Abstract
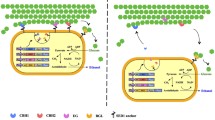
Enzymatic hydrolysis of cellulosic material is an essential step in the bioethanol production process. However, complete cellulose hydrolysis by cellulase is difficult due to the irreversible adsorption of cellulase onto cellulose. Thus, part of the cellulose remains in crystalline form after hydrolysis. In this study, after 96-h hydrolysis of Avicel crystalline cellulose, 47.1 % of the cellulase was adsorbed on the cellulose surface with 10.8 % crystalline cellulose remaining. In simultaneous saccharification and fermentation of 100 g/L Avicel with 1.0 filter paper unit/mL cellulase, a wild-type yeast strain produced 44.7 g/L ethanol after 96 h. The yield of ethanol was 79.7 % of the theoretical yield. On the other hand, a recombinant yeast strain displaying various cellulases, such as β-glucosidase, cellobiohydrolase, and endoglucanase, produced 48.9 g/L ethanol, which corresponds to 87.3 % of the theoretical yield. Higher ethanol production appears to be attributable to higher efficiency of cellulase displayed on the cell surface. These results suggest that cellulases displayed on the yeast cell surface improve hydrolysis of Avicel crystalline cellulose. Indeed, after the 96-h simultaneous saccharification and fermentation using the cellulase-displaying yeast, the amount of residual cellulose was 1.5 g/L, one quarter of the cellulose remaining using the wild-type strain, a result of the alleviation of irreversible adsorption of cellulases on the crystalline cellulose.




Similar content being viewed by others
References
Adney B, Baker J (1996) Measurement of cellulase activities (LAP). NREL TP-510-42628. National Renewable Energy Laboratory, Golden CO
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Desai SG, Converse AO (1997) Substrate reactivity as a function of the extent of reaction in the enzymatic hydrolysis of lignocellulose. Biotechnol Bioeng 56:650–655
Eriksson T, Karlsson J, Tjerneld F (2002) A model explaining declining rate in hydrolysis of lignocellulose substrates with cellobiohydrolase I (Cel7A) and endoglucanase I (Cel7B) of Trichoderma reesei. Appl Biochem Biotechnol 101:41–60
Fujita Y, Ito J, Ueda M, Fukuda H, Kondo A (2004) Synergistic saccharification, and direct fermentation to ethanol, of amorphous cellulose by use of an engineered yeast strain codisplaying three types of cellulolytic enzyme. Appl Environ Microbiol 70:1207–1212
Galbe M, Zacchi G (2007) Pretreatment of lignocellulosic materials for efficient bioethanol production. Adv Biochem Eng Biotechnol 108:41–65
Gao D, Chundawat SPS, Uppugundla N, Balan V, Dale BE (2011) Binding characteristics of Trichoderma reesei cellulases on untreated, ammonia fiber expansion (AFEX), and dilute-acid pretreated lignocellulosic biomass. Biotechnol Bioeng 108:1788–1800
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 59:257–268
Gole A, Vyas S, Sainkar SR, Lachke A, Sastry M (2001) Enhanced temperature and pH stability of fatty amine-endoglucanase composites: fabrication, substrate protection, and biological activity. Langmuir 17:5964–5970
Hasunuma T, Kondo A (2012) Consolidated bioprocessing and simultaneous saccharification and fermentation of lignocellulose to ethanol with thermotolerant yeast strains. Process Biochem 47:1287–1294
Hasunuma T, Sung KM, Sanda T, Yoshimura K, Matsuda F, Kondo A (2011) Efficient fermentation of xylose to ethanol at high formic acid concentrations by metabolically engineered Saccharomyces cerevisiae. Appl Microbiol Biotechnol 90:997–1004
Karlsson M, Ekeroth J, Elwing H, Carlsson U (2005) Reduction of irreversible protein adsorption on solid surfaces by protein engineering for increased stability. J Biol Chem 280:25558–25564
Kondo A, Shigechi H, Abe M, Uyama K, Matsumoto T, Takahashi S, Ueda M, Tanaka A, Kishimoto M, Fukuda H (2002) High-level ethanol production from starch by a flocculent Saccharomyces cerevisiae strain displaying cell-surface glucoamylase. Appl Microbiol Biotechnol 58:291–296
Kumar R, Wyman CE (2009) Cellulase adsorption and relationship to features of corn stover solids produced by leading pretreatments. Biotechnol Bioeng 103:252–267
Lupoi JS, Smith EA (2011) Evaluation of nanoparticle-immobilized cellulase for improved ethanol yield in simultaneous saccharification and fermentation reactions. Biotechnol Bioeng 108:2835–2843
Ma A, Hu Q, Qu Y, Bai Z, Liu W, Zhuang G (2008) The enzymatic hydrolysis rate of cellulose decreases with irreversible adsorption of cellobiohydrolase I. Enzyme Microb Technol 42:543–547
Matano Y, Hasunuma T, Kondo A (2012a) Display of cellulases on the cell surface of Saccharomyces cerevisiae for high yield ethanol production from high-solid lignocellulosic biomass. Bioresour Technol 108:128–133
Matano Y, Hasunuma T, Kondo A (2012b) Cell recycle batch fermentation of high-solid lignocellulose using a recombinant cellulase-displaying yeast strain for high yield ethanol production in consolidated bioprocessing. Bioresour Technol (in press)
Nakamura N, Yamada R, Katahira S, Tanaka T, Fukuda H, Kondo A (2008) Effective xylose/cellobiose co-fermentation and ethanol production by xylose-assimilating S. cerevisiae via expression of β-glucosidase on its cell surface. Enzyme Microb Technol 43:233–236
Otter DE, Munro PA, Scott GK, Geddes R (1989) Desorption of Trichoderma reesei cellulase from cellulose by a range of desorbents. Biotechnol Bioeng 34:291–298
Palonen H, Tenkanen M, Linder M (1999) Dynamic interaction of Trichoderma reesei cellobiohydrolases Cel6A and Cel7A and cellulose at equilibrium and during hydrolysis. Appl Environ Microbiol 65:6–11
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton (2008) Determination of structural carbohydrates and lignin in biomass: laboratory 476 analytical procedure (LAP). NREL/TP-510-42618. National Renewable Energy Laboratory, Golden, CO
Taherzadeh MJ, Karimi K (2008) Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: a review. Int J Mol Sci 9:1621–1651
Ueda M, Tanaka A (2000) Cell surface engineering of yeast: construction of arming yeast with biocatalyst. J Biosci Bioeng 90:125–136
Väljamäe P, Sild V, Pettersson G, Johansson G (1998) The initial kinetics of hydrolysis by cellobiohydrolases I and II is consistent with a cellulose surface-erosion model. Eur J Biochem 253:469–475
Xiao Z, Zhang X, Gregg DJ, Saddler JN (2004) Effects of sugar inhibition on cellulases and beta-glucosidase during enzymatic hydrolysis of softwood substrates. Appl Biochem Biotechnol 113–116:1115–1126
Yamada R, Taniguchi N, Tanaka T, Ogino C, Fukuda H, Kondo A (2011) Direct ethanol production from cellulosic materials using a diploid strain of Saccharomyces cerevisiae with optimized cellulase expression. Biotechnol Biofuels 4:8
Yang B, Wyman CE (2006) BSA treatment to enhance enzymatic hydrolysis of cellulose in lignin containing substrates. Biotechnol Bioeng 94:611–617
Yang B, Willies DM, Wyman CE (2006) Changes in the enzymatic hydrolysis rate of avicel cellulose with conversion. Biotechnol Bioeng 94:1122–1128
Yang J, Zhang X, Yong Q, Yu S (2010) Three-stage hydrolysis to enhance enzymatic saccharification of steam-exploded corn stover. Bioresour Technol 101:4930–4935
Zhang YP, Lynd LR (2004) Toward an aggregated understanding of enzymatic hydrolysis of cellulose: noncomplexed cellulase systems. Biotechnol Bioeng 88:797–824
Zhu Z, Sathitsuksanoh N, Percival Zhang Y-H (2009) Direct quantitative determination of adsorbed cellulase on lignocellulosic biomass with its application to study cellulase desorption for potential recycling. Analyst 134:2267–2272
Acknowledgments
This work was supported through the project P07015 by the New Energy and Industrial Technology Development Organization (NEDO) under the sponsorship of the Ministry of Economy, Trade, and Industry (METI) of Japan. This work was also supported in part by Grants-in-Aid for Young Scientists (B) to TH from the Ministry of Education, Culture, Sports and Technology (MEXT) of Japan and a Special Coordination Fund for Promoting Science and Technology, Creation of Innovative Centers for Advanced Interdisciplinary Research Areas (Innovative Bioproduction Kobe) from MEXT, Japan.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matano, Y., Hasunuma, T. & Kondo, A. Simultaneous improvement of saccharification and ethanol production from crystalline cellulose by alleviation of irreversible adsorption of cellulase with a cell surface-engineered yeast strain. Appl Microbiol Biotechnol 97, 2231–2237 (2013). https://doi.org/10.1007/s00253-012-4587-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4587-x