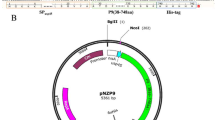
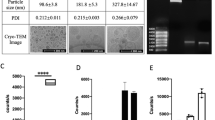
Abstract
Wild-type human interleukin-10 (hIL-10) is a non-covalent homodimer with a short half-life, thus limiting its therapeutic applications in vivo. To avoid loss of function due to dimer dissociation, we designed a synthetic hIL-10 analog by bridging both monomers via a 15 amino acid-long peptide spacer in a C-terminal to N-terminal fashion. For secretory expression in Escherichia coli, a 1156 bp fragment was generated from template vector pAZ1 by fusion PCR encoding a T7 promoter region and the signal sequence of the E. coli outer membrane protein F fused in frame to two tandem E. coli codon-optimized mature hIL-10 genes connected via a 45 nucleotide linker sequence. The construct was cloned into pUC19 for high-level expression in E. coli BL21 (DE3). The mean concentrations of hIL-10 fusion protein in the periplasm and supernatant of E. coli at 37 °C growth temperature were 130 ± 40 and 2 ± 1 ng/ml, respectively. The molecular mass of the recombinant protein was assessed via matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) analysis, indicating correct processing of the signaling sequence in E. coli. In vitro biological activity was shown by phosphorylation of signal transducer and activator of transcription protein 3 and suppression of tumor necrosis factor α secretion in lipopolysaccharide-stimulated macrophages.






Similar content being viewed by others
References
Asadullah K, Docke WD, Ebeling M, Friedrich M, Belbe G, Audring H, Volk HD, Sterry W (1999) Interleukin 10 treatment of psoriasis: clinical results of a phase 2 trial. Arch Dermatol 135(2):187–192
Berlett BS, Stadtman ER (1997) Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 272(33):20313–20316
Braat H, Rottiers P, Hommes DW, Huyghebaert N, Remaut E, Remon JP, van Deventer SJ, Neirynck S, Peppelenbosch MP, Steidler L (2006) A phase I trial with transgenic bacteria expressing interleukin-10 in Crohn’s disease. Clin Gastroenterol Hepatol 4(6):754–759
Chamekh M, Phalipon A, Quertainmont R, Salmon I, Sansonetti P, Allaoui A (2008) Delivery of biologically active anti-inflammatory cytokines IL-10 and IL-1ra in vivo by the Shigella type III secretion apparatus. J Immunol 180(6):4292–4298
Cowan SW, Schirmer T, Rummel G, Steiert M, Ghosh R, Pauptit RA, Jansonius JN, Rosenbusch JP (1992) Crystal structures explain functional properties of two E. coli porins. Nature 358(6389):727–733
D’Hauteville H, Khan S, Maskell DJ, Kussak A, Weintraub A, Mathison J, Ulevitch RJ, Wuscher N, Parsot C, Sansonetti PJ (2002) Two msbB genes encoding maximal acylation of lipid a are required for invasive Shigella flexneri to mediate inflammatory rupture and destruction of the intestinal epithelium. J Immunol 168(10):5240–5251
Domingo A, Marco R (1989) Visualization under ultraviolet light enhances 100-fold the sensitivity of peroxidase-stained blots. Anal Biochem 182(1):176–181
Dower WJ, Miller JF, Ragsdale CW (1988) High efficiency transformation of E. coli by high voltage electroporation. Nucleic Acids Res 16(13):6127–6145
El Kasmi KC, Holst J, Coffre M, Mielke L, de Pauw A, Lhocine N, Smith AM, Rutschman R, Kaushal D, Shen Y, Suda T, Donnelly RP, Myers MG Jr, Alexander W, Vignali DA, Watowich SS, Ernst M, Hilton DJ, Murray PJ (2006) General nature of the STAT3-activated anti-inflammatory response. J Immunol 177(11):7880–7888
Fedorak RN, Gangl A, Elson CO, Rutgeerts P, Schreiber S, Wild G, Hanauer SB, Kilian A, Cohard M, LeBeaut A, Feagan B (2000) Recombinant human interleukin 10 in the treatment of patients with mild to moderately active Crohn’s disease. The interleukin 10 inflammatory bowel disease cooperative study group. Gastroenterology 119(6):1473–1482
Giacalone MJ, Gentile AM, Lovitt BT, Berkley NL, Gunderson CW, Surber MW (2006) Toxic protein expression in Escherichia coli using a rhamnose-based tightly regulated and tunable promoter system. Biotechniques 40(3):355–364
Huston JS, Levinson D, Mudgett-Hunter M, Tai MS, Novotny J, Margolies MN, Ridge RJ, Bruccoleri RE, Haber E, Crea R, Oppermann H (1988) Protein engineering of antibody binding sites: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli. Proc Natl Acad Sci U S A 85(16):5879–5883
Joly JC, Swartz JR (1994) Protein folding activities of Escherichia coli protein disulfide isomerase. Biochemistry 33(14):4231–4236
Josephson K, DiGiacomo R, Indelicato SR, Iyo AH, Nagabhushan TL, Parker MH, Walter MR (2000) Design and analysis of an engineered human interleukin-10 monomer. J Biol Chem 275(32):25054
Kang D, Gho YS, Suh M, Kang C (2002) Highly sensitive and fast protein detection with Coomassie brilliant blue in sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Bull Kor Chem Soc 23(11):1511–1512
Luque-Garcia JL, Zhou G, Spellman DS, Sun TT, Neubert TA (2008) Analysis of electroblotted proteins by mass spectrometry: protein identification after western blotting. Mol Cell Proteomics 7(2):308–314
Miller JH (1992) A short course in bacterial genetics: a laboratory manual and handbook for Escherichia coli and related bacteria. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Milligan ED, Langer SJ, Sloane EM, He L, Wieseler-Frank J, O’Connor K, Martin D, Forsayeth JR, Maier SF, Johnson K, Chavez RA, Leinwand LA, Watkins LR (2005) Controlling pathological pain by adenovirally driven spinal production of the anti-inflammatory cytokine, interleukin-10. Eur J Neurosci 21(8):2136–2148
Moore KW, de Waal MR, Coffman RL, O’Garra A (2001) Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol 19:683–765
Nagahari K, Kanaya S, Munakata K, Aoyagi Y, Mizushima S (1985) Secretion into the culture medium of a foreign gene product from Escherichia coli: use of the ompF gene for secretion of human beta-endorphin. EMBO J 4(13 A):3589–3592
Neu HC, Heppel LA (1965) The release of enzymes from Escherichia coli by osmotic shock and during the formation of spheroplasts. J Biol Chem 240(9):3685–3692
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8(10):785–786
Pöhlmann C, Brandt M, Mottok DS, Zschüttig A, Campbell JW, Blattner FR, Frisch D, Gunzer F (2012) Periplasmic delivery of biologically active human interleukin-10 in Escherichia coli via a sec-dependent signal peptide. J Mol Microbiol Biotechnol 22(1):1–9
Pöhlmann C, Thomas M, Förster S, Brandt M, Hartmann M, Bleich A, Gunzer F (2013) Improving health from the inside: use of engineered intestinal microorganisms as in situ cytokine delivery system. Bioengineered 4(3):172–179
Posfai G, Plunkett G 3rd, Feher T, Frisch D, Keil GM, Umenhoffer K, Kolisnychenko V, Stahl B, Sharma SS, de Arruda M, Burland V, Harcum SW, Blattner FR (2006) Emergent properties of reduced-genome Escherichia coli. Science 312(5776):1044–1046
Rachmawati H, Beljaars L, Reker-Smit C, Bakker HI, Van Loenen-Weemaes AM, Lub-De Hooge MN, Poelstra K (2011) Intravenous administration of recombinant human IL-10 suppresses the development of anti-thy 1-induced glomerulosclerosis in rats. PDA J Pharm Sci Technol 65(2):116–130
Rockenbach SK, Dupuis MJ, Pitts TW, Marschke CK, Tomich CS (1991) Secretion of active truncated CD4 into Escherichia coli periplasm. Appl Microbiol Biotechnol 35(1):32–37
Roy A, Kucukural A, Zhang Y (2010) I-TASSER: a unified platform for automated protein structure and function prediction. Nat Protoc 5(4):725–738
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A 74(12):5463–5467
Schreiber S, Fedorak RN, Nielsen OH, Wild G, Williams CN, Nikolaus S, Jacyna M, Lashner BA, Gangl A, Rutgeerts P, Isaacs K, van Deventer SJ, Koningsberger JC, Cohard M, LeBeaut A, Hanauer SB (2000) Safety and efficacy of recombinant human interleukin 10 in chronic active Crohn’s disease. Crohn’s disease IL-10 cooperative study group. Gastroenterology 119(6):1461–1472
Signor L, Boeri Erba E (2013) Matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometric analysis of intact proteins larger than 100 kDa. J Vis Exp (79)
Soderquist RG, Milligan ED, Harrison JA, Chavez RA, Johnson KW, Watkins LR, Mahoney MJ (2010) PEGylation of interleukin-10 for the mitigation of enhanced pain states. J Biomed Mater Res A 93(3):1169–1179
Strohalm M, Kavan D, Novák P, Volný M, Havlícek V (2010) mMass 3: a cross-platform software environment for precise analysis of mass spectrometric data. Anal Chem 82(11):4648–4651
Studier FW, Moffatt BA (1986) Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol 189(1):113–130
Sun G, Anderson VE (2004) Prevention of artifactual protein oxidation generated during sodium dodecyl sulfate-gel electrophoresis. Electrophoresis 25(7–8):959–965
Syto R, Murgolo NJ, Braswell EH, Mui P, Huang E, Windsor WT (1998) Structural and biological stability of the human interleukin 10 homodimer. Biochemistry 37(48):16943–16951
Tan JC, Indelicato SR, Narula SK, Zavodny PJ, Chou CC (1993) Characterization of interleukin-10 receptors on human and mouse cells. J Biol Chem 268(28):21053–21059
van Deventer SJ, Elson CO, Fedorak RN (1997) Multiple doses of intravenous interleukin 10 in steroid-refractory Crohn’s disease. Crohn’s disease study group. Gastroenterology 113(2):383–389
Varma S, Chiu SW, Jakobsson E (2006) The influence of amino acid protonation states on molecular dynamics simulations of the bacterial porin OmpF. Biophys J 90(1):112–123
Vieira P, de Waal-Malefyt R, Dang MN, Johnson KE, Kastelein R, Fiorentino DF, deVries JE, Roncarolo MG, Mosmann TR, Moore KW (1991) Isolation and expression of human cytokine synthesis inhibitory factor cDNA clones: homology to Epstein-Barr virus open reading frame BCRFI. Proc Natl Acad Sci U S A 88(4):1172–1176
Walter MR, Nagabhushan TL (1995) Crystal structure of interleukin 10 reveals an interferon gamma-like fold. Biochemistry 34(38):12118–12125
Wenzel T, Sparbier K, Mieruch T, Kostrzewa M (2006) 2,5-Dihydroxyacetophenone: a matrix for highly sensitive matrix-assisted laser desorption/ionization time-of-flight mass spectrometric analysis of proteins using manual and automated preparation techniques. Rapid Commun Mass Spectrom 20(5):785–789
Windsor WT, Syto R, Tsarbopoulos A, Zhang R, Durkin J, Baldwin S, Paliwal S, Mui PW, Pramanik B, Trotta PP, Tindall SH (1993) Disulfide bond assignments and secondary structure analysis of human and murine interleukin 10. Biochemistry 32(34):8807–8815
Woodcock DM, Crowther PJ, Doherty J, Jefferson S, DeCruz E, Noyer-Weidner M, Smith SS, Michael MZ, Graham MW (1989) Quantitative evaluation of Escherichia coli host strains for tolerance to cytosine methylation in plasmid and phage recombinants. Nucleic Acids Res 17(9):3469–3478
Wülfing C, Plückthun A (1994) Correctly folded T-cell receptor fragments in the periplasm of Escherichia coli. Influence of folding catalysts. J Mol Biol 242(5):655–669
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33(1):103–119
Yoon SI, Logsdon NJ, Sheikh F, Donnelly RP, Walter MR (2006) Conformational changes mediate interleukin-10 receptor 2 (IL-10R2) binding to IL-10 and assembly of the signaling complex. J Biol Chem 281(46):35088–35096
Zdanov A, Schalk-Hihi C, Gustchina A, Tsang M, Weatherbee J, Wlodawer A (1995) Crystal structure of interleukin-10 reveals the functional dimer with an unexpected topological similarity to interferon gamma. Structure 3(6):591–601
Zdanov A, Schalk-Hihi C, Wlodawer A (1996) Crystal structure of human interleukin-10 at 1.6 A resolution and a model of a complex with its soluble receptor. Protein Sci 5(10):1955–1962
Zhang Y (2008) I-TASSER server for protein 3D structure prediction. BMC Bioinf 9:40
Acknowledgments
This work was funded by a grant of the “MeDDrive38” program of the TU Dresden, Medical Faculty, Carl Gustav Carus to C.P. and by a grant of the Federal Ministry of Education and Research (BMBF Genomik-Transfer, PROTumor consortium, project number 0315590A) to F.G. We would like to thank Manuela Brandt for excellent technical assistance and Christian Auerbach and Claudia Dittrich for help with database submission of plasmid sequences and bacterial strains, and we would also like to acknowledge Dr. Michael Hogardt (Max von Pettenkofer Institute, Munich, Germany) for providing the murine macrophage cell line J-774.1.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The study neither comprised the use of human material and data nor did the research involve animals. Thus, an approval by the ethics committee was not necessary for the study.
Conflict of interest
All authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Guggenbichler, F., Büttner, C., Rudolph, W. et al. Design of a covalently linked human interleukin-10 fusion protein and its secretory expression in Escherichia coli . Appl Microbiol Biotechnol 100, 10479–10493 (2016). https://doi.org/10.1007/s00253-016-7667-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7667-5