Abstract
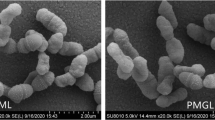
The effects of weightlessness on enteric microorganisms have been extensively studied, but have mainly been focused on pathogens. As a major component of the microbiome of the human intestinal tract, probiotics are important to keep the host healthy. Accordingly, understanding their changes under weightlessness conditions has substantial value. This study was carried out to investigate the characteristics of Lactobacillus acidophilus, a typical probiotic for humans, under simulated microgravity (SMG) conditions. The results revealed that SMG had no significant impact on the morphology of L. acidophilus, but markedly shortened its lag phase, enhanced its growth rate, acid tolerance ability up to pH < 2.5, and the bile resistance at the bile concentration of <0.05%. SMG also decreased the sensitivity of L. acidophilus to cefalexin, sulfur gentamicin, and sodium penicillin. No obvious effect of SMG was observed on the adhesion ability of L. acidophilus to Caco-2 cells. Moreover, after SMG treatment, both the culture of L. acidophilus and its liquid phase exhibited higher antibacterial activity against S. typhimurium and S. aureus in a time-dependent manner. The SMG treatment also increased the in vitro cholesterol-lowering ability of L. acidophilus by regulating the expression of the key cholesterol metabolism genes CYP7A1, ABCB11, LDLR, and HMGCR in the HepG2 cell line. Thus, the SMG treatment did have considerable influence on some biological activities and characteristics of L. acidophilus related to human health. These findings provided valuable information for understanding the influence of probiotics on human health under simulated microgravity conditions, at least.









Similar content being viewed by others
References
Bartley GE, Yokoyama W, Young SA, Anderson WHK, Hung SC, Albers DR, Langhorst ML, Kim H (2010) Hypocholesterolemic effects of hydroxypropyl methylcellulose are mediated by altered gene expression in hepatic bile and cholesterol pathways of male hamsters. J Nutr 140:1255–1260. doi:10.3945/jn.109.118349
Brown R, Klaus D, Todd P (2002) Effects of space flight, clinorotation, and centrifugation on the substrate utilization efficiency of E. coli. Microgravity Sci Tec 13:24–29
Chopra V, Fadl A, Sha J, Chopra S, Galindo C, Chopra A (2006) Alterations in the virulence potential of enteric pathogens and bacterial–host cell interactions under simulated microgravity conditions. J Toxicol Env Heal A 69:1345–1370. doi:10.1080/15287390500361792
Chou LS, Weimer B (1999) Isolation and characterization of acid and biletolerant isolates from strains of Lactobacillus acidophilus. J Dairy Sci 82:23–31. doi:10.3168/jds.S0022-0302(99)75204-5
Collado MC, Gueimonde M, Hernandez M, Sanz Y, Salminen S (2005) Adhesion of selected Bifidobacterium strains to human intestinal mucus and its role in enteropathogen exclusion. J Food Protect 68:2672–2678
Crabbé A, Schurr MJ, Monsieurs P, Morici L, Schurr J, Wilson JW, Ott CM, Tsaprailis G, Pierson DL, Stefanyshyn-Piper H (2011) Transcriptional and proteomic responses of Pseudomonas aeruginosa PAO1 to spaceflight conditions involve Hfq regulation and reveal a role for oxygen. Appl Environ Microb 77:1221–1230. doi:10.1128/AEM.01582-10
Crucian B, Stowe R, Pierson D, Sams C (2008) Immune system dysregulation following short- vs long-duration spaceflight. Aviat Space Envir Md 79:835–843. doi:10.3357/ASEM.2276.2008
Darilmaz D, Ashm B, Suludere Z, Akca G (2011) Influence of gastrointestinal system conditions on adhesion of exopolysaccharide-producing Lactobacillus delbrueckii subsp. bulgaricus strains to Caco-2 cells. Braz Arch Biol Technol 54:917–926. doi:10.1590/S1516-89132011000500009
Edsall SC, Franzodendaal TA (2014) An assessment of the long-term effects of simulated microgravity on cranial neural crest cells in zebrafish embryos with a focus on the adult skeleton. PLoS One 9:e89296. doi:10.1371/journal.pone.0089296
Fang A, Pierson D, Koenig D, Mishra S, Demain A (1997) Effect of simulated microgravity and shear stress on microcin B17 production by Escherichia coli and its excretion into the medium. Appl Environ Microb 63:4090–4092
Huang Y, Zheng Y (2010) The probiotic Lactobacillus acidophilus reduces cholesterol absorption through the down-regulation of Niemann-pick C1-like 1 in Caco-2 cells. Brit. J Nutr 103:473–478. doi:10.1017/S0007114509991991
Ilyin V (2005) Microbiological status of cosmonauts during orbital spaceflights on Salyut and Mir orbital stations. Acta Astronaut 56:839–850. doi:10.1016/j.actaastro.2005.01.009
Kacena M, Merrell G, Manfredi B, Smith E, Klaus D, Todd P (1999) Bacterial growth in space flight: logistic growth curve parameters for Escherichia coli and Bacillus subtilis. Appl Microbiol Biot 51:229–234. doi:10.1007/s002530051386
Klaus DM, Howard HN (2006) Antibiotic efficacy and microbial virulence during space flight. Trends Biotechnol 24:131–136. doi:10.1016/j.tibtech.2006.01.008
Li Q, Huyan T, Ye LJ, Li J, Shi JL, Huang QS (2014) Concentration-dependent biphasic effects of resveratrol on human natural killer cells in Vitro. J Agric Food Chem 62:10928–10935. doi:10.1021/jf502950u
Lizko N (1991) Problems of microbial ecology in man space mission. Acta Astronaut 23:163–169. doi:10.1016/0094-5765(91)90115-L
Lynch S, Brodie E, Matin A (2004) Role and regulation of sigma S in general resistance conferred by low-shear simulated microgravity in Escherichia coli. J Bacteriol 186:8207–8212. doi:10.1128/JB.186.24.8207-8212.2004
Lynch S, Mukundakrishnan K, Benoit M, Ayyaswamy P, Matin A (2006) Escherichia coli Biofilms formed under low shear modelled microgravity in ground-based system. Appl Environ Microb 72:7701–7710. doi:10.1128/AEM.01294-06
Mauclaire L, Egli M (2010) Effect of simulated microgravityon growth and production of exopolymeric substances of Micrococcus luteus space and earth isolates. FEMS Immunol Med Microbiol 59:350–356. doi:10.1111/j.1574-695X.2010.00683.x
McLean RJC, Cassanto JM, Barnes MB, Koo JH (2001) Bacterial biofilm formation under microgravity conditions. FEMS Microbiol Lett 195:115–119. doi:10.1111/j.1574-6968.2001.tb10507.x
Meier PJ, Stieger B (2002) Bile salt transporters. Annu Rev Physiol 64:635–661. doi:10.1146/annurev.physiol.64.082201.100300
Mermel LA (2013) Infection prevention and control during prolonged human space travel. Healthcare. Epidemiology 56:123–130. doi:10.1093/cid/cis861
Min BR, Pinchak WE, Merkel R, Walker S, Tomita G, Anderson RC (2008) Comparative antimicrobial activity of tannin extracts from perennial plants on mastitis pathogens. Sci Res Essays 3:66–73
Nickerson C, Ott C, Mister S, Morrow B, Burns-Keliher L, Pierson D (2000) Microgravity as a novel environmental signal affecting Salmonella enterica Serovar Typhimurium virulence. Infect Immun 68:3147–3152. doi:10.1128/IAI.68.6.3147-3152.2000
Ouwehand AC, Isolauri E, Kirjavainen PV, Salminen SJ (1999) Adhesion of four Bifidobacterium strains to human intestinal mucus from subjects in different age groups. FEMS Microbiol Lett 172:61–64. doi:10.1111/j.1574-6968.1999.tb13450.x
Rosenzweig J, Abogunde O, Thomas K, Lawal A, Nguyen Y, Sodipe A, Jejelowo O (2010) Spaceflight and modeled microgravity effects on microbial growth and virulence. App Microbiol Biot 85:885–891. doi:10.1007/s00253-009-2237-8
Rosado H, Doyle M, Hinds J, Taylor PW (2010) Low-shear modelled microgravity alters expression of virulence determinants of Staphylococcus aureus. Acta Astronaut 66:408–413. doi:10.1016/j.actaastro.2009.06.007
Shao D, Bartley GE, Yokoyama W, Pan Z, Zhang H, Zhang A (2013) Plasma and hepatic cholesterol-lowering effects of tomato pomace, tomato seed oil and defatted tomato seed in hamsters fed with high-fat diets. Food Chem 139:589–596. doi:10.1016/j.foodchem.2013.01.043
Shi F, Wang YC, Zhao TZ, Zhang S, TY D, Yang CB, Li YH, Sun XQ (2012) Effects of simulated microgravity on human umbilical vein endothelial cell angiogenesis and role of the PI3K-Akt-eNOS signal pathway. PLoS One 7:e40365. doi:10.1371/journal.pone.0040365
Soccol C, Vandenberghe L, Spier M, Medeiros A, Yamaguishi C, Lindner J (2010) The potential of probiotics: a review. Food Technol. Biotech 48:413–434
Taylor PW, Sommer AP (2005) Towards rational treatment of bacterial infections during extended space travel. Int J Antimicrob Ag 26:183–187. doi:10.1016/j.ijantimicag.2005.06.002
Vesterlund S (2009) Mechanisms of probiotics: production of antimicrobial substances. In: Lee YK, Salminen S (eds) Handbook of probiotics and prebiotics, 2nd edn. John Wiley & Sons, Inc., Hoboken
Vukanti R, Model MA, Leff LG (2012) Effect of modeled reduced gravity conditions on bacterial morphology and physiology. BMC Microbiol 12:1–11. doi:10.1186/1471-2180-12-4
Vukanti R, Mintz E, Leff L (2008) Changes in gene expression of E. coli under conditions of modeled reduced gravity. Microgravity Sci Tec 20(1):41–57. doi:10.1007/s12217-008-9012-9
Wang H, Yang YJ, Qian HY, Zhang Q, Xu H, Li JJ (2012) Resveratrol in cardiovascular disease: what is known from current research? Heart Fail Rev 17:437–448. doi:10.1007/s10741-011-9260-4
Yin S, Zhai Z, Wang G, Na H, Luo Y, Hao Y (2011) A novel vector for lactic acid bacteria that uses a bile salt hydrolase gene as a potential food-grade selection marker. J Biotechnol 152:49–53. doi:10.1016/j.jbiotec.2011.01.018
Acknowledgements
This study was funded by the project of National Natural Science Funding (No. 31471718), National Key Technology R&D Program (No. 2015BAD16B02), the Agriculture Department of China (No. CARS-30), Shaanxi Provincial Natural Science Foundation (No. 2015JQ3083), Fundamental Research Funds for the Central Universities (No. 3102016QD075 and 3102014JCQ15001), and the Foundation of He’nan Educational Committee (17B550005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Shao, D., Yao, L., riaz, M.s. et al. Simulated microgravity affects some biological characteristics of Lactobacillus acidophilus . Appl Microbiol Biotechnol 101, 3439–3449 (2017). https://doi.org/10.1007/s00253-016-8059-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-8059-6