Abstract
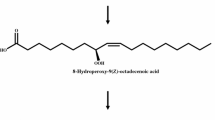
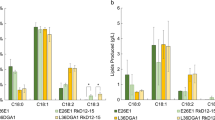
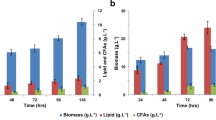
Among several fatty acids tested, oleic acid was selected as the most efficient inducer for the production of 4-hydroxydodecanoic acid, a metabolite of β-oxidation, by Waltomyces lipofer. Cells were induced by incubation for 12 h in a medium containing 10 g l−1 yeast extract, 10 g l−1 peptone, 5 g l−1 oleic acid, 1 g l−1 glucose, and 0.05 % (w/v) Tween 80. The optimal reaction conditions for the production of γ-lactones by induced cells were pH 6.5, 35 °C, 200 rpm, 0.71 M Tris, 60 g l−1 hydroxy fatty acid, and 20 g l−1 cells. Non-induced cells produced 38 g l−1 γ-dodecalactone from 60 g l−1 10-hydroxystearic acid after 30 h, with a conversion yield of 63 % (w/w) and a productivity of 1.3 g l−1 h−1 under the optimized conditions, whereas induced cells produced 51 g l−1 γ-dodecalactone from 60 g l−1 10-hydroxystearic acid after 30 h, with a conversion yield of 85 % (w/w) and a productivity of 1.7 g l−1 h−1. The conversion yield and productivity of induced cells were 22 % and 1.3-fold higher, respectively, than those of non-induced cells. Induced cells also produced 28 g l−1 γ-decalactone and 12 g l−1 γ-butyrolactone from 60 g l−1 12-hydroxystearic acid and 60 g l−1 10-hydroxydecanoic acid, respectively, after 30 h. The concentration, conversion yield, and productivity of γ-dodecalactone and γ-decalactone are the highest reported thus far. This is the first study on the biotechnological production of γ-butyrolactone.




Similar content being viewed by others
References
Aguedo M, Beney L, Wache Y, Belin JM (2003) Mechanisms underlying the toxicity of lactone aroma compounds towards the producing yeast cells. J Appl Microbiol 94:258–265
Alchihab M, Destain J, Aguedo M, Majad L, Ghalfi H, Wathelet JP, Thonart P (2009) Production of γ-decalactone by a psychrophilic and a mesophilic strain of the yeast Rhodotorula aurantiaca. Appl Biochem Biotechnol 158:41–50
An JU, Joo YC, Oh DK (2013) New biotransformation process for the production of the fragrance compound γ-dodecalactone from 10-hydroxystearate by permeabilized Waltomyces lipofer cells. Appl Environ Microbiol 79:2636–2641
Carrau FM, Medina K, Farina L, Boido E, Henschke PA, Dellacassa E (2008) Production of fermentation aroma compounds by Saccharomyces cerevisiae wine yeasts: effects of yeast assimilable nitrogen on two model strains. FEMS Yeast Res 8:1196–1207
Chen TC, Ju YH (2001) An improved fractional crystallization method for the enrichment of γ-linolenic acid in borage oil fatty acid. Ind Eng Chem Res 40:3781–3784
Dommes P, Dommes V, Kunau WH (1983) β-Oxidation in Candida tropicalis. Partial purification and biological function of an inducible 2,4-dienoyl coenzyme A reductase. J Biol Chem 258:10846–10852
Elss S, Preston C, Hertzig C, Heckel F, Richling E, Schreier P (2005) Aroma profiles of pineapple fruit (Ananas comosus [L.] Merr.) and pineapple products. Lwt-Food Sci Technol 38:263–274
Endrizzi A, Awade AC, Belin JM (1993) Presumptive involvement of methyl ricinoleate β-oxidation in the production of γ-decalactone by the yeast Pichia guilliermondii. FEMS Microbiol Lett 114:153–160
Endrizzi A, Pagot Y, Le Clainche A, Nicaud JM, Belin JM (1996) Production of lactones and peroxisomal β-oxidation in yeasts. Crit Rev Biotechnol 16:301–329
Farbood MI, Morris JA, Mclean LB (1998) Fermentation process for preparing 10-hydroxy-C18-carboxylic acid and γ-dodecalactone derivatives. European Patent 0578388
Farbood MI, Willis BJ, Ramsey NJ (1985) Production of γ-decalactone. U S Patent 4560656
Feron G, Dufosse L, Pierard E, Bonnarme P, Quere JL, Spinnler H (1996) Production, identification, and toxicity of γ-decalactone and 4-hydroxydecanoic acid from Sporidiobolus spp. Appl Environ Microbiol 62:2826–2831
Fickers P, Benetti PH, Wache Y, Marty A, Mauersberger S, Smit MS, Nicaud JM (2005) Hydrophobic substrate utilisation by the yeast Yarrowia lipolytica, and its potential applications. FEMS Yeast Res 5:527–543
Gatfield I (1988) Production of flavor and aroma compounds by biotechnology. Crit Rev Biotechnol 42:110–122
Han O, Han SR (1995) Process for production of C10 and/or C12 gamma-lactones from the corresponding C10 and/or C12 carboxylic acid by means of microbial biotransformation in the presence of mineral oil. United States Patent 5457036
Hara Y, Takahashi K (2002) A novel production of γ-butyrolactone catalyzed by homogeneous ruthenium complexes. Catal Surv Jpn 6:73–78
Joo YC, Seo ES, Kim YS, Kim KR, Park JB, Oh DK (2012) Production of 10-hydroxystearic acid from oleic acid by whole cells of recombinant Escherichia coli containing oleate hydratase from Stenotrophomonas maltophilia. J Biotechnol 158:17–23
Kawamoto S, Nozaki C, Tanaka A, Fukui S (1978) Fatty acid β-oxidation system in microbodies of n-alkane-grown Candida tropicalis. Eur J Biochem 83:609–613
Kondo A, Liu Y, Furuta M, Fujita Y, Matsumoto T, Fukuda H (2000) Preparation of high activity whole cell biocatalyst by permeabilization of recombinant flocculent yeast with alcohol. Enzyme Microb Technol 27:806–811
Lalel HJD, Singh Z, Tan SC (2003) Aroma volatiles production during fruit ripening of ‘Kensington Pride’ mango. Postharvest Biol Technol 27:323–336
Lee SL, Chen HY, Chen WC, Chou CC (1998) Production of -decalactone from ricinoleic acid by immobilized cells of Sporidiobolus salmonicolor. Process Biochem 33:453–459
Maga JA (1976) Lactones in foods. CRC Crit Rev Food Sci Nutr 8:1–56
Nicaud JM, Belin JM, Pagot Y, Joran EA (1996) Bioconversion of substrate with microbe auxotrophic for specific compound in medium deficient in this compound. French Patent 2734843
Pagot Y, Le Clainche A, Nicaud JM, Wache Y, Belin JM (1998) Peroxisomal β-oxidation activities and γ-decalactone production by the yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 49:295–300
Rabenhorst J, Gatfield I (2002) Method of producing γ-decalactone using Yarrowia lipolytica strain HR 145 (DSM 12397). United States Patent 6451565
Romero-Guido C, Belo I, Ta TM, Cao-Hoang L, Alchihab M, Gomes N, Thonart P, Teixeira JA, Destain J, Wache Y (2011) Biochemistry of lactone formation in yeast and fungi and its utilisation for the production of flavour and fragrance compounds. Appl Microbiol Biotechnol 89:535–547
Schöttler M, Boland W (1995) On the biosynthesis of γ-dodecanolactone in ripening fruits: flavor constituents from strawberries (Fragaria ananassa) and peaches (Prunus persica). Helv Chim Acta 78:847–856
Sulter GJ, Waterham HR, Goodman JM, Veenhuis M (1990) Proliferation and metabolic significance of peroxisomes in Candida boidinii during growth on D-alanine or oleic acid as the sole carbon source. Arch Microbiol 153:485–489
Ta TM, Cao-Hoang L, Phan-Thi H, Tran HD, Souffou N, Gresti J, Marechal PA, Cavin JF, Wache Y (2010) New insights into the effect of medium-chain-length lactones on yeast membranes. Importance of the culture medium. Appl Microbiol Biotechnol 87:1089–1099
Titorenko VI, Nicaud JM, Wang H, Chan H, Rachubinski RA (2002) Acyl-CoA oxidase is imported as a heteropentameric, cofactor-containing complex into peroxisomes of Yarrowia lipolytica. J Cell Biol 156:481–494
Valenciano S, Lucas JR, Pedregosa A, Monistrol IF, Laborda F (1996) Induction of β-oxidation enzymes and microbody proliferation in Aspergillus nidulans. Arch Microbiol 166:336–341
Veenhuis M, Mateblowski M, Kunau WH, Harder W (1987) Proliferation of microbodies in Saccharomyces cerevisiae. Yeast 3:77–84
Vose J, Tighe T, Schwartz M, Buel E (2001) Detection of γ-butyrolactone (GBL) as a natural component in wine. J Forensic Sci 46:1164–1167
Wache Y, Aguedo M, Nicaud JM, Belin JM (2003) Catabolism of hydroxyacids and biotechnological production of lactones by Yarrowia lipolytica. Appl Microbiol Biotechnol 61:393–404
Wilson WA, Hawley SA, Hardie DG (1996) Glucose repression/derepression in budding yeast: SNF1 protein kinase is activated by phosphorylation under derepressing conditions, and this correlates with a high AMP:ATP ratio. Curr Biol 6:1426–1434
Acknowledgments
This study was supported by a grant (no. 112002–3) from the Bio-industry Technology Development Program, Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
An, JU., Oh, DK. Increased production of γ-lactones from hydroxy fatty acids by whole Waltomyces lipofer cells induced with oleic acid. Appl Microbiol Biotechnol 97, 8265–8272 (2013). https://doi.org/10.1007/s00253-013-5089-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5089-1