Abstract
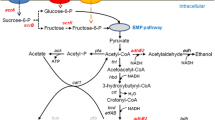
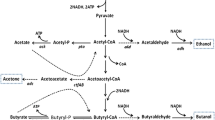
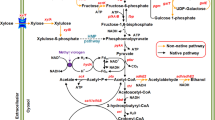
Clostridium tyrobutyricum does not have the enzymes needed for using maltose or starch. Two extracellular α-glucosidases encoded by agluI and agluII from Clostridium acetobutylicum ATCC 824 catalyzing the hydrolysis of α-1,4-glycosidic bonds in maltose and starch from the non-reducing end were cloned and expressed in C. tyrobutyricum (Δack, adhE2), and their effects on n-butanol production from maltose and soluble starch in batch fermentations were studied. Compared to the parental strain grown on glucose, mutants expressing agluI showed robust activity in breaking down maltose and produced more butanol (17.2 vs. 9.5 g/L) with a higher butanol yield (0.20 vs. 0.10 g/g) and productivity (0.29 vs. 0.16 g/L h). The mutant was also able to use soluble starch as substrate, although at a slower rate compared to maltose. Compared to C. acetobutylicum ATCC 824, the mutant produced more butanol from maltose (17.2 vs. 11.2 g/L) and soluble starch (16.2 vs. 8.8 g/L) in batch fermentations. The mutant was stable in batch fermentation without adding antibiotics, achieving a high butanol productivity of 0.40 g/L h. This mutant strain thus can be used in industrial production of n-butanol from maltose and soluble starch.




Similar content being viewed by others
References
Albasheri KA, Mitchell WJ (1995) Identification of two α-glucosidase activities in Clostridium acetobutylicum NCIB 8052. J Appl Microbiol 78:149–156
Al-Hinai MA, Fast AG, Papoutsakis ET (2012) Novel system for efficient isolation of Clostridium double-crossover allelic exchange mutants enabling markerless chromosomal gene deletions and DNA integration. Appl Environ Microbiol 78:8112–21
Annous BA, Blaschek HP (1990) Regulation and localization of amylolytic enzymes in Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 56:2559–2661
Annous BA, Blaschek HP (1991) Isolation and characterization of Clostridium acetobutylicum mutants with enhanced amylolytic activity. Appl Environ Microbiol 57:2544–8
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Boulton C, Quain D (2006) Brewing yeast and fermentation. Wiley-Blackwell, New York, NY
Chojecki A, Blaschek HP (1986) Effect of carbohydrate source on α-amylase and glucoamylase formation by Clostridium acetobutylicum SA-1. J Ind Microbiol 1:63–67
Du Y, Jiang W, Yu M, Tang IC, Yang ST (2015) Metabolic process engineering of Clostridium tyrobutyricum Δack-adhE2 for enhanced n-butanol production from glucose: effects of methyl viologen on NADH availability, flux distribution and fermentation kinetics. Biotechnol Bioeng 112:705–715
Dwidar M, Park JY, Mitchell RJ, Sang BI (2012) The future of butyric acid in industry. Sci World J 2012:471417
Fontaine L, Meynial-Salles I, Girbal L, Yang X, Croux C, Soucaille P (2002) Molecular characterization and transcriptional analysis of adhE2, the gene encoding the NADH-dependent aldehyde/alcohol dehydrogenase responsible for butanol production in alcohologenic cultures of Clostridium acetobutylicum ATCC 824. J Bacteriol 184:821–830
Green EM (2011) Fermentative production of butanol—the industrial perspective. Curr Opin Biotechnol 22:337–343
Gu Y, Jiang Y, Wu H, Liu X, Li Z, Li J, Xiao H, Shen Z, Dong H, Yang Y, Li Y, Jiang W, Yang S (2011) Economical challenges to microbial producers of butanol: feedstock, butanol ratio and titer. Biotechnol J 6:1348–1357
Heap JT, Pennington OJ, Cartman ST, Carter GP, Minton NP (2007) The ClosTron: a universal gene knock-out system for the genus Clostridium. J Microbiol Methods 70:452–464
Heap JT, Pennington OJ, Cartman ST, Minton NP (2009) A modular system for Clostridium shuttle plasmids. J Microbiol Methods 78:79–85
Huang L, Forsberg CW, Gibbins LN (1986) Influence of external pH and fermentation products on Clostridium acetobutylicum intracellular pH and cellular distribution of fermentation products. Appl Environ Microbiol 51:1230–1234
Jiang W, Zhao J, Wang Z, Yang ST (2014) Stable high-titer n-butanol production from sucrose and sugarcane juice by Clostridium acetobutylicum JB200 in repeated batch fermentations. Bioresour Technol 163:172–179
Jones DT, Woods DR (1986) Acetone-butanol fermentation revisited. Microbiol Rev 50:484–524
Kovács K, Willson BJ, Schwarz K, Heap JT, Jackson A, Bolam DN, Winzer K, Minton NP (2013) Secretion and assembly of functional mini-cellulosomes from synthetic chromosomal operons in Clostridium acetobutylicum ATCC 824. Biotechnol Biofuels 6:117
Lee SY, Park JH, Jang SH, Nielsen LK, Kim J, Jung KS (2008) Fermentative butanol production by clostridia. Biotechnol Bioeng 101:209–228
Liu X, Zhu Y, Yang ST (2005) Butyric acid and hydrogen production by Clostridium tyrobutyricum ATCC 25755 and mutants. Enzym Microb Technol 38:521–528
Liu X, Zhu Y, Yang ST (2006) Construction and characterization of ack deleted mutant of Clostridium tyrobutyricum for enhanced butyric acid and hydrogen production. Biotechnol Prog 22:1265–1275
Lütke-Eversloh T (2014) Application of new metabolic engineering tools for Clostridium acetobutylicum. Appl Microbiol Biotechnol 98:5823–5837
Madihah MS, Ariff AB, Khalil MS, Suraini AA, Karim MI (2001) Anaerobic fermentation of gelatinized sago starch-derived sugars to acetone-1-butanol-ethanol solvent by Clostridium acetobutylicum. Folia Microbiol (Praha) 46:197–204
Mariano AP, Dias MO, Junqueira TL, Cunha MP, Bonomi A, Filho RM (2013) Butanol production in a first-generation Brazilian sugarcane biorefinery: technical aspects and economics of greenfield projects. Bioresour Technol 135:316–323
McMahon H, Zoecklein BW, Fugelsang K, Jasinski Y (1999) Quantification of glycosidase activities in selected yeasts and lactic acid bacteria. J Ind Microbiol Biotechnol 23:198–203
Ni Y, Sun Z (2009) Recent progress on industrial fermentative production of acetone-butanol-ethanol by Clostridium acetobutylicum in China. Appl Microbiol Biotechnol 83:415–23
Nicolaou SA, Gaida SM, Papoutsakis ET (2010) A comparative view of metabolite and substrate stress and tolerance in microbial bioprocessing: from biofuels and chemicals, to biocatalysis and bioremediation. Metab Eng 12:307–331
Novichkov PS, Laikova ON, Novichkova ES, Gelfand MS, Arkin AP, Dubchak I, Rodionov DA (2010) RegPrecise: a database of curated genomic inferences of transcriptional regulatory interactions in prokaryotes. Nucleic Acids Res 38:D111–D118
Papoutsakis ET (2008) Engineering solventogenic clostridia. Curr Opin Biotechnol 19:420–429
Paquet V, Croux C, Goma G, Soucaille P (1991) Purification and characterization of the extracellular α-amylase from Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 57:212–218
Qureshi N, Blaschek HP (2001) ABE production from corn: a recent economic evaluation. J Ind Microbiol Biotechnol 27:292–297
Servinsky MD, Kiel JT, Dupuy NF, Sund CJ (2010) Transcriptional analysis of differential carbohydrate utilization by Clostridium acetobutylicum. Microbiology 156:3478–91
Soni BK, Kapp C, Goma G, Soucaille P (1992) Solvent production from starch: effect of pH on α-amylase and glucoamylase localization and synthesis in synthetic medium. Appl Microbiol Biotechnol 37:539–534
Tangney M, Winters GT, Mitchell WJ (2001) Characterization of a maltose transport system in Clostridium acetobutylicum ATCC 824. J Ind Microbiol Biotechnol 27:298–306
Thompson J, Hess S, Pikis A (2004) Genes malh and pagl of Clostridium acetobutylicum ATCC 824 encode NAD+- and Mn2+-dependent phospho-α-glucosidase(s). J Biol Chem 279:1553–1561
Wang J, Yang X, Chen CC, Yang ST (2014) Engineering clostridia for butanol production from biorenewable resources: from cells to process integration. Curr Opin Chem Eng 6:43–54
Williams DR, Young DI, Young M (1990) Conjugative plasmid transfer from Escherichia coli to Clostridium acetobutylicum. Microbiology 136:819–826
Xu M, Zhao J, Yu L, Tang IC, Xue C, Yang ST (2015) Engineering Clostridium acetobutylicum with a histidine kinase knockout for enhanced n-butanol tolerance and production. Appl Microbiol Biotechnol 99:1011–1022
Xue C, Zhao X-Q, Liu CG, Chen L-J, Bai F-W (2013) Prospective and development of butanol as an advanced biofuel. Biotechnol Adv 31:1575–1584
Yu M, Zhang Y, Tang IC, Yang ST (2011) Metabolic engineering of Clostridium tyrobutyricum for n-butanol production. Metab Eng 13:373–382
Yu M, Du Y, Jiang W, Chang WL, Yang ST, Tang IC (2012) Effects of different replicons in conjugative plasmids on transformation efficiency, plasmid stability, gene expression and n-butanol biosynthesis in Clostridium tyrobutyricum. Appl Microbiol Biotechnol 93:881–889
Zhao J, Lu C, Chen CC, Yang ST (2013) Biological production of butanol and higher alcohols. In: Yang ST, El-Enshasy HA, Thongchul N (eds) Bioprocessing technologies in biorefinery for sustainable production of fuels, chemicals, and polymers, vol 13. Wiley, Hoboken, NJ, pp 235–261
Zheng Y, Li L, Xian M, Ma Y, Yang J, Xu X, He D (2009) Problems with the microbial production of butanol. J Ind Microbiol Biotechnol 36:1127–1138
Zhu Y, Yang ST (2003) Adaptation of Clostridium tyrobutyricum for enhanced tolerance to butyric acid in a fibrous-bed bioreactor. Biotechnol Prog 19:365–372
Acknowledgments
This work was supported in part by the National Science Foundation STTR program (IIP-1026648).
Compliance with ethical standards
This research does not involve human participants or animals.
Conflict of interest
The authors declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 113 kb)
Rights and permissions
About this article
Cite this article
Yu, L., Xu, M., Tang, IC. et al. Metabolic engineering of Clostridium tyrobutyricum for n-butanol production from maltose and soluble starch by overexpressing α-glucosidase. Appl Microbiol Biotechnol 99, 6155–6165 (2015). https://doi.org/10.1007/s00253-015-6680-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6680-4