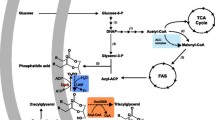
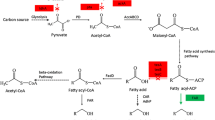
Abstract
This study investigated the production of triacylglycerols in cells of the wild type of Escherichia coli and of a strain with a deleted diacylglycerol kinase gene (dgkA). By overexpression of atfA from Acinetobacter baylyi ADP1 and fadD from E. coli in the dgkA deletion mutant, cellular contents of up to 4.9 % (w/w) triacylglycerols could be achieved in batch cultivation. Furthermore, heterologous expression of atfA relieves the negative effects of dgkA deletion on growth. Process optimization and fed-batch fermentation resulted in the production of 530 mg l−1 triacylglycerols and a maximal content of 8.5 % (w/w) triacylglycerols of the cell dry mass. This clearly exceeded all previous results concerning triacylglycerol production in E. coli. Furthermore, the production of extracellular free fatty acids and fatty acid ethyl esters was investigated. Like triacylglycerols, both products are potential biofuels, and we show their continuous production in a repeated batch process, with recovery of the production cells.





Similar content being viewed by others
References
Albright FR, White DA, Lennarz WJ (1973) Studies on enzymes involved in the catabolism of phospholipids in Escherichia coli. J Biol Chem 248:3968–3977
Alvarez HM, Mayer F, Fabritius D, Steinbüchel A (1996) Formation of intracytoplasmic lipid inclusions by Rhodococcus opacus strain PD630. Arch Microbiol 165:377–386
Arabolaza A, Rodriguez E, Altabe S, Alvarez H, Gramajo H (2008) Multiple pathways for triacylglycerol biosynthesis in Streptomyces coelicolor. Appl Environ Microbiol 74:2573–2582
Bokinsky G, Peralta-Yahya PP, George A, Holmes BM, Steen EJ, Dietrich J, Lee TS, Tullman-Ercek D, Voigt CA, Simmons BA, Keasling JD (2011) Synthesis of three advanced biofuels from ionic liquid-pretreated switchgrass using engineered Escherichia coli. Proc Natl Acad Sci U S A 108:19949–19954
Brandl H, Gross RA, Lenz RW, Fuller RC (1988) Pseudomonas oleovorans as a source of poly(beta-hydroxyalkanoates) for potential applications as biodegradable polyesters. Appl Environ Microbiol 54:1977–1982
Cho H, Cronan JE Jr (1995) Defective export of a periplasmic enzyme disrupts regulation of fatty acid synthesis. J Biol Chem 270:4216–4219
Comba S, Menendez-Bravo S, Arabolaza A, Gramajo H (2013) Identification and physiological characterization of phosphatidic acid phosphatase enzymes involved in triacylglycerol biosynthesis in Streptomyces coelicolor. Microb Cell Fact 12:9
Desbois AP, Smith VJ (2010) Antibacterial free fatty acids: activities, mechanisms of action and biotechnological potential. Appl Microbiol Biotechnol 85:1629–1642
Fixter LM, McCormack JG (1976) The effect of growth conditions on the wax content of various strains of Acinetobacter. Biochem Soc Trans 4:504–505
Heath RJ, Jackowski S, Rock CO (1994) Guanosine tetraphosphate inhibition of fatty acid and phospholipid synthesis in Escherichia coli is relieved by overexpression of glycerol-3-phosphate acyltransferase (plsB). J Biol Chem 269:26584–26590
Holtzapple E, Schmidt-Dannert C (2007) Biosynthesis of isoprenoid wax ester in Marinobacter hydrocarbonoclasticus DSM 8798: Identification and characterization of isoprenoid coenzyme A synthetase and wax ester synthases. J Bacteriol 189:3804–3812
Kalscheuer R, Steinbüchel A (2003) A novel bifunctional wax ester synthase/acyl-CoA:diacylglycerol acyltransferase mediates wax ester and triacylglycerol biosynthesis in Acinetobacter calcoaceticus ADP1. J Biol Chem 278:8075–8082
Kalscheuer R, Stölting T, Steinbüchel A (2006) Microdiesel: Escherichia coli engineered for fuel production. Microbiology (SGM) 152:2529–2536
Kennedy EP (1982) Osmotic regulation and the biosynthesis of membrane-derived oligosaccharides in Escherichia coli. Proc Natl Acad Sci U S A 79:1092–1095
Kol MA, Kuster DW, Boumann HA, de Cock H, Heck AJ, de Kruijff B, de Kroon AI (2004) Uptake and remodeling of exogenous phosphatidylethanolamine in E. coli. Biochim Biophys Acta 1636:205–212
Kovach ME, Elzer PH, Hill DS, Robertson GT, Farris MA, Roop RM II, Peterson KM (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166:175–176
Kurosawa K, Boccazzi P, de Almeida NM, Sinskey AJ (2010) High-cell-density batch fermentation of Rhodococcus opacus PD630 using a high glucose concentration for triacylglycerol production. J Biotechnol 147:212–218
Lageveen RG, Huisman GW, Preusting H, Ketelaar P, Eggink G, Witholt B (1988) Formation of polyesters by Pseudomonas oleovorans: Effect of substrates on formation and composition of poly-(R)-3-hydroxyalkanoates and poly-(R)-3-hydroxyalkenoates. Appl Environ Microbiol 54:2924–2932
Lennen RM, Pfleger BF (2013) Microbial production of fatty acid-derived fuels and chemicals. Curr Opin Biotechnol 21:1–10
Lennen RM, Kruziki MA, Kumar K, Zinkel RA, Burnum KE, Lipton MS, Hoover SW, Ranatunga DR, Wittkopp TM, Marner WD II, Pfleger BF (2011) Membrane stresses induced by overproduction of free fatty acids in Escherichia coli. Appl Environ Microbiol 77:8114–8128
Lin F, Chen Y, Levine R, Lee K, Yuan Y, Lin XN (2013) Improving fatty acid availability for bio-hydrocarbon production in Escherichia coli by metabolic engineering. PLOS ONE 8:e78595
Liu H, Yu C, Feng D, Cheng T, Meng X, Liu W, Zou H, Xian M (2012) Production of extracellular fatty acid using engineered Escherichia coli. Microb Cell Fact 11:41
Pech-Canul A, Nogales J, Miranda-Molina A, Alvarez L, Geiger O, Soto MJ, Lopez-Lara IM (2011) FadD is required for utilization of endogenous fatty acids released from membrane lipids. J Bacteriol 193:6295–6304
Raetz CR, Newman KF (1978) Neutral lipid accumulation in the membranes of Escherichia coli mutants lacking diglyceride kinase. J Biol Chem 253:3882–3887
Raetz CR, Newman KF (1979) Diglyceride kinase mutants of Escherichia coli: inner membrane association of 1,2-diglyceride and its relation to synthesis of membrane-derived oligosaccharides. J Bacteriol 137:860–868
Ray TK, Cronan JE Jr (1975) Acylation of sn-glycerol 3-phosphate in Escherichia coli. Study of reaction with native palmitoyl-acyl carrier protein. J Biol Chem 250:8422–8427
Rucker J, Paul J, Pfeifer BA, Lee K (2013) Engineering E. coli for triglyceride accumulation through native and heterologous metabolic reactions. Appl Microbiol Biotechnol 97:2753–2759
Ruffing AM, Jones HD (2012) Physiological effects of free fatty acid production in genetically engineered Synechococcus elongatus PCC 7942. Biotechnol Bioeng 109:2190–2199
Sambrook J, Fritsch EF, Maniatisch T (1989) Molecular cloning a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schlegel HG, Gottschalk G, von Bartha R (1961) Formation and utilization of poly-beta-hydroxybutyric acid by Knallgas bacteria (Hydrogenomonas). Nature 191:463–465
Shi S, Valle-Rodríguez JO, Khoomrung S, Siewers V, Nielsen J (2012) Functional expression and characterization of five wax ester synthases in Saccharomyces cerevisiae and their utility for biodiesel production. Biotechnol Biofuels 5:7
Steen EJ, Kang Y, Bokinsky G, Hu Z, Schirmer A, McClure A, Del Cardayre SB, Keasling JD (2010) Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 463:559–562
Touze T, Blanot D, Mengin-Lecreulx D (2008) Substrate specificity and membrane topology of Escherichia coli PgpB, an undecaprenyl pyrophosphate phosphatase. J Biol Chem 283:16573–16583
Uthoff S, Bröker D, Steinbüchel A (2009) Current state and perspectives of producing biodiesel-like compounds by biotechnology. Microb Biotechnol 2:551–565
Wältermann M, Luftmann H, Baumeister D, Kalscheuer R, Steinbüchel A (2000) Rhodococcus opacus strain PD630 as a new source of high-value single-cell oil? Isolation and characterization of triacylglycerols and other storage lipids. Microbiology (SGM) 146:1143–1149
Zhang YM, Rock CO (2008) Membrane lipid homeostasis in bacteria. Nat Rev Microbiol 6:222–233
Zhang F, Rodriguez S, Keasling JD (2011) Metabolic engineering of microbial pathways for advanced biofuels production. Curr Opin Biotechnol 22:775–783
Zhang F, Carothers JM, Keasling JD (2012) Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids. Nat Biotechnol 30:354–359
Zheng Y, Li L, Liu Q, Qin W, Yang J, Cao Y, Jiang X, Zhao G, Xian M (2012) Boosting the free fatty acid synthesis of Escherichia coli by expression of a cytosolic Acinetobacter baylyi thioesterase. Biotechnol Biofuels 5:76-6834-5-76
Competing interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Janßen, H.J., Steinbüchel, A. Production of triacylglycerols in Escherichia coli by deletion of the diacylglycerol kinase gene and heterologous overexpression of atfA from Acinetobacter baylyi ADP1. Appl Microbiol Biotechnol 98, 1913–1924 (2014). https://doi.org/10.1007/s00253-013-5460-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5460-2