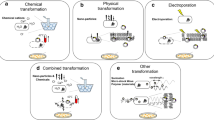
Abstract
The ability to introduce individual molecules of plasmid DNA into cells by transformation has been of central importance to the recent rapid advancement of plasmid biology and to the development of DNA cloning methods. Molecular genetic manipulation of bacteria requires the development of plasmid-mediated transformation systems that include (1) chemical transformation, (2) electro-transformation, (3) biolistic transformation, and (4) sonic transformation, leading to the introduction of exogenous plasmid DNA into bacterial cells. In this review, the manipulation properties and transformation efficiencies of these techniques are described. In addition to these methods, a conceptually novel transformation technique, namely the hydrogel exposure method, was developed. The hydrogel exposure method, based on the Yoshida effect, provides a significant advance over chemical means for transforming many strains of Escherichia coli and a variety of other bacterial species. The new term “tribos transformation” has been proposed for this novel technique. We also determined that, compared to conventional methods, the hydrogel exposure method is a novel and convenient method by which to transform bacteria.

Similar content being viewed by others
References
Anagnostopoulos C, Spizizen J (1961) Requirements for transformation in Bacillus subtilis. J Bacteriol 81:741–746
Antonov PA, Maximova VA, Pancheva RP (1993) Heat shock and osmotically dependent steps by DNA uptake after Escherichia coli electroporation. Biochim Biophys Acta 1216:286–288
Azakami H, Sugino H, Murooka Y (1992) Cloning and nucleotide sequence of a negative regulator gene for Klebsiella aerogenes arylsulfatase synthesis and identification of the gene as folA. J Bacteriol 174:2344–2351
Azakami H, Sugino H, Iwata N, Yokoro N, Yamashita M, Murooka Y (1995) A Klebsiella aerogenes moaEF operon is controlled by the positive MoaR regulator of the monoamine regulon. Gene 164:89–94
Bao S, Thrall BD, Miller DL (1997) Transfection of a reporter plasmid into cultured cells by sonoporation in vitro. Ultrasound Med Biol 23:953–959
Bättig P, Mülemann K (2008) Influence of the spxB gene on competence in Streptococcus pneumoniae. J Bacteriol 190:1184–1189
Chang S, Cohen SN (1979) High frequency transformation of Bacillus subtilis protoplasts by plasmid DNA. Mol Gen Genet 168:111–115
Chassy BM, Flickinger JL (1987) Transformation of Lactobacillus casei by electroporation. FEMS Microbiol Lett 44:173–177
Chung CT, Niemela SL, Miller RH (1987) One-step preparation of competent Escherichia coli: transformation and storage of bacterial cells in the same solution. Proc Natl Acad Sci U S A 86:2172–2175
Cohen SN, Chang AC, Hsu L (1972) Nonchromosomal antibiotic resistance in bacteria: genetic transformation of Escherichia coli by R-factor DNA. Proc Natl Acad Sci U S A 69:2110–2114
de Vos WM, Venema G (1981) Fate of plasmid DNA in transformation of Bacillus subtilis protoplasts. Mol Gen Genet 182:39–43
de Vos WM, Venema G, Canosi U, Trautner TA (1981) Plasmid transformation in Bacillus subtilis: fate of plasmid DNA. Mol Gen Genet 181:424–433
Dower WJ, Miller JF, Ragsdale CW (1988) High efficiency transformation of E. coli by high voltage electroporation. Nucleic Acids Res 16:6127–6145
Ehrlich SD (1977) Replication and expression of plasmids from Staphylococcus aureus in Bacillus subtilis. Proc Natl Acad Sci U S A 74:1680–1682
Elliott AR, Silvert PY, Xue GP, Simpson GD, Tekaia-Elhsissen K, Aylward JH (1999) Transformation of Bacillus subtilis using the particle inflow gun and submicrometer particles obtained by the polyol process. Anal Biochem 269:418–420
Fiedler S, Wirth R (1988) Transformation of bacteria with plasmid DNA by electroporation. Anal Biochem 170:38–44
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Hauser PM, Karamata D (1994) A rapid and simple method for Bacillus subtilis transformation on solid media. Microbiology 140:1613–1617
Huff JP, Grant BJ, Penning CA, Sullivan KF (1990) Optimization of routine transformation of Escherichia coli with plasmid DNA. BioTechniques 9:570–577
Inoue H, Nojima H, Okayama H (1990) High efficiency transformation of Escherichia coli with plasmids. Gene 96:23–28
Ishiwa H, Tsuchida N (1984) New shuttle vectors for Escherichia coli and Bacillus subtilis. I. Construction and characterization of plasmid pHY460 with twelve unique cloning sites. Gene 32:129–134
Klein T, Wolf E, Wu R, Sanford JC (1987) High-velocity microprojectiles for delivering nucleic acids into living cells. Nature 327:70–73
Lederberg EM, Cohen SN (1974) Transformation of Salmonella typhimurium by plasmid deoxyribonucleic acid. J Bacteriol 119:1072–1074
Löblom J, Kronqvist N, Uhlén M, Ståhl S, Wernérus H (2007) Optimization of electroporation-mediated transformation: Staphylococcus carnosus as model organism. J Appl Microbiol 102:736–747
Ludwig A, Heimbucher T, Gregor W, Czerny T, Schmetterer G (2008) Transformation and gene replacement in the facultatively chemoheterotrophic, unicellular cyanobacterium Synechocystis sp. PCC6714 by electroporation. Appl Microbiol Biotechnol 78:729–735
MacNeil DJ (1987) Introduction of plasmid DNA into Streptomyces lividans by electroporation. FEMS Microbiol Lett 42:239–244
Mandel M, Higa A (1970) Calcium-dependent bacteriophage DNA infection. J Mol Biol 53:159–162
Martinez MA, Dezar C, Baigorí M, Siñeriz F (1999) Simple method for plasmid mediated transformation of different Bacillus species. Biotechnol Tech 13:337–340
Neumann E, Schaefer-Ridder M, Wang Y, Hofschneider PH (1982) Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J 1:841–845
Ohse M, Takahashi K, Kadowaki Y, Kusaoke H (1995) Effects of plasmid DNA sizes and several other factors on transformation of Bacillus subtilis ISW1214 with plasmid DNA by electroporation. Biosci Biotechnol Biochem 59:1433–1437
Panja S, Saha S, Jana B, Basu T (2006) Role of membrane potential on artificial transformation of E. coli with plasmid DNA. J Biotechnol 127:14–20
Powell IB, Achen MG, Hillier AJ, Davidson BE (1988) A simple and rapid method for genetic transformation of lactic Streptococci by electroporation. Appl Environ Microbiol 54:655–660
Priest FG (1977) Extracellular enzyme synthesis in the genus Bacillus. Bacteriol Rev 41:711–753
Quinn CP, Dancer BN (1990) Transformation of vegetative cells of Bacillus anthracis with plasmid DNA. J Gen Microbiol 136:1211–1215
Rodríuez MC, Alegre MT, Mesas JM (2007) Optimization of technical conditions for the transformation of Pediococcus acidilactici P60 by electroporation. Plasmid 58:44–50
Sadaie Y, Kada T (1983) Formation of competent Bacillus subtilis cells. J Bacteriol 153:813–821
Shark KB, Smith FD, Harpending PR, Rasmussen JL, Sanford JC (1991) Biolistic transformation of a procaryote, Bacillus megaterium. Appl Environ Microbiol 57:480–485
Sheng Y, Mancino V, Birren B (1995) Transformation of Escherichia coli with large DNA molecules by electroporation. Nucleic Acids Res 23:1990–1996
Shivarova N, Foster W, Jacob HE, Grigorova R (1983) Microbiological implications of electric field effects. VII: stimulation of plasmid transformation of Bacillus cereus protoplasts by electric field pulses. Z Allg Mikrobiol 23:595–599
Silvert PY, Tekaia-Elhsissen K (1995) Synthesis of monodisperse submicronic gold particles by the polyol process. Solid State Ionics 82:53–60
Song Y, Hahn T, Thompson IP, Mason TJ, Preston GM, Li G, Paniwnyk L, Huang WE (2007) Ultrasound-mediated DNA transfer for bacteria. Nucleic Acids Res 35:e129
Takeshita S, Sato M, Toba M, Masahashi W, Hashimoto-Gotoh T (1987) High-copy-number and low-copy-number plasmid vectors for lacZ alpha-complementation and chloramphenicol- or kanamycin-resistance selection. Gene 61:63–74
Turgeon N, Laflamme C, Ho J, Duchaine C (2006) Elaboration of an electroporation protocol for Bacillus cereus ATCC 14579. J Microbiol Methods 67:543–548
Tyurin MV, Desai SG, Lynd LR (2004) Electrotransformation of Clostridium thermocellum. Appl Environ Microbiol 70:883–890
VanWagoner TM, Whitby PW, Morton DJ, Seale TW, Stull TL (2004) Characterization of three new competence-regulated operons in Haemophilus influenzae. J Bacteriol 186:6409–6421
Wong TK, Neumann E (1982) Electric field mediated gene transfer. Biochem Biophys Res Commun 107:584–587
Yoshida N (2007) Discovery and application of the Yoshida effect: nano-sized acicular materials enable penetration of bacterial cells by sliding friction force. Recent Pat Biotechnol 1:194–201
Yoshida N, Saeki Y (2004) Chestnut bur-shaped aggregates of chrysotile particles enable inoculation of Escherichia coli cells with plasmid DNA. Appl Microbiol Biotechnol 65:566–575
Yoshida N, Ikeda T, Yoshida T, Sengoku T, Ogawa K (2001) Chrysotile asbestos fibers mediate transformation of Escherichia coli by exogenous plasmid DNA. FEMS Microbiol Lett 195:133–137
Yoshida N, Kodama K, Nakata K, Yamashita M, Miwa T (2002) Escherichia coli cells penetrated by chrysotile fibers are transformed to antibiotics resistance by incorporation of exogenous plasmid DNA. Appl Microbiol Biotechnol 60:461–468
Yoshida N, Nakajima-Kambe T, Matsuki K, Shigeno T (2007) Novel plasmid transformation method mediated by chrysotile, sliding friction, and elastic body exposure. Anal Chem Insights 2:9–15
Zarnitsyn VG, Prausnitz MR (2004) Physical parameters influencing optimization of ultrasound-mediated DNA transfection. Ultrasound Med Biol 30:527–538
Zimmermann U, Vienken J (1982) Electric field-induced cell-to-cell fusion. J Membr Biol 67:165–182
Zimmermann U, Vienken J, Scheurich P (1980) Electric field induced fusion of biological cells. Eur Biophys J 6:86
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, N., Sato, M. Plasmid uptake by bacteria: a comparison of methods and efficiencies. Appl Microbiol Biotechnol 83, 791–798 (2009). https://doi.org/10.1007/s00253-009-2042-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2042-4