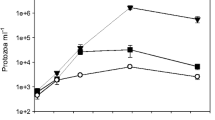
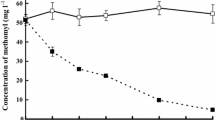
Abstract
Micropollutants in groundwater are given significant attention by water companies and authorities due to an increasing awareness that they might be present even above the legal threshold values. As part of our investigations of the possibility to remove the common groundwater pollutant 2,6-dichlorobenzamide (BAM) by introducing the efficient BAM degrader Aminobacter sp. MSH1 into biologically active sand filters, we investigated if the strain adheres to filters containing various filter materials and if the initial adherence and subsequent degradation of BAM could be optimized. We found that most of the inoculated MSH1 cells adhered fast and that parameters like pH and ionic strength had only a minor influence on the adhesion despite huge influence on cell surface hydrophobicity. At the given growth protocol, the MSH1 strain apparently developed a subpopulation that had lost its ability to adhere to the filter materials, which was supported by attempted reinoculation of non-adhered cells. Analysis by quantitative PCR showed that most cells adhered in the top of the filters and that some of these were lost from the filters during initial operation, while insignificant losses occurred after 1 day of operation. The inoculated filters were found to degrade 2.7 μg/L BAM to below 0.1 μg/L at a 1.1-h residence time with insignificant formation of known degradation products. In conclusion, most filter materials and water types should be feasible for inoculation with the MSH1 strain, while more research into degradation at low concentrations and temperatures is needed before this technology is ready for use at actual waterworks.




Similar content being viewed by others
References
Ahammed MM, Meera V (2010) Metal oxide/hydroxide-coated dual-media filter for simultaneous removal of bacteria and heavy metals from natural waters. J Haz Mat 181:788–793
Arvin E, Nielsen L, Tully A, Albrechtsen H-J, Mosbæk H (2004) MTBE removal by biofiltration in a water works. In: Van Loosdrecht M, Clement J (eds) 2nd IWA leading-edge conference on water and wastewater treatment technologies. IWA, London, pp 120–121
Baumgarten T, Sperling S, Seifert J, Von Bergen M, Steiniger F, Wick LY, Heipieper HJ (2012) Membrane vesicle formation as a multiple-stress response mechanism enhances Pseudomonas putida DOT-T1E cell surface hydrophobicity and biofilm formation. Appl Environ Microbiol 78:6217–6224
Bolster CH, Mills AL, Hornberger GM, Herman JS (1999) Spatial distribution of deposited bacteria following Miscible Displacement Experiments in intact cores. Wat Resour Res 35:1797–1807
Bolster CH, Cook KL, Marcus IM, Haznedaroglu BZ, Walker SL (2010) Correlating transport behavior with cell properties for eight porcine Escherichia coli isolates. Environ Sci Technol 44:5008–5014
DeFlaun M, Murray C, Holben W, Scheibe T, Mills A, Ginn T, Griffin T, Majer E, Wilson J (1997) Preliminary observations on bacterial transport in a coastal plain aquifer. FEMS Microbiol Rev 20:473–487
Gregg SJ, Sing KSW (1991) Adsorption, surface area, and porosity, 2nd edn. Academic, London
Harms H, Zehnder AJ (1994) Influence of substrate diffusion on degradation of dibenzofuran and 3-chlorodibenzofuran by attached and suspended bacteria. Appl Environ Microbiol 60:2736–2745
Harvey RW, Harms H (2002) Transport of microorganisms in the terrestrial subsurface: in situ and laboratory methods. In: Hurst CJ, Knudsen GR, McInerney MJ, Stetzenbach LD, Crawford R (eds) Manual of environmental microbiology, 2nd edn. ASM, Washington, DC, pp 753–776
Heise S, Gust G (1999) Influence of the physiological status of bacteria on their transport into permeable sediments. Mar Ecol Prog Ser 190:141–153
Ho L, Meyn T, Keegan A, Hoefel D, Brookes J, Saint CP, Newcombe G (2006) Bacterial degradation of microcystin toxins within a biologically active sand filter. Wat Res 40:768–774
Kinoshita T, Bales RC, Yahya MT, Gerba CP (1993) Bacteria transport in a porous medium: retention of Bacillus and Pseudomonas on silica surfaces. Wat Res 27:1295–1301
Kosmulski M (2009) Surface charging and points of zero charge. CRC, Boca Raton
Lee C-G, Park S-J, Han Y-U, Park J-A, Kim S-B (2010) Bacterial attachment and detachment in aluminum-coated quartz sand in response to ionic strength change. Wat Environ Res 82:499–505
Li B, Logan BE (2004) Bacterial adhesion to glass and metal-oxide surfaces. Coll Surf B Biointerfaces 36:81–90
Manem J, Rittmann B (1992) Removing trace-level organic pollutants in a biological filter. J AWWA 84:152–157
McDowall B, Hoefel D, Newcombe G, Saint CP, Ho L (2009) Enhancing the biofiltration of geosmin by seeding sand filter columns with a consortium of geosmin-degrading bacteria. Wat Res 43:433–440
Meffe R, Kohfahl C, Holzbecher E, Massmann G, Richter D, Dünnbier U, Pekdeger A (2010) Modelling the removal of p-TSA (para-toluenesulfonamide) during rapid sand filtration used for drinking water treatment. Wat Res 44:205–213
Mills AL, Herman JS, Hornberger GM, DeJesus TH (1994) Effect of solution ionic strength and iron coatings on mineral grains on the sorption of bacterial cells to quartz sand. Appl Environ Microbiol 60:3300–3306
Park S-J, Kim S-B (2010) Influence of (bi)carbonate on bacterial interaction with quartz and metal oxide-coated surfaces. Coll Surf B Biointerfaces 76:57–62
Park SJ, Lee C-G, Kim SB (2008) Quantification of bacterial attachment-related parameters in porous media. Environ Eng Res 13:141–146
Rijnaarts HHM, Norde W, Bouwer EJ, Lyklema J, Zehnder AJB (1996) Bacterial deposition in porous media: effects of cell-coating, substratum hydrophobicity, and electrolyte concentration. Environ Sci Technol 30:2877–2883
Rosenberg M, Gutnick D, Rosenberg E (1980) Adherence of bacteria to hydrocarbons: a simple method for measuring cell-surface hydrophobicity. FEMS Microbiol Lett 9:29–33
Simoni SF, Harms H, Bosma TNP, Zehnder AJB (1998) Transport of bacteria through sand columns at low flow rates. Environ Sci Technol 32:2100–2105
Simoni SF, Schäfer A, Harms H, Zehnder AJ (2001) Factors affecting mass transfer limited biodegradation in saturated porous media. J Cont Hydrol 50:99–120
Simonsen A (2010) Physiological characterisation of bacteria degrading dichlobenil and linuron herbicides. PhD thesis, Geological Survey of Denmark & Greenland
Simonsen A, Badawi N, Anskjær GG, Albers CN, Sørensen SR, Sørensen J, Aamand J (2012) Intermediate accumulation of metabolites results in a bottleneck for mineralisation of the herbicide metabolite 2,6-dichlorobenzamide (BAM) by Aminobacter spp. Appl Microbiol Biotechnol 94:237–245
Sjøholm OR, Aamand J, Sørensen J, Nybroe O (2010a) Degrader density determines spatial variability of 2,6-dichlorobenzamide mineralisation in soil. Environ Poll 158:292–298
Sjøholm OR, Nybroe O, Aamand J, Sørensen J (2010b) 2,6-Dichlorobenzamide (BAM) herbicide mineralisation by Aminobacter sp. MSH1 during starvation depends on a subpopulation of intact cells maintaining vital membrane functions. Environ Poll 158:3618–3625
Sørensen SR, Aamand J (2003) Rapid mineralisation of the herbicide isoproturon in soil from a previously treated Danish agricultural field. Pest Manag Sci 59:1118–1124
Sørensen SR, Holtze MS, Simonsen A, Aamand J (2007) Degradation and mineralization of nanomolar concentrations of the herbicide dichlobenil and its persistent metabolite 2,6-dichlorobenzamide by Aminobacter spp. isolated from dichlobenil-treated soils. Appl Environ Microbiol 73:399–406
Thorling L, Hansen B, Langtofte L, Brüsch W, Møller RR, Mielby S, Højbjerg AL (2011) Groundwater monitoring 2011 (Grundvandsovervågning 2011) in Danish with English summary (www.grundvandsovervaagning.dk)
Tros M, Schraa G, Zehnder AJB, Bosma TNP (1998) Anomalies in the transformation of 3-chlorobenzoate in percolation columns with Pseudomonas sp. strain b13. Wat Sci Technol 37:89–96
Zearley TL, Summers RS (2012) Removal of trace organic micropollutants by drinking water biological filters. Environ Sci Technol 46:9412–9419
Acknowledgments
The study was financially supported by the MIRESOWA project (Danish Strategic Research Council grant no. 2104-08-0012). Kerteminde, Hvidovre, Astrup, and Aike Waterworks are thanked for allowing sampling of filter materials.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 3080 kb)
Rights and permissions
About this article
Cite this article
Albers, C.N., Jacobsen, O.S. & Aamand, J. Using 2,6-dichlorobenzamide (BAM) degrading Aminobacter sp. MSH1 in flow through biofilters—initial adhesion and BAM degradation potentials. Appl Microbiol Biotechnol 98, 957–967 (2014). https://doi.org/10.1007/s00253-013-4942-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-4942-6