Abstract
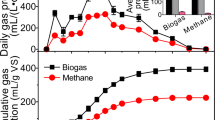
Two-phasic anaerobic digestion processes (hydrolysis/acidogenesis separated from acetogenesis/methanogenesis) can be used for biogas production on demand or a combined chemicals/bioenergy production. For an effective process control, detailed knowledge about the microbial catalysts and their correlation to process conditions is crucial. In this study, maize silage was digested in a two-phase process and interrelationships between process parameters and microbial communities were revealed. In the first-phase reactor, alternating metabolic periods were observed which emerged independently from the feeding frequency. During the L-period, up to 11.8 g L-1 lactic acid was produced which significantly correlated to lactic acid bacteria of the genus Lactobacillus as the most abundant community members. During the alternating G-period, the production of volatile fatty acids (up to 5.3, 4.0 and 3.1 g L−1 for propionic, n-butyric and n-caproic acid, respectively) dominated accompanied by a high gas production containing up to 28 % hydrogen. The relative abundance of various Clostridiales increased during this metabolic period. In the second-phase reactor, the metabolic fluctuations of the first phase were smoothed out resulting in a stable biogas production as well as stable bacterial and methanogenic communities. However, the biogas composition followed the metabolic dynamics of the first phase: the hydrogen content increased during the L-period whereas highest CH4/CO2 ratios (up to 2.8) were reached during the G-period. Aceticlastic Methanosaeta as well as hydrogenotrophic Methanoculleus and Methanobacteriaceae were identified as dominant methanogens. Consequently, a directed control of the first-phase stabilizing desired metabolic states can lead to an enhanced productivity regarding chemicals and bioenergy.






Similar content being viewed by others
References
Agler MT, Wrenn BA, Zinder SH, Angenent LT (2011) Waste to bioproduct conversion with undefined mixed cultures: the carboxylate platform. Trends Biotechnol 29(2):70–78. doi:10.1016/j.tibtech.2010.11.006
Allison SD, Martiny JB (2008) Colloquium paper: resistance, resilience, and redundancy in microbial communities. Proc Natl Acad Sci U S A 105(Suppl 1):11512–11519. doi:10.1073/pnas.0801925105
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336. doi:10.1038/nmeth.f.303
Chanakya HN, Borgaonkar S, Meena G, Jagadish KS (1993) Solid-phase biogas production with garbage or water hyacinth. Bioresour Technol 46(3):227–231
Chang H, Kim N-J, Kang J, Jeong C (2010) Biomass-derived volatile fatty acid platform for fuels and chemicals. Biotechnol Bioproc E 15(1):1–10. doi:10.1007/s12257-009-3070-8
Chojnacka A, Blaszczyk MK, Szczesny P, Nowak K, Suminska M, Tomczyk-Zak K, Zielenkiewicz U, Sikora A (2011) Comparative analysis of hydrogen-producing bacterial biofilms and granular sludge formed in continuous cultures of fermentative bacteria. Bioresour Technol 102(21):10057–10064. doi:10.1016/j.biortech.2011.08.063
Cirne DG, Lehtomaki A, Bjornsson L, Blackall LL (2007) Hydrolysis and microbial community analyses in two-stage anaerobic digestion of energy crops. J Appl Microbiol 103(3):516–527. doi:10.1111/j.1365-2672.2006.03270.x
Cysneiros D, Banks CJ, Heaven S (2008) Anaerobic digestion of maize in coupled leach-bed and anaerobic filter reactors. Water Sci Technol 58(7):1505–1511. doi:10.2166/wst.2008.518
Demirel B, Yenigün O (2002) Two-phase anaerobic digestion processes: a review. J Chem Technol Biot 77(7):743–755. doi:10.1002/Jctb.630
Demirer GN, Chen S (2005) Two-phase anaerobic digestion of unscreened dairy manure. Process Biochem 40(11):3542–3549. doi:10.1016/j.procbio.2005.03.062
Desvaux M (2006) Unravelling carbon metabolism in anaerobic cellulolytic bacteria. Biotechnol Prog 22(5):1229–1238. doi:10.1021/bp060016e
Ding S-Y, Himmel ME (2006) The maize primary cell wall microfibril: a new model derived from direct visualization. J Agr Food Chem 54(3):597–606. doi:10.1021/jf051851z
Dodsworth JA, Blainey PC, Murugapiran SK, Swingley WD, Ross CA, Tringe SG, Chain PS, Scholz MB, Lo C-C, Raymond J (2013) Single-cell and metagenomic analyses indicate a fermentative and saccharolytic lifestyle for members of the OP9 lineage. Nat Commun 4:1854. doi:10.1038/ncomms2884
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26(19):2460–2461. doi:10.1093/bioinformatics/btq461
Fennell DE, Gossett JM, Zinder SH (1997) Comparison of butyric acid, ethanol, lactic acid, and propionic acid as hydrogen donors for the reductive dechlorination of tetrachloroethene. Environ Sci Technol 31(3):918–926. doi:10.1021/Es960756r
Gies EA, Konwar KM, Beatty JT, Hallam SJ (2014) Illuminating microbial dark matter in meromictic Sakinaw Lake. Appl Environ Microbiol 80(21):6807–6818. doi:10.1128/AEM.01774-14
Grootscholten TI, Steinbusch KJ, Hamelers HV, Buisman CJ (2013) Chain elongation of acetate and ethanol in an upflow anaerobic filter for high rate MCFA production. Bioresour Technol 135:440–445. doi:10.1016/j.biortech.2012.10.165
Hawkes FR, Dinsdale R, Hawkes DL, Hussy I (2002) Sustainable fermentative hydrogen production: challenges for process optimisation. Int J Hydrog Energ 27(11–12):1339–1347. doi:10.1016/S0360-3199(02)00090-3
Heckel M (2007) Einfluß von Siliermitteln auf Siliererfolg und Biogasproduktion verschiedener Energiepflanzen. University of Potsdam, Diploma thesis
Hofvendahl K, Hahn-Hägerdal B (2000) Factors affecting the fermentative lactic acid production from renewable resources. Enzym Microb Tech 26(2–4):87–107. doi:10.1016/S0141-0229(99)00155-6
Horiuchi J-I, Shimizu T, Tada K, Kanno T, Kobayashi M (2002) Selective production of organic acids in anaerobic acid reactor by pH control. Bioresour Technol 82(3):209–213. doi:10.1016/S0960-8524(01)00195-X
Hussy I, Hawkes FR, Dinsdale R, Hawkes DL (2003) Continuous fermentative hydrogen production from a wheat starch co-product by mixed microflora. Biotechnol Bioeng 84(6):619–626. doi:10.1002/bit.10785
Jo JH, Jeon CO, Lee DS, Park JM (2007) Process stability and microbial community structure in anaerobic hydrogen-producing microflora from food waste containing kimchi. J Biotechnol 131(3):300–308. doi:10.1016/j.jbiotec.2007.07.492
Kusch S, Oechsner H, Jungbluth T (2012) Effect of various leachate recirculation strategies on batch anaerobic digestion of solid substrates. IJEWM 9(1/2):69–88. doi:10.1504/IJEWM.2012.044161
Lee DJ, Show KY, Su A (2011) Dark fermentation on biohydrogen production: pure culture. Bioresour Technol 102(18):8393–8402. doi:10.1016/j.biortech.2011.03.041
Lehtomäki A, Huttunen S, Lehtinen TM, Rintala JA (2008) Anaerobic digestion of grass silage in batch leach bed processes for methane production. Bioresour Technol 99(8):3267–3278. doi:10.1016/j.biortech.2007.04.072
Lucas R, Kuchenbuch A, Fetzer I, Harms H, Kleinsteuber S (2015) Long-term monitoring reveals stable and remarkably similar microbial communities in parallel full-scale biogas reactors digesting energy crops. FEMS Microbiol Ecol 91(3). doi:10.1093/femsec/fiv004
Lv W, Schanbacher FL, Yu Z (2010) Putting microbes to work in sequence: recent advances in temperature-phased anaerobic digestion processes. Bioresour Technol 101(24):9409–9414. doi:10.1016/j.biortech.2010.07.100
Lykidis A, Chen CL, Tringe SG, McHardy AC, Copeland A, Kyrpides NC, Hugenholtz P, Macarie H, Olmos A, Monroy O, Liu WT (2011) Multiple syntrophic interactions in a terephthalate-degrading methanogenic consortium. ISME J 5(1):122–130. doi:10.1038/ismej.2010.125
Mähnert P (2007) Kinetik der Biogasproduktion aus nachwachsenden Rohstoffen und Gülle. Humboldt University Berlin, Dissertation
McCarty PL, Smith DP (1986) Anaerobic wastewater treatment. Environ Sci Technol 20(12):1200–1206. doi:10.1021/es00154a002
McDonald D, Price MN, Goodrich J, Nawrocki EP, DeSantis TZ, Probst A, Andersen GL, Knight R, Hugenholtz P (2012) An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J 6(3):610–618. doi:10.1038/ismej.2011.139
Naumann C, Bassler R (2006) Handbuch der landwirtschaftlichen Versuchs- und Untersuchungsmethodik (VDLUFA-Methodenbuch), vol Bd. III Die chemische Untersuchung von Futtermitteln, VDLUFA-Verlag, Darmstadt, Germany
Nizami A-S, Korres NE, Murphy JD (2009) Review of the integrated process for the production of grass biomethane. Environ Sci Technol 43(22):8496–8508. doi:10.1021/es901533j
Noike T, Ko I, Yokoyama S, Kohno Y, Li Y (2005) Continuous hydrogen production from organic waste. Water Sci Technol 52(1–2):145–151
Noike T, Takabatake H, Mizuno O, Ohba M (2002) Inhibition of hydrogen fermentation of organic wastes by lactic acid bacteria. Int J Hydrog Energ 27(11–12):1367–1371. doi:10.1016/S0360-3199(02)00120-9
Oksanen J (2011) Multivariate analysis of ecological communities in R: vegan tutorial. 2.0-6 edn
Parawira W, Murto M, Read JS, Mattiasson B (2004) Volatile fatty acid production during anaerobic mesophilic digestion of solid potato waste. J Chem Technol Biot 79(7):673–677. doi:10.1002/jctb.1012
Pelletier E, Kreimeyer A, Bocs S, Rouy Z, Gyapay G, Chouari R, Riviere D, Ganesan A, Daegelen P, Sghir A, Cohen GN, Medigue C, Weissenbach J, Le Paslier D (2008) “Candidatus Cloacamonas acidaminovorans”: genome sequence reconstruction provides a first glimpse of a new bacterial division. J Bacteriol 190(7):2572–2579. doi:10.1128/JB.01248-07
Pohland FG, Ghosh S (1971) Developments in anaerobic stabilization of organic wastes—the two-phase concept. Environ Lett 1(4):255–266. doi:10.1080/00139307109434990
Popp D, Schrader S, Kleinsteuber S, Harms H, Sträuber H (2015) Biogas production from coumarin-rich plants—impact of coumarin on process parameters and microbial community. FEMS Microbiol Ecol 91(9). doi:10.1093/femsec/fiv103
R Development Core Team (2011) R: a language and environment for statistical computing. 0.97.551 edn. R Foundation for Statistical Computing, Vienna, Austria
Ramasamy EV, Abbasi SA (2000) High-solids anaerobic digestion for the recovery of energy from municipal solid waste (MSW). Environ Technol 21(3):345–349. doi:10.1080/09593332108618120
Seedorf H, Fricke WF, Veith B, Brüggemann H, Liesegang H, Strittmatter A, Miethke M, Buckel W, Hinderberger J, Li F, Hagemeier C, Thauer RK, Gottschalk G (2008) The genome of Clostridium kluyveri, a strict anaerobe with unique metabolic features. Proc Natl Acad Sci U S A 105(6):2128–2133. doi:10.1073/pnas.0711093105
Sikora A, Zielenkiewicz U, Błaszczyk M, Jurkowski M (2013) Lactic acid bacteria in hydrogen-producing consortia: on purpose or by coincidence? In: Kongo JM (ed) Lactic acid bacteria—R & D for food, health and livestock purposes. InTech. doi:10.5772/50364
Spirito CM, Richter H, Rabaey K, Stams AJ, Angenent LT (2014) Chain elongation in anaerobic reactor microbiomes to recover resources from waste. Curr Opin Biotechnol 27C:115–122. doi:10.1016/j.copbio.2014.01.003
Staley BF, Reyes FL D l, Barlaz MA (2011) Effect of spatial differences in microbial activity, pH, and substrate levels on methanogenesis initiation in refuse. Appl Environ Microbiol 77(7):2381–2391. doi:10.1128/aem.02349-10
Steinberg LM, Regan JM (2008) Phylogenetic comparison of the methanogenic communities from an acidic, oligotrophic fen and an anaerobic digester treating municipal wastewater sludge. Appl Environ Microbiol 74(21):6663–6671. doi:10.1128/AEM.00553-08
Sträuber H, Bühligen F, Kleinsteuber S, Nikolausz M, Porsch K (2015) Improved anaerobic fermentation of wheat straw by alkaline pre-treatment and addition of alkali-tolerant microorganisms. Bioengineering 2(2):66–93. doi:10.3390/bioengineering2020066
Sträuber H, Schröder M, Kleinsteuber S (2012) Metabolic and microbial community dynamics during the hydrolytic and acidogenic fermentation in a leach-bed process. Energ Sustain Soc 2(1):13. doi:10.1186/2192-0567-2-13
Wei T (2011) corrplot: visualization of a correlation matrix. http://CRAN.R-project.org/package=corrplot.
Van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74(10):3583–3597. doi:10.3168/jds.S0022-0302(91)78551-2
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267. doi:10.1128/AEM.00062-07
Weimer PJ, Moen GN (2013) Quantitative analysis of growth and volatile fatty acid production by the anaerobic ruminal bacterium Megasphaera elsdenii T81. Appl Microbiol Biotechnol 97(9):4075–4081. doi:10.1007/s00253-012-4645-4
Weißbach F, Strubelt C (2008) Die Korrektur des Trockensubstanzgehaltes von Maissilagen als Substrat für Biogasanlagen. Landtechnik 63(2):2–4
Wong BT, Show KY, Su A, Wong RJ, Lee DJ (2008) Effect of volatile fatty acid composition on upflow anaerobic sludge blanket (UASB) performance. Energ Fuel 22(1):108–112. doi:10.1021/Ef700282r
Zielonka S, Lemmer A, Oechsner H, Jungbluth T (2010) Energy balance of a two-phase anaerobic digestion process for energy crops. Eng Life Sci 10(6):515–519. doi:10.1002/elsc.201000071
Ziganshin AM, Liebetrau J, Pröter J, Kleinsteuber S (2013) Microbial community structure and dynamics during anaerobic digestion of various agricultural waste materials. Appl Microbiol Biotechnol 97(11):5161–5174. doi:10.1007/s00253-013-4867-0
Acknowledgments
This work was supported by the Initiative and Networking Fund of the Helmholtz Association. We would like to thank Ronny Kirbach and Peter Keil for operating the two-phase reactor system and analysis of process parameters. We thank our collaboration partners from the Department Biochemical Conversion of the Deutsches Biomasseforschungszentrum (DBFZ) for contributing to the analytical measurements. Furthermore, we thank Dorota Rzechonek and Anne Kuchenbuch for T-RFLP analyses of bacterial communities, Franziska Bühligen for T-RFLP analyses of the methanogenic community and Ute Lohse for technical assistance with 454 pyrosequencing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 508 kb)
Rights and permissions
About this article
Cite this article
Sträuber, H., Lucas, R. & Kleinsteuber, S. Metabolic and microbial community dynamics during the anaerobic digestion of maize silage in a two-phase process. Appl Microbiol Biotechnol 100, 479–491 (2016). https://doi.org/10.1007/s00253-015-6996-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6996-0