wang X, Law BMP, Kassianos AJ, Healy HG (2018) The role of oxidative stress and inflammation in acute oxalate nephropathy associated with ethylene glycol intoxication. Kidney Int Rep 3(5):1217–1221. https://doi.org/10.1016/j.ekir.2018.05.005
Wolk K, Witte E, Witte K, Warszawska K, Sabat R (2010) Biology of interleukin-22. Semin Immunopathol 32(1):17–31. https://doi.org/10.1007/s00281-009-0188-x
Wu Y, Min J, Ge C, Shu J, Tian D, Yuan Y, Zhou D (2020) Interleukin 22 in liver injury, inflammation and cancer. Int J Biol Sci 16(13):2405–2413. https://doi.org/10.7150/ijbs.38925
Wyatt CM, Drueke TB (2020) Stiripentol for the treatment of primary hyperoxaluria and calcium oxalate nephropathy. Kidney Int 97(1):17–19. https://doi.org/10.1016/j.kint.2019.06.011
Yang W, Hekimi S (2010) A mitochondrial superoxide signal triggers increased longevity in Caenorhabditis elegans. PLoS Biol 8(12):e1000556. https://doi.org/10.1371/journal.pbio.1000556
Zhao YW, Guo D, Li CY, Ouyang JM (2019) Comparison of the adhesion of calcium oxalate monohydrate to HK-2 cells before and after repair using tea polysaccharides. Int J Nanomedicine 14:4277–4292. https://doi.org/10.2147/ijn.S198644
Funding
This study was supported by National Natural Science Foundation of China (82073752, 81773620, 32070935, 31872746) and Shanghai Science and Technology Fund (20S11904700, 20JC1411000).
Author information
Authors and Affiliations
Contributions
HL, XM, and DJ designed the study and revised the paper. YG performed the major part of the research and drafted the manuscript. YS and WC carried out some confocal microscopy experiments. YM and HH performed the western blot assay. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All animal experiments were conducted with the approval of the Ethics Committee of Fudan University School of Pharmacy (2019-06-YL-LHR-01).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gu, Y., Shen, Y., Chen, W. et al. Protective effects of interleukin-22 on oxalate-induced crystalline renal injury via alleviating mitochondrial damage and inflammatory response. Appl Microbiol Biotechnol 106, 2637–2649 (2022). https://doi.org/10.1007/s00253-022-11876-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-11876-4