Abstract
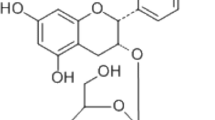
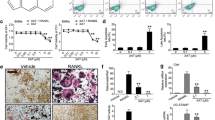
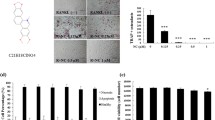
Mycoepoxydiene (MED) is a compound isolated from the marine fungal Diaporthe sp. HLY-1 associated with mangroves. MED has various biological effects such as anti-microbial, anti-cancer, and anti-inflammatory activities. However, the effect of MED on the differentiation of osteoclasts, the multinucleated bone-resorbing cells which play a crucial role in bone remodeling, is still unknown. In this study, we showed that MED could inhibit receptor activator of NF-κB ligand (RANKL)-induced osteoclast differentiation and the expression of three well-known osteoclast markers such as tartrate-resistant acid phosphatase, calcitonin receptor, and cathepsin K in bone marrow-derived macrophages. Furthermore, we found that MED inhibited the expression of nuclear factor of activated T cells c1, a key transcriptional factor in osteoclast differentiation, via inhibiting the phosphorylation of TAK1 and then blocking the activation of NF-κB and ERK1/2 pathways. Moreover, MED could prevent bone loss in ovariectomized mice. Taken together, we demonstrate for the first time that MED can suppress RANKL-induced osteoclast differentiation in vitro and ovariectomy-induced osteoporosis in vivo, suggesting that MED is a potential lead compound for the development of novel drugs for osteoporosis treatment.






Similar content being viewed by others
References
Arai F, Miyamoto T, Ohneda O, Inada T, Sudo T, Brasel K, Miyata T, Anderson DM, Suda T (1999) Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-Fms and receptor activator of nuclear factor kappaB (RANK) receptors. J Exp Med 190:1741–1754
Asagiri M, Takayanagi H (2007) The molecular understanding of osteoclast differentiation. Bone 40:251–264
Asagiri M, Sato K, Usami T, Ochi S, Nishina H, Yoshida H, Morita I, Ef W, Tw M, Serfling E, Takayanagi H (2005) Autoamplification of NFATc1 expression determines its essential role in bone homeostasis. J Exp Med 202:1261–1269
Baron R (2004) Arming the osteoclast. Nat Med 10:458–460
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342
Chen J, Sun Y, Mao X, Liu Q, Wu H, Chen Y (2010) RANKL up-regulates brain-type creatine kinase via poly(ADP-ribose) polymerase-1 during osteoclastogenesis. J Biol Chem 285:36315–36321
Darnay BG, Ni J, Moore PA, Aggarwal BB (1999) Activation of NF-kappaB by RANK requires tumor necrosis factor receptor-associated factor (TRAF) 6 and NF-kappaB-inducing kinase. Identification of a novel TRAF6 interaction motif. J Biol Chem 274:7724–7731
Franzoso G, Carlson L, Xing L, Poljak L, Ew S, Kd B, Leonardi A, Tran T, Bf B, Siebenlist U (1997) Requirement for NF-kappaB in osteoclast and B-cell development. Genes Dev 11:3482–3496
He Y, Staser K, Sd R, Liu Y, Wu X, Sj P, Yuan J, Yang X, Li X, Jiang L, Chen S, Fc Y (2011a) Erk1 positively regulates osteoclast differentiation and bone resorptive activity. PLoS One 6:e24780
He YX, Zhang G, Pan Xh, Liu Z, Lz Z, Cw C, Km L, Yp C, Li G, Wei L, Lk H, Ks L, Qin L (2011b) Impaired bone healing pattern in mice with ovariectomy-induced osteoporosis: a drill-hole defect model. Bone 48:1388–1400
Hirata K, Taki H, Shinoda K, Hounoki H, Miyahara T, Tobe K, Ogawa H, Mori H, Sugiyama E (2010) Inhibition of tumor progression locus 2 protein kinase suppresses receptor activator of nuclear factor-kappaB ligand-induced osteoclastogenesis through down-regulation of the c-Fos and nuclear factor of activated T cells c1 genes. Biol Pharm Bull 33:133–137
Hirotani H, Na T, Jt W, Stern Ph, Na C (2004) The calcineurin/nuclear factor of activated T cells signaling pathway regulates osteoclastogenesis in RAW264.7 cells. J Biol Chem 279:13984–13992
Huber DM, Ac B, Pathrose P, Srivastava S, Km D, Nk S, Jw P (2001) Androgens suppress osteoclast formation induced by RANKL and macrophage-colony stimulating factor. Endocrinology 142:3800–3808
Ishida N, Hayashi K, Hoshijima M, Ogawa T, Koga S, Miyatake Y, Kumegawa M, Kimura T, Takeya T (2002) Large scale gene expression analysis of osteoclastogenesis in vitro and elucidation of NFAT2 as a key regulator. J Biol Chem 277:41147–41156
Jiang J, Li H, Fs F, Filbert E, Ke S, Ls S, Zhu Q (2006) Quantitative analysis of osteoclast-specific gene markers stimulated by lipopolysaccharide. J Endod 32:742–746
Kobayashi N, Kadono Y, Naito A, Matsumoto K, Yamamoto T, Tanaka S, Inoue J (2001) Segregation of TRAF6-mediated signaling pathways clarifies its role in osteoclastogenesis. EMBO J 20:1271–1280
Kong YY, Yoshida H, Sarosi I, Tan HL, Timms E, Capparelli C, Morony S, Oliveira-Dos-Santos AJ, Van G, Itie A, Khoo W, Wakeham A, Dunstan CR, Lacey DL, Mak TW, Boyle WJ, Penninger JM (1999) OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397:315–323
Lin X, Huang Y, Fang M, Wang J, Zheng Z, Su W (2005) Cytotoxic and antimicrobial metabolites from marine lignicolous fungi, Diaporthe sp. FEMS Microbiol Lett 251:53–58
Mellis DJ, Itzstein C, Helfrich Mh, Jc C (2011) The skeleton: a multi-functional complex organ: the role of key signalling pathways in osteoclast differentiation and in bone resorption. J Endocrinol 211:131–143
Miyamoto T, Suda T (2003) Differentiation and function of osteoclasts. Keio J Med 52:1–7
Miyazaki T, Katagiri H, Kanegae Y, Takayanagi H, Sawada Y, Yamamoto A, Mp P, Asano T, Im V, Oda H, Nakamura K, Tanaka S (2000) Reciprocal role of ERK and NF-kappaB pathways in survival and activation of osteoclasts. J Cell Biol 148:333–342
Nakamura H, Hirata A, Tsuji T, Yamamoto T (2003) Role of osteoclast extracellular signal-regulated kinase (ERK) in cell survival and maintenance of cell polarity. J Bone Miner Res 18:1198–1205
Nakashima T, Hayashi M, Fukunaga T, Kurata K, Oh-Hora M, Jq F, Lf B, Kodama T, Wutz A, Ef W, Jm P, Takayanagi H (2011) Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat Med 17:1231–1234
Omata Y, Tanaka S (2011) RANKL/RANK signaling in rheumatoid arthritis. Clin Calcium 21:1175–1180
Remen KM, Henning P, Lerner Uh, Gustafsson JA, Andersson G (2011) Activation of liver X receptor (LXR) inhibits receptor activator of nuclear factor kappaB ligand (RANKL)-induced osteoclast differentiation in an LXRbeta-dependent mechanism. J Biol Chem 286:33084–33094
Suda T, Kobayashi K, Jimi E, Udagawa N, Takahashi N (2001) The molecular basis of osteoclast differentiation and activation. Novartis Found Symp 232:235–247, discussion 247-250
Takami M, Es C, Sy L, Kamijo R, Yim M (2005) Phosphodiesterase inhibitors stimulate osteoclast formation via TRANCE/RANKL expression in osteoblasts: possible involvement of ERK and p38 MAPK pathways. FEBS Lett 579:832–838
Takao K, Watanabe G, Yasui H, Tadano K (2002) Total synthesis of (+/−)-mycoepoxydiene, a novel fungal metabolite having an oxygen-bridged cyclooctadiene skeleton. Org Lett 4:2941–2943
Takatsuna H, Asagiri M, Kubota T, Oka K, Osada T, Sugiyama C, Saito H, Aoki K, Ohya K, Takayanagi H, Umezawa K (2005) Inhibition of RANKL-induced osteoclastogenesis by (−)-DHMEQ, a novel NF-kappaB inhibitor, through downregulation of NFATc1. J Bone Miner Res 20:653–662
Takayanagi H (2005) Inflammatory bone destruction and osteoimmunology. J Periodontal Res 40:287–293
Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H, Saiura A, Isobe M, Yokochi T, Inoue J, Ef W, Tw M, Kodama T, Taniguchi T (2002) Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell 3:889–901
Teitelbaum SL (2000) Bone resorption by osteoclasts. Science 289:1504–1508
Uchida R, Chiba H, Ishimi Y, Uehara M, Suzuki K, Kim H, Matsumoto A (2011) Combined effects of soy isoflavone and fish oil on ovariectomy-induced bone loss in mice. J Bone Miner Metab 29:404–413
Wang J, Zhao B, Zhang W, Wu X, Wang R, Huang Y, Chen D, Park K, Bc W, Shen Y (2010) Mycoepoxydiene, a fungal polyketide, induces cell cycle arrest at the G2/M phase and apoptosis in HeLa cells. Bioorg Med Chem Lett 20:7054–7058
Wiktor-Jedrzejczak W, Bartocci A, Aw Ferrante Jr, Ahmed-Ansari A, Sell Kw, Pollard Jw, Stanley Er (1990) Total absence of colony-stimulating factor 1 in the macrophage-deficient osteopetrotic (op/op) mouse. Proc Natl Acad Sci U S A 87:4828–4832
Yamashita M, Otsuka F, Mukai T, Yamanaka R, Otani H, Matsumoto Y, Nakamura E, Takano M, Ke S, Makino H (2010) Simvastatin inhibits osteoclast differentiation induced by bone morphogenetic protein-2 and RANKL through regulating MAPK, AKT and Src signaling. Regul Pept 162:99–108
Acknowledgments
This work was supported by grants from the National Basic Research Program of China (973 Program, 2009CB522200 and 2010CB833802), the Natural Science Foundation of Fujian Province of China (2010 J06014), the Program for New Century Excellent Talents in University of the Ministry of Education (NCET-10-0718), and the Natural Science Foundation of China (30770455 and 31170819).
Conflict of interest
The authors disclose no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhu, J., Chen, Q., Xia, X. et al. Mycoepoxydiene suppresses RANKL-induced osteoclast differentiation and reduces ovariectomy-induced bone loss in mice. Appl Microbiol Biotechnol 97, 767–774 (2013). https://doi.org/10.1007/s00253-012-4146-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4146-5