Abstract
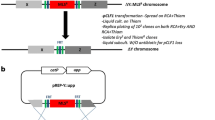
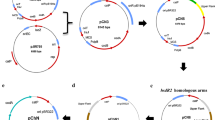
To better understand the DNA restriction-modification (R-M) systems for more amenable strain development of the alternative industrial ethanologen, Zymomonas mobilis, three gene knockout mutants were constructed. The gene knockout mutants were tested for their DNA restriction activities by the determination of transformation efficiency using methylated and unmethylated foreign plasmid DNAs. Inactivation of a putative mrr gene encoded by ZMO0028 (zmrr) resulted in a 60-fold increase in the transformation efficiency when unmethylated plasmid DNA was used. This indicated that the putative mrr gene may serve as a type IV restriction-modification system in Z. mobilis ZM4. To assign the function of a putative type I DNA methyltransferase encoded by ZMO1933 (putative S subunit) and ZMO1934 (putative M subunit), the putative S subunit was inactivated. The gene inactivation of ZMO1933 resulted in a 30-fold increase in the transformation efficiency when methylated plasmid DNA was introduced, indicating that the putative S subunit possibly serves as a part of functional type I R-M system(s). Growth studies performed on the mutant strains indicate inactivation of the type I S subunit resulted in a lower maximum specific glucose consumption rate and biomass yield, while inactivation of the type IV Zmrr had the opposite effect, with an increase in the maximum specific growth rate and biomass yield.






Similar content being viewed by others
References
Bair CL, Black LW (2007) A type IV modification dependent restriction nuclease that targets glucosylated hydroxymethyl cytosine modified DNAs. J Mol Biol 366:768–778
Bickle TA (1987) In: Neidhardt FC (ed) DNA restriction and modification in Escherichia coli and Salmonella typhimurium. ASM Press, Washington, DC, pp 692–696
Bonamy C, Guyonvarch A, Reyes O, David F, Leblon G (1990) Interspecies electro-transformation in Corynebacteria. FEMS Microbiol Lett 66(1–3):263–269
Bujnicki JM, Rychlewski L (2001) Identification of a PD-(D/E)XK-like domain with a novel configuration of the endonuclease active site in the methyl-directed restriction enzyme Mrr and its homologs. Gene 267:183–191
Choi KH, Schweizer HP (2005) An improved method for rapid generation of unmarked Pseudomonas aeruginosa deletion mutants. BMC Microbiol 5(30):110–121
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K12 using PCR products. Proc Natl Acad Sci USA 97(12):6640–6645
Goodman AE, Rogers PL, Skotnicki ML (1982) Minimal medium for isolation of auxotrophic Zymomonas mobilis. Appl Environ Microbiol 44:496–498
Hoang TT, Karkhoff-Schweizer RR, Kutchma AJ, Schweizer HP (1998) A broad-host-range Flp-FRT recombination system for site-specific excision of chromosomally-located DNA sequences: application for isolation of unmarked Pseudomonas aeruginosa mutants. Gene 212:77–86
Janulaitis A, Petrusyte M, Maneliene Z, Klimasauskas S, Butkus V (1992) Purification and properties of the Eco57I restriction endonuclease and methylase: prototypes of a new class (type IV). Nucleic Acids Res 20(22):6043–6049
Jeon YJ, Svenson CJ, Joachimsthal EL, Rogers PL (2002) Kinetic analysis of ethanol production by an acetate-resistant strain of recombinant Zymomonas mobilis. Biotechnol Lett 24:819–824
Jeon YJ, Svenson CJ, Rogers PL (2005) Over-expression of xylulokinase in a xylose-metabolising recombinant strain of Zymomonas mobilis. FEMS Microbiol Lett 244:85–92
Joachimsthal EL, Rogers PL (2000) Characterization of a high productivity recombinant strain of Zymomonas mobilis for ethanol production from glucose/xylose mixture. Appl Biochem Biotechnol 84–86:343–356
Joachimsthal EL, Haggett KD, Rogers PL (1999) Evaluation of recombinant strains of Zymomonas mobilis for ethanol production from glucose/xylose media. Appl Biochem Biotechnol 77(1–3):147–157
Jore JPM, van Luijk N, Luiten RG M, van der Werf MJ, Pouwels PH (2001) Efficient transformation system for Propionibacterium freudenreichii based on a novel vector. Appl Environ Microbiol 67:499–503
Kwak J, Jiang H, Kendrick KE (2002) Transformation using in vivo and in vitro methylation in Streptomyces griseus. FEMS Microbiol Lett 209:243–248
Lefrancois J, Gasc AM, Sicard M (1997) Electrotransformation of Streptococcus pneumoniae: evidence for restriction of the DNA on entry. Microb Drug Resist 3:101–104
Lepikhov K, Tchernov A, Zheleznaja L, Matvienko N, Walter J, Trautner TA (2001) Characterization of the type IV restriction modification system BspLU11III from Bacillus sp. U11. Nucleic Acids Res 29:4691–4698
Liang CC, Lee WC (1998) Characteristics and transformation of Zymomonas mobilis with plasmid pKT230 by electroporation. Bioprocess Eng 19:81–85
Luria SE, Delbruck M (1943) Mutations of bacteria from virus sensitivity to virus resistance. Genetics 28(6):491–511
Marchler-Bauer A, Anderson JB, Chitsaz F, Derbyshire MK, DeWeese-Scott C, Fong JH, Geer LY, Geer RC, Gonzales NR, Gwadz M, He S, Hurwitz DI, Jackson JD, Ke Z, Lanczycki CJ, Liebert CA, Liu C, Lu F, Lu S, Marchler GH, Mullokandov M, Song JS, Tasneem A, Thanki N, Yamashita RA, Zhang D, Zhang N, Bryant SH (2009) CDD: specific functional annotation with the conserved domain database. Nucleic Acids Res 37:D205–D210
Miller JF, Dower WJ, Tompkins LS (1988) High-voltage electroporation of bacterial genetic transformation of Campylobacter jejuni with plasmid DNA. Proc Natl Acad Sci USA 85:856–860
Miller WG, Pearson BM, Wells JM, Parker CT, Kapitonov VV, Mandrell RE (2005) Diversity within the Campylobacter jejuni type I restriction-modification loci. Microbiol 151:337–351
Rogers PL, Jeon YJ, Lee KJ, Lawford HG (2007) Zymomonas mobilis for fuel ethanol and higher value products. Adv Biochem Eng Biotechnol 108:263–288
Schouler C, Clier F, Lerayer AL, Ehrlich SD, Chopin MC (1998) A type IC restriction-modification system in Lactococcus lactis. J Bacteriol 180(2):407–411
Seo JS, Chong HY, Park HS, Yoon KO, Jung C, Kim JJ, Hong JH, Kim H, Kim JH, Kil JI, Park CJ, Oh HM, Lee JS, Jin SJ, Um HW, Lee HJ, Oh SJ, Kim JY, Kang HL, Lee SY, Lee KJ, Kang HS (2005) The genome sequence of the ethanologenic bacterium Zymomonas mobilis ZM4. Nat Biotechnol 23:63–68
Skotnicki ML, Goodman AE, Warr RG, Rogers PL (1984) Isolation and characterization of Zymomonas mobilis plasmids. Microbios 40:53–61
Suh SJ, Silo-Suh LA, Ohman DE (2004) Development of tools for the genetic manipulation of Pseudomonas aeruginosa. J Microbiol Meth 58:203–212
Sutherland E, Coe L, Raleigh EA (1992) McrBC—a multi-subunit GTP dependent restriction endonuclease. J Mol Biol 225(2):327–348
Tock MR, Drydan DTF (2005) The biology of restriction and anti restriction. Curr Opin Microbiol 8:466–472
Vertes AA, Hatakeyama K, Inui M, Kobayashi M, Kurusu Y, Yukawa H (1993) Replacement recombination in Coryneform bacteria—high-efficiency integration requirement for nonmethylated plasmid DNA. Biosci Biotechnol Biochem 57(12):2036–2038
Walder RY, HartleyJL DJE, Walder JA (1981) Cloning and expression of the Pst-I restriction-modification system in Escherichia coli. Proc Natl Acad Sci USA 78(3):1503–1507
Walkinshaw MD, Taylor S, Sturrock SS, Atanasiu C, Berge T, Henderson RM, Edwardson JM, Dryden DTF (2002) Structure of Ocr from bacteriophage T7, a protein that mimics B-form DNA. Mol Cell 9:187–194
Wilson GG, Murray NE (1991) Restriction and modification systems. Annu Rev Genet 25:585–627
Yang S, Pappas KM, Hauser LJ, Land ML, Chen GL, Hurst GB, Pan C, Kouvelis VN, Typas MA, Pelletier DA, Klingeman DM, Chang Y-J, Samatova NF, Brown SD (2009) Improved genome annotation for Zymomonas mobilis. Nat Biotechnol 27:893–894
Yang S, Land ML, Klingeman DM, Pelletier DA, Lu TY, Martin SL, Guo HB, Smith JC, Brown SD (2010) Paradigm for industrial strain improvement identifies sodium acetate tolerance loci in Zymomonas mobilis and Saccharomyces cerevisiae. PNAS 107(23):10395–10400
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kerr, A.L., Jeon, Y.J., Svenson, C.J. et al. DNA restriction-modification systems in the ethanologen, Zymomonas mobilis ZM4. Appl Microbiol Biotechnol 89, 761–769 (2011). https://doi.org/10.1007/s00253-010-2936-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2936-1