Abstract
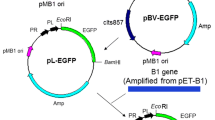
One way to reduce the potential risk of genetically engineered microorganisms (GEMs) to the environment is to use a containment system that does not interfere with the performance of the GEM until activated. Such a system can be created by inserting a suicide cassette consisting of a toxin-encoding gene controlled by an inducible promoter. We constructed a GEM that can degrade organophosphorus compounds, emit green fluorescence, and commit suicide when required by putting the genes that control these different functions under different promoters. The genes for enhanced green fluorescent protein (EGFP) and organophosphorus hydrolase (OPH) were cloned downstream of the lambda PL promoter in the plasmid pBV220. These genes could be expressed freely as long as the GEM was metabolizing because the repressor sequence cIts857 had been deleted. The extracellular nuclease gene of Serratia marcescens, without its leader-coding sequence, provided the suicide mechanism. This was put under the control of the T7 promoter to form a suicide cassette activated by the presence of an environmental signal, in this case, arabinose. To improve the reliability of this containment system, the suicide cassette was duplicated within the conditional suicide plasmid. The plasmid carrying the EGFP and OPH fusion genes and that containing the suicide cassette were compatible and coexisted in the same host.





Similar content being viewed by others
References
Ahrenhotz I, Lorenz MG, Wackernagel W (1994) A conditional suicide system in Escherichia coli based on the intracellular degradation of DNA. Appl Environ Microbiol 60:3746–3751
Balan A, Schenberg ACG (2005) A conditional suicide system for Saccharomyces cerevisiae relying on the intracellular production of the Serratia marcescens nuclease. Yeast 22:203–212
Bej AK, Perlin MH, Atlas RM (1988) Model suicide vector for containment of genetically engineered microorganisms. Appl Environ Microbiol 54:2472–2477
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chae MY, Postula JF, Raushel FM (1994) Stereospecific enzymatic hydrolysis of phosphorus-sulfur bonds in chiral organophosphate triesters. Bioorg Med Chem Lett 4:1473–1478
Cormack BP, Valdivia R, Falkow S (1996) FACS-Optimized mutants of the green fluorescent protein (GFP). Gene 173:371–374
Cranenburgh RM, Hanak JA, Williams SG, Sherratt DJ (2001) Escherichia coli strains that allow antibiotic-free plasmid selection and maintenance by repressor titration. Nucleic Acids Res 29:E26
Cranenburgh RM, Lewis KS, Hanak JA (2004) Effect of plasmid copy number and lac operator sequence on antibiotic-free plasmid selection by operator-repressor titration in Escherichia coli. J Mol Microbiol Biotechnol 7:197–203
Gory L, Montel MC, Zagorec M (2001) Use of green fluorescent protein to monitor Lactobacillus sakei in fermented meat products. FEMS Microbiol Lett 194:127–133
Jensen LB, Ramos JL, Kaneva Z, Molin S (1993) A substrate-dependent biological containment system for Pseudomonas putida based on the Escherichia coli gef gene. Appl Environ Microbiol 59:3713–3717
Kang DG, Choi SS, Cha HJ (2006) Enhanced biodegradation of toxic organophosphate compounds using recombinant Escherichia coli with sec pathway-driven periplasmic secretion of organophosphorus hydrolase. Biotechnol Prog 22:406–410
Khan AA, Jones RA, Cerniglia CE (1998) Rapid method for the detection of genetically engineered microorganisms by polymerase chain reaction from soil and sediments. J Ind Microbiol Biotechnol 20:90–94
Klemm P, Jensen LB, Molin S (1995) A stochastic killing system for biological containment of Escherichia coli. Appl Environ Microbiol 61:481–486
Knudsen S, Saadbye P, Hansen LH, Collier A, Jacobsen BL, Schlundt J, Karlstrom OH (1995) Development and testing of improved suicide functions for biological containment of bacteria. Appl Environ Microbiol 61:985–991
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lai K, Stolowich NJ, Wild JR (1995) Characterization of P–S bond hydrolysis in organophosphorothioate pesticides by organophosphorus hydrolase. Arch Biochem Biophys 318:59–64
Leff LG, Leff AA (1996) Use of green fluorescent protein to monitor survival of genetically engineered bacteria in aquatic environments. Appl Environ Microbiol 62:3486–3488
Lindow SE (1995) The use of reporter genes in the study of microbial ecology. Mol Ecol 4:555–566
March JC, Rao G, Bentley WE (2003) Biotechnological applications of green fluorescent protein. Appl Microbiol Biotechnol 62:303–315
Matheson VG, Munakata-Marr J, Hopkins GD, McCarty PL, Tiedje JM, Forney LJ (1997) A novel means to develop strain-specific DNA probes for detecting bacteria in the environment. Appl Environ Microbiol 63:2863–2869
Munnecke DM (1980) Enzymatic detoxification of waste organophosphate pesticides. J Agric Food Chem 28:105–111
Ramos-Gonzalez MI, Ruiz-Cabello F, Brettar I, Garrido F, Ramos JL (1992) Tracking genetically engineered bacteria: monoclonal antibodies against surface determinants of the soil bacterium Pseudomonas putida 2440. J Bacteriol 174:2978–2985
Ripp S, Nivens DE, Werner C, Sayler GS (2000) Bioluminescent most-probable-number monitoring of a genetically engineered bacterium during a long-term contained field release. Appl Microbiol Biotechnol 53:736–741
Ronchel MC, Ramos JL (2001) Dual system to reinforce biological containment of recombinant bacteria designed for rhizoremediation. Appl Environ Microbiol 67:2649–2656
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Shimomura O, Johnson FH, Saiga Y (1962) Extraction, purification and properties of aequorin, a bioluminescent protein from luminous hydromedusan, Aequorea. J Cell Comp Physiol 59:223–239
Torres B, Jaenecke S, Timmis KN, García JL, Díaz E (2003) A dual lethal system to enhance containment of recombinant micro-organisms. Microbiology 149:3595–3601
Wang AA, Mulchandani A, Chen W (2002) Specific adhesion to cellulose and hydrolysis of organophosphate nerve agents by a genetically engineered Escherichia coli strain with a surface-expressed cellulose-binding domain and organophosphorus hydrolase. Appl Environ Microbiol 68:1684–1689
Wu CF, Cha HJ, Rao G, Valdes JJ, Bentley WE (2000) A green fluorescent protein fusion strategy for monitoring the expression, cellular location, and separation of biologically active organophosphorus hydrolase. Appl Microbiol Biotechnol 54:78–83
You L, Cox RS 3rd, Weiss R, Arnold FH (2004) Programmed population control by cell–cell communication and regulated killing. Nature 428:868–871
Acknowledgments
We thank Prof. W. Wackernagel for providing the E. coli TGE900 containing the plasmid pAH12, Prof. C. L. Qiao for providing the plasmid pGEM-T-opd, and Ms. R. Liu for the plasmid pBV220. This research was supported by the National High Technology R&D Program of China (863 Program, no. 2002AA601160, no. 2006AA06Z423) and The CAS Innovation Program (KSCX3-IOZ-0701).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material
Suppl. Table 1
Primers used in this study (DOC 23 kb)
Rights and permissions
About this article
Cite this article
Li, Q., Wu, YJ. A fluorescent, genetically engineered microorganism that degrades organophosphates and commits suicide when required. Appl Microbiol Biotechnol 82, 749–756 (2009). https://doi.org/10.1007/s00253-009-1857-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-1857-3