Abstract
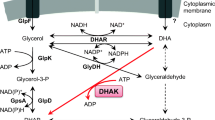
Corynebacterium glutamicum can consume glucose to excrete glycerol under oxygen deprivation. Although glycerol synthesis from 1,3-dihydroxyacetone (DHA) has been speculated, no direct evidence has yet been provided in C. glutamicum. Enzymatic and genetic investigations here indicate that the glycerol is largely produced from DHA and, unexpectedly, the reaction is catalyzed by (S,S)-butanediol dehydrogenase (ButA) that inherently catalyzes the interconversion between S-acetoin and (S,S)-2,3-butanediol. Consequently, the following pathway for glycerol biosynthesis in the bacterium emerges: dihydroxyacetone phosphate is dephosphorylated by HdpA to DHA, which is subsequently reduced to glycerol by ButA. This study emphasizes the importance of promiscuous activity of the enzyme in vivo.



Similar content being viewed by others
References
Becker J, Klopprogge C, Wittmann C (2008) Metabolic responses to pyruvate kinase deletion in lysine producing Corynebacterium glutamicum. Microb Cell Factories 7:8
Becker J, Klopprogge C, Zelder O, Heinzle E, Wittmann C (2005) Amplified expression of fructose 1,6-bisphosphatase in Corynebacterium glutamicum increases in vivo flux through the pentose phosphate pathway and lysine production on different carbon sources. Appl Environ Microbiol 71:8587–8596
Blomberg A, Adler L (1992) Physiology of osmotolerance in fungi. Adv Microb Physiol 33:145–212
Dominguez H, Rollin C, Guyonvarch A, Guerquin-Kern JL, Cocaign-Bousquet M, Lindley ND (1998) Carbon-flux distribution in the central metabolic pathways of Corynebacterium glutamicum during growth on fructose. Eur J Biochem 254:96–102
García-Martínez J, Castrillo M, Avalos J (2014) The gene cutA of Fusarium fujikuroi, encoding a protein of the haloacid dehalogenase family, is involved in osmotic stress and glycerol metabolism. Microbiology 160:26–36
Gutknecht R, Beutler R, Garcia-Alles LF, Baumann U, Erni B (2001) The dihydroxyacetone kinase of Escherichia coli utilizes a phosphoprotein instead of ATP as phosphoryl donor. EMBO J 20:2480–2486
Hasegawa S, Uematsu K, Natsuma Y, Suda M, Hiraga K, Jojima T, Inui M, Yukawa H (2011) Improvement of the redox balance increases L-valine production by Corynebacterium glutamicum under oxygen deprivation. Appl Environ Microbiol 78:865–875
Horbach S, Strohhacker J, Welle R, de Graaf A, Sahm H (1994) Enzymes involved in the formation of glycerol 3-phosphate and the by-products dihydroxyacetone and glycerol in Zymomonas mobilis. FEMS Microbiol Lett 120:37–44
Hult K, Berglund P (2007) Enzyme promiscuity: mechanism and applications. Trends Biotechnol 25:231–238
Ikeda M, Takeno S (2013) Amino acid production by Corynebacterium glutamicum. In: Yukawa H, Inui M (ed) Corynebacterium glutamicum, vol 23. Springer, Berlin Heidelberg, pp 107–147
Inui M, Kawaguchi H, Murakami S, Vertès AA, Yukawa H (2004) Metabolic engineering of Corynebacterium glutamicum for fuel ethanol production under oxygen-deprivation conditions. J Mol Microbiol Biotechnol 8:243–254
Ji XJ, Huang H, Ouyang PK (2011) Microbial 2,3-butanediol production: a state-of-the-art review. Biotechnol Adv 29:351–364
Jin RZ, Tang JC, Lin EC (1983) Experimental evolution of a novel pathway for glycerol dissimilation in Escherichia coli. J Mol Evol 19:429–436
Jojima T, Igari T, Gunji W, Suda M, Inui M, Yukawa H (2012) Identification of a HAD superfamily phosphatase, HdpA, involved in 1,3-dihydroxyacetone production during sugar catabolism in Corynebacterium glutamicum. FEBS Lett 586:4228–4232
Jojima T, Inui M, Yukawa H (2013) Biorefinery applications of Corynebacterium glutamicum. In: Yukawa H, Inui M (ed) Corynebacterium glutamicum, vol 23. Springer, Berlin Heidelberg, pp 149–172
Kiefer P, Heinzle E, Zelder O, Wittmann C (2004) Comparative metabolic flux analysis of lysine-producing Corynebacterium glutamicum cultured on glucose or fructose. Appl Environ Microbiol 70:229–239
Krömer JO, Sorgenfrei O, Klopprogge K, Heinzle E, Wittmann C (2004) In-depth profiling of lysine-producing Corynebacterium glutamicum by combined analysis of the transcriptome, metabolome, and fluxome. J Bacteriol 186:1769–1784
Lin EC (1976) Glycerol dissimilation and its regulation in bacteria. Annu Rev Microbiol 30:535–578
Lindner SN, Meiswinkel TM, Panhorst M, Youn JW, Wiefel L, Wendisch VF (2012) Glycerol-3-phosphatase of Corynebacterium glutamicum. J Biotechnol 159:216–224
Machielsen R, Uria AR, Kengen SWM, van der Oost J (2006) Production and characterization of a thermostable alcohol dehydrogenase that belongs to the Aldo-keto reductase superfamily. Appl Environ Microbiol 72:233–238
Neish AC, Blackwood AC, Ledingham GA (1945) Dissimilation of glucose by Bacillus subtilis (Ford’s strain). Can J Res 23:290–296
Nissen TL, Hamann CW, Kielland-Brandt MC, Nielsen J, Villadsen J (2000) Anaerobic and aerobic batch cultivations of Saccharomyces cerevisiae mutants impaired in glycerol synthesis. Yeast 16:463–474
Otagiri M, Ui S, Takusagawa Y, Ohtsuki T, Kurisu G, Kusunoki M (2010) Structural basis for chiral substrate recognition by two 2,3-butanediol dehydrogenases. FEBS Lett 584:219–223
Pepper RE, Costilow RN (1964) Glucose catabolism by Bacillus popilliae and Bacillus lentimorbus. J Bacteriol 87:303
Raspoet D, Pot B, De Deyn D, De Vos P, Kersters K, De Ley J (1991) Differentiation between 2, 3-butanediol producing Bacillus licheniformis and B. polymyxa strains by fermentation product profiles and whole-cell protein electrophoretic patterns. Syst Appl Microbiol 14:1–7
Suzuki N, Okai N, Nonaka H, Tsuge Y, Inui M, Yukawa H (2006) High-throughput transposon mutagenesis of Corynebacterium glutamicum and construction of a single-gene disruptant mutant library. Appl Environ Microbiol 72:3750–3755
Takusagawa Y, Otagiri M, Ui S, Ohtsuki T, Mimura A, Ohkuma M, Kudo T (2001) Purification and characterization of L-2, 3-butanediol dehydrogenase of Brevibacterium saccharolyticum C-1012 expressed in Escherichia coli. Biosci Biotechnol Biochem 65:1876–1878
Ui S, Masuda H, Muraki H (1983) Stereopecific and electrophoretic natures of bacterial 2, 3-butanediol dehydrogenases. J Ferment Technol 61:467–471
Ui S, Otagiri M, Mimura A, Dohmae N, Takio K, Ohkuma M, Kudo T (1998) Cloning, expression and nucleotide sequence of the l-2,3-butanediol dehydrogenase gene from Brevibacterium saccharolyticum C-1012. J Ferment Bioeng 86:290–295
Veiga-DA-Cunha M, Santos H, Van Schaftingen E (1993) Pathway and regulation of erythritol formation in Leuconostoc oenos. J Bacteriol 175:3941–3948
Viikari L, Berry DR (1988) Carbohydrate metabolism in Zymomonas. Crit Rev Biotechnol 7:237–261
Wang Y, Tao F, Xu P (2014) Glycerol dehydrogenase plays a dual role in glycerol metabolism and 2,3-butanediol formation in Klebsiella pneumoniae. J Biol Chem 289:6080–6090
Wang ZX, Zhuge J, Fang H, Prior BA (2001) Glycerol production by microbial fermentation: a review. Biotechnol Adv 19:201–223
Wittmann C, Kiefer P, Zelder O (2004) Metabolic fluxes in Corynebacterium glutamicum during lysine production with sucrose as carbon source. Appl Environ Microbiol 70:7277–7287
Wolf A, Krämer R, Morbach S (2003) Three pathways for trehalose metabolism in Corynebacterium glutamicum ATCC13032 and their significance in response to osmotic stress. Mol Microbiol 49:1119–1134
Yukawa H, Omumasaba CA, Nonaka H, Kos P, Okai N, Suzuki N, Suda M, Tsuge Y, Watanabe J, Ikeda Y, Vertes AA, Inui M (2007) Comparative analysis of the Corynebacterium glutamicum group and complete genome sequence of strain R. Microbiology 153:1042–1058
Acknowledgments
We thank Crispinus A. Omumasaba (RITE) for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 178 kb)
Rights and permissions
About this article
Cite this article
Jojima, T., Igari, T., Moteki, Y. et al. Promiscuous activity of (S,S)-butanediol dehydrogenase is responsible for glycerol production from 1,3-dihydroxyacetone in Corynebacterium glutamicum under oxygen-deprived conditions. Appl Microbiol Biotechnol 99, 1427–1433 (2015). https://doi.org/10.1007/s00253-014-6170-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6170-0