Abstract
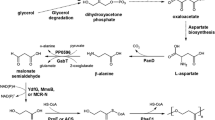
Ethylene glycol (EG) is an important platform chemical with steadily expanding global demand. Its commercial production is currently limited to fossil resources; no biosynthesis route has been delineated. Herein, a biosynthesis route for EG production from d-xylose is reported. This route consists of four steps: d-xylose → d-xylonate → 2-dehydro-3-deoxy-d-pentonate → glycoaldehyde → EG. Respective enzymes, d-xylose dehydrogenase, d-xylonate dehydratase, 2-dehydro-3-deoxy-d-pentonate aldolase, and glycoaldehyde reductase, were assembled. The route was implemented in a metabolically engineered Escherichia coli, in which the d-xylose → d-xylulose reaction was prevented by disrupting the d-xylose isomerase gene. The most efficient construct produced 11.7 g L−1 of EG from 40.0 g L−1 of d-xylose. Glycolate is a carbon-competing by-product during EG production in E. coli; blockage of glycoaldehyde → glycolate reaction was also performed by disrupting the gene encoding aldehyde dehydrogenase, but from this approach, EG productivity was not improved but rather led to d-xylonate accumulation. To channel more carbon flux towards EG than the glycolate pathway, further systematic metabolic engineering and fermentation optimization studies are still required to improve EG productivity.







Similar content being viewed by others
References
Aggarwal SL, Sweeting OJ (1957) Polyethylene: preparation, structure, and properties. Chem Rev 57:665–742
Baba T, Ara T, Hasegawa M, Takai Y, Okumura Y, Baba M, Datsenko KA, Tomita M, Wanner BL, Mori H (2006) Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol Syst Biol 2:2006.0008
Baldomà L, Aguilar J (1987) Involvement of lactaldehyde dehydrogenase in several metabolic pathways of Escherichia coli K12. J Biol Chem 262:13991–13996
Baldomà L, Aguilar J (1988) Metabolism of l-fucose and l-rhamnose in Escherichia coli: aerobic–anaerobic regulation of l-lactaldehyde dissimilation. J Bacteriol 170:416–421
Baudot A, Odagescu V (2004) Thermal properties of ethylene glycol aqueous solutions. Cryobiology 48:283–294
Benko Z, Andersson A, Szengyel Z, Gáspár M, Réczey K, Stålbrand H (2007) Heat extraction of corn fiber hemicelluloses. Appl Biochem Biotechnol 137–140:253–265
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Cherepanov PP, Wackernagel W (1995) Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158:9–14
Dahms AS (1974) 3-Deoxy-d-pentulosonic acid aldolase and its role in a new pathway of d-xylose degradation. Biochem Biophys Res Commun 60:1433–1439
Frost JW (2008) MEWG: interagency conference on metabolic engineering. Work presentation, North Bethesda. <URL: http://www.powershow.com/view1/10021d-Zjc0O/> (accessed 12 February 2010)
Frost JW, Niu W (2008) Microbial synthesis of d-1,2,4-butanetriol. Patent, Pub. No.: WO 2008/091288 A2
Himfr T (2011) Analysts predict overcapacity critical situation of glycol worldwide. <URL: http://www.articleforge.com.> (accessed 6 March 2012)
Jarboe LR (2010) YqhD: a broad-substrate range aldehyde reductase with various applications in production of biorenewable fuels and chemicals. Appl Microbiol Biotechnol 89:249–257
Jiang X, Malley K, Jensen HM (2009) Nanobio: protocol for gene knockout. <URL: http://openwetware.org/index.php?title=NanoBio: Protocol_for_gene_knockout &oldid=339609> (accessed 7 June 2010)
Lee C, Kim I, Lee J, Lee KL, Min B, Park C (2010) Transcriptional activation of the aldehyde reductase YqhD by YqhC and its implication in glyoxal metabolism of Escherichia coli K-12. J Bacteriol 192:4205–4214
Liu H, Valdehuesa KN, Nisola GM, Ramos KR, Chung WJ (2012) High yield production of d-xylonic acid from d-xylose using engineered Escherichia coli. Bioresour Technol 115:244–248
Macgee J, Doudoroff M (1954) A new phosphorylated intermediate in glucose oxidation. J Biol Chem 210:617–626
Mavrovouniotis ML (1999) Group contributions for estimating standard Gibbs energies of formation of biochemical compounds in aqueous solution. Biotechnol Bioeng 36:1070–1082
Novagen (2005) λDE3 lysogenization kit user protocol TB031 Rev. C 0805 <URL: http://www.emdmillipore.com/life-science-research/technical-bulletins/c_IMOb.s1OXkUAAAEj2xsYzMkq> (accessed 10 June 2010)
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor
Sathitsuksanoh N, Zhu Z, Zhang YH (2012) Cellulose solvent- and organic solvent-based lignocellulose fractionation enabled efficient sugar release from a variety of lignocellulosic feedstocks. Bioresour Technol 117C:228–233
Sun J, Liu H (2011) Selective hydrogenolysis of biomass-derived xylitol to ethylene glycol and propylene glycol on supported Ru catalysts. Green Chem 13:135–142
Vispute TP, Zhang H, Sanna A, Xiao R, Huber GW (2010) Renewable chemical commodity feedstocks from integrated catalytic processing of pyrolysis oils. Science 330:1222–1227
Wargacki AJ, Leonard E, Win MN, Regitsky DD, Santos CN, Kim PB, Cooper SR, Raisner RM, Herman A, Sivitz AB, Lakshmanaswamy A, Kashiyama Y, Baker D, Yoshikuni Y (2012) An engineered microbial platform for direct biofuel production from brown macroalgae. Science 335:308–313
Warren L (1959) The thiobarbituric acid assay of sialic acids. J Biol Chem 234:1971–1975
Yim H, Haselbeck R, Niu W, Pujol-Baxley C, Burgard A, Boldt J, Khandurina J, Trawick JD, Osterhout RE, Stephen R, Estadilla J, Teisan S, Schreyer HB, Andrae S, Yang TH, Lee SY, Burk MJ, Van Dien S (2011) Metabolic engineering of Escherichia coli for direct production of 1,4-butanediol. Nat Chem Biol 7:445–452
Yue H, Zhao Y, Ma X, Gong J (2012) Ethylene glycol: properties, synthesis, and applications. Chem Soc Rev 41:4218–4244
Zeng AP, Sabra W (2011) Microbial production of diols as platform chemicals: recent progresses. Curr Opin Biotechnol 22:749–757
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea funded by the Ministry of Education, Science, and Technology (2012–0006693). The authors would like to thank Professor Chankyu Park from Korea Advanced Institute of Science and Technology (KAIST) for generously providing the pET15a-yqhD plasmid and Professor Jaejoon Han from Sungkyunkwan University for kindly providing a reagent for the enzyme activity assays.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H., Ramos, K.R.M., Valdehuesa, K.N.G. et al. Biosynthesis of ethylene glycol in Escherichia coli . Appl Microbiol Biotechnol 97, 3409–3417 (2013). https://doi.org/10.1007/s00253-012-4618-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4618-7