Abstract
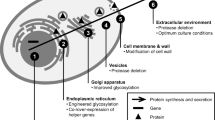
Mammalian peptidoglycan recognition proteins (PGLYRPs) are highly conserved pattern-recognition molecules of the innate immune system with considerable bactericidal activity, which manifest their potential values for the application to food and pharmaceutical industry. However, the effective expression of porcine PGLYRP-1 in Pichia pastoris has not been reported so far. In this study, expression in P. pastoris was explored as an efficient way to produce functional porcine PGLYRP-1. Cooperation of chaperones co-expression and gene dosage (including protein disulfide isomerase (PDI)/binding protein (BiP) and pglyrp-1) were used to enhance functional expression of antimicrobial protein in P. pastoris. Overexpression of PDI was certainly able to increase secretion level of PGLYRP-1 protein because the increase in secreted PGLYRP-1 secretion was correlated with the copy numbers of PDI in high copy pglyrp-1 clones. However, co-expression of BiP was proved to be detrimental to PGLYRP-1 secretion. In addition, we also found that excessive expression of PDI and/or BiP could decrease the mRNA expression of pglyrp-1 gene. This showed that PDI and BiP as the target genes of unfolded protein response (UPR) might regulate the transcription of the target protein. These data demonstrated for the first time that the combination of chaperones and gene dosages could improve the yield of PGLYRP-1, which could facilitate the application to food and pharmaceutical industry.






Similar content being viewed by others
References
Boman HG (1995) Peptide antibiotics and their role in innate immunity. Annu Rev Immunol 13:61–92. doi:10.1146/annurev.immunol.13.1.61
Boman HG (2003) Antibacterial peptides: basic facts and emerging concepts. J Intern Med 254(3):197–215. doi:10.1046/j.1365-2796.2003.01228.x
Chen L, Wang F, Wang X, Liu YG (2013) Robust one-tube Omega-PCR strategy accelerates precise sequence modification of plasmids for functional genomics. Plant Cell Physiol 54(4):634–642. doi:10.1093/pcp/pct009
Cho S, Wang Q, Swaminathan CP, Hesek D, Lee M, Boons GJ, Mobashery S, Mariuzza RA (2007) Structural insights into the bactericidal mechanism of human peptidoglycan recognition proteins. Proc Natl Acad Sci U S A 104(21):8761–8766. doi:10.1073/pnas.0701453104
Cregg JM, Cereghino JL, Shi J, Higgins DR (2000) Recombinant protein expression in Pichia pastoris. Mol Biotechnol 16(1):23–52. doi:10.1385/mb:16:1:23
Dziarski R, Gupta D (2006) The peptidoglycan recognition proteins (PGRPs). Genome Biol 7(8):232. doi:10.1186/gb-2006-7-8-232
Esposito D, Chatterjee DK (2006) Enhancement of soluble protein expression through the use of fusion tags. Curr Opin Biotechnol 17(4):353–358. doi:10.1016/j.copbio.2006.06.003
Fomenko DE, Gladyshev VN (2003) Genomics perspective on disulfide bond formation. Antioxid Redox Signal 5(4):397–402. doi:10.1089/152308603768295131
Gasser B, Saloheimo M, Rinas U, Dragosits M, Rodriguez-Carmona E, Baumann K, Giuliani M, Parrilli E, Branduardi P, Lang C, Porro D, Ferrer P, Tutino ML, Mattanovich D, Villaverde A (2008) Protein folding and conformational stress in microbial cells producing recombinant proteins: a host comparative overview. Microb Cell Factories 7:11. doi:10.1186/1475-2859-7-11
Gross E, Kastner DB, Kaiser CA, Fass D (2004) Structure of Ero1p, source of disulfide bonds for oxidative protein folding in the cell. Cell 117(5):601–610. doi:10.1016/s0092-8674(04)00418-0
Gustafsson C, Govindarajan S, Minshull J (2004) Codon bias and heterologous protein expression. Trends Biotechnol 22(7):346–353. doi:10.1016/j.tibtech.2004.04.006
Hartner FS, Ruth C, Langenegger D, Johnson SN, Hyka P, Lin-Cereghino GP, Lin-Cereghino J, Kovar K, Cregg JM, Glieder A (2008) Promoter library designed for fine-tuned gene expression in Pichia pastoris. Nucleic Acids Res 36(12) doi:10.1093/nar/gkn369
Hohenblum H, Gasser B, Maurer M, Borth N, Mattanovich D (2004) Effects of gene dosage, promoters, and substrates on unfolded protein stress of recombinant Pichia pastoris. Biotechnol Bioeng 85(4):367–375. doi:10.1002/bit.10904
Idiris A, Tohda H, Kumagai H, Takegawa K (2010) Engineering of protein secretion in yeast: strategies and impact on protein production. Appl Microbiol Biotechnol 86(2):403–417. doi:10.1007/s00253-010-2447-0
Inan M, Aryasomayajula D, Sinha J, Meagher MM (2006) Enhancement of protein secretion in Pichia pastoris by overexpression of protein disulfide isomerase. Biotechnol Bioeng 93(4):771–778. doi:10.1002/bit.20762
Janeway CA, Medzhitov R (2002) Innate immune recognition. Annu Rev Immunol 20:197–216. doi:10.1146/annurev.immunol.20.083001.084359
Kang D, Lu G, Lundstrom A, Gelius E, Steiner H (1998) A peptidoglycan recognition protein in innate immunity conserved from insects to humans. Proc Natl Acad Sci U S A 95(17):10078–10082. doi:10.1073/pnas.95.17.10078
Kappeler SR, Heuberger C, Farah Z, Puhan Z (2004) Expression of the peptidoglycan recognition protein, PGRP, in the lactating mammary gland. J Dairy Sci 87(8):2660–2668
Kashyap DR, Rompca A, Gaballa A, Helmann JD, Chan J, Chang CJ, Hozo I, Gupta D, Dziarski R (2014) Peptidoglycan recognition proteins kill bacteria by inducing oxidative, thiol, and metal stress. PLoS Pathog 10(7):e1004280. doi:10.1371/journal.ppat.1004280
Kashyap DR, Wang M, Liu L-H, Boons G-J, Gupta D, Dziarski R (2011) Peptidoglycan recognition proteins kill bacteria by activating protein-sensing two-component systems. Nat Med 17(6):676–683. doi:10.1038/nm.2357
Lehrer RI, Rosenman M, Harwig SS, Jackson R, Eisenhauer P (1991) Ultrasensitive assays for endogenous antimicrobial polypeptides. J Immunol Methods 137(2):167–173. doi:10.1016/0022-1759(91)90021-7
Liang S, Lin Y, Li C, Ye Y (2012) Internal ribosome entry site mediates protein synthesis in yeast Pichia pastoris. Biotechnol Lett 34(5):957–964. doi:10.1007/s10529-012-0862-2
Liu H, Qin Y, Huang Y, Chen Y, Cong P, He Z (2014a) Direct evaluation of the effect of gene dosage on secretion of protein from yeast Pichia pastoris by expressing EGFP. J Microbiol Biotechnol 24(2):144–151
Liu X, Deng R, Wang J, Wang X (2014b) COStar: a D-star Lite-based dynamic search algorithm for codon optimization. J Theor Biol 344:19–30. doi:10.1016/j.jtbi.2013.11.022
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25(4):402–408. doi:10.1006/meth.2001.1262
Lo D, Tynan W, Dickerson J, Mendy J, Chang H-W, Scharf M, Byrne D, Brayden D, Higgins L, Evans C, O’Mahony DJ (2003) Peptidoglycan recognition protein expression in mouse Peyer’s Patch follicle associated epithelium suggests functional specialization. Cell Immunol 224(1):8–16. doi:10.1016/s0008-8749(03)00155-2
Lodi T, Neglia B, Donnini C (2005) Secretion of human serum albumin by Kluyveromyces lactis overexpressing KlPDI1 and KlERO1. Appl Environ Microbiol 71(8):4359–4363. doi:10.1128/AEM.71.8.4359-4363.2005
Loewen MC, Liu X, Davies PL, Daugulis AJ (1997) Biosynthetic production of type II fish antifreeze protein: Fermentation by Pichia pastoris. Appl Microbiol Biotechnol 48(4):480–486
Lu X, Wang M, Qi J, Wang H, Li X, Gupta D, Dziarski R (2006) Peptidoglycan recognition proteins are a new class of human bactericidal proteins. J Biophys Chem 281(9):5895–5907. doi:10.1074/jbc.M511631200
Macauley-Patrick S, Fazenda ML, McNeil B, Harvey LM (2005) Heterologous protein production using the Pichia pastoris expression system. Yeast 22(4):249–270. doi:10.1002/yea.1208
Marx H, Mecklenbrauker A, Gasser B, Sauer M, Mattanovich D (2009) Directed gene copy number amplification in Pichia pastoris by vector integration into the ribosomal DNA locus. FEMS Yeast Res 9(8):1260–1270. doi:10.1111/j.1567-1364.2009.00561.x
Montano AM, Tsujino F, Takahata N, Satta Y (2011) Evolutionary origin of peptidoglycan recognition proteins in vertebrate innate immune system. BMC Evol Biol 11:79. doi:10.1186/1471-2148-11-79
Osanai A, Sashinami H, Asano K, Li SJ, Hu DL, Nakane A (2011) Mouse peptidoglycan recognition protein PGLYRP-1 plays a role in the host innate immune response against Listeria monocytogenes infection. Infect Immun 79(2):858–866. doi:10.1128/IAI.00466-10
Ruottinen M, Bollok M, Kogler M, Neubauer A, Krause M, Hamalainen ER, Myllyharju J, Vasala A, Neubauer P (2008) Improved production of human type II procollagen in the yeast Pichia pastoris in shake flasks by a wireless-controlled fed-batch system. BMC Biotechnol 8:33. doi:10.1186/1472-6750-8-33
Sharma P, Dube D, Singh A, Mishra B, Singh N, Sinha M, Dey S, Kaur P, Mitra DK, Sharma S, Singh TP (2011) Structural basis of recognition of pathogen-associated molecular patterns and inhibition of proinflammatory cytokines by camel peptidoglycan recognition protein. J Biophys Chem 286(18):16208–16217. doi:10.1074/jbc.M111.228163
Sharma P, Dube D, Yadav SP, Sinha M, Kaur P, Sharma S, Singh TP (2014) Structural and functional studies of mammalian peptidoglycan recognition protein, PGRP-S. J Indian Inst Sci 94(1):109–117
Sharma P, Yamini S, Dube D, Singh A, Sinha M, Dey S, Mitra DK, Kaur P, Sharma S, Singh TP (2012) Structural studies on molecular interactions between camel peptidoglycan recognition protein, CPGRP-S, and peptidoglycan moieties N-acetylglucosamine and N-acetylmuramic acid. J Biophys Chem 287(26):22153–22164. doi:10.1074/jbc.M111.321307
Shen Q, Wu M, Wang HB, Naranmandura H, Chen SQ (2012) The effect of gene copy number and co-expression of chaperone on production of albumin fusion proteins in Pichia pastoris. Appl Microbiol Biotechnol 96(3):763–772. doi:10.1007/s00253-012-4337-0
Smith JD, Tang BC, Robinson AS (2004) Protein disulfide isomerase, but not binding protein, overexpression enhances secretion of a non-disulfide-bonded protein in yeast. Biotechnol Bioeng 85(3):340–350. doi:10.1002/bit.10853
Tydell CC, Yount N, Tran D, Yuan J, Selsted ME (2002) Isolation, characterization, and antimicrobial properties of bovine oligosaccharide-binding protein. A microbicidal granule protein of eosinophils and neutrophils. J Biophys Chem 277(22):19658–19664. doi:10.1074/jbc.M200659200
Tydell CC, Yuan J, Tran P, Selsted ME (2006) Bovine peptidoglycan recognition protein-S: antimicrobial activity, localization, secretion, and binding properties. J Immunol 176(2):1154–1162
Ueda W, Tohno M, Shimazu T, Fujie H, Aso H, Kawai Y, Numasaki M, Saito T, Kitazawa H (2011) Molecular cloning, tissue expression, and subcellular localization of porcine peptidoglycan recognition proteins 3 and 4. Vet Immunol Immunopathol 143(1–2):148–154. doi:10.1016/j.vetimm.2011.05.026
Vassileva A, Chugh DA, Swaminathan S, Khanna N (2001) Effect of copy number on the expression levels of hepatitis B surface antigen in the methylotrophic yeast Pichia pastoris. Protein Expr Purif 21(1):71–80. doi:10.1006/prep.2000.1335
Wilkinson B, Gilbert HF (2004) Protein disulfide isomerase. Biochim Biophys Acta 1699(1–2):35–44. doi:10.1016/j.bbapap.2004.02.017
Yoshida H (2007) ER stress and diseases. FEBS J 274(3):630–658. doi:10.1111/j.1742-4658.2007.05639.x
Zhu T, Guo M, Tang Z, Zhang M, Zhuang Y, Chu J, Zhang S (2009) Efficient generation of multi-copy strains for optimizing secretory expression of porcine insulin precursor in yeast Pichia pastoris. J Appl Microbiol 107(3):954–963. doi:10.1111/j.1365-2672.2009.04279.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Natural Science Foundation of Guangdong Province (No: 2015 A030312005, 2014 A030313449).
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic Supplementary Material
ESM 1
(PDF 438kb)
Rights and permissions
About this article
Cite this article
Yang, J., Lu, Z., Chen, J. et al. Effect of cooperation of chaperones and gene dosage on the expression of porcine PGLYRP-1 in Pichia pastoris . Appl Microbiol Biotechnol 100, 5453–5465 (2016). https://doi.org/10.1007/s00253-016-7372-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7372-4