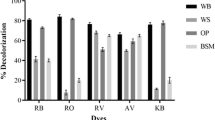
Abstract
In this study, we investigated the efficacy of phenolic extract of wheat bran and lignin-related phenolic compounds as natural redox mediators on laccase-mediated transformation of malachite green (MG) using purified laccase from the white-rot fungus Ganoderma lucidum. G. lucidum laccase was able to decolorize 40.7% MG dye (at 25 mg l−1) after 24 h of incubation. Whereas, the addition of phenolic extract of wheat bran enhanced the decolorization significantly (p < 0.001) by two- to threefold than that of purified laccase alone. Among various natural phenolic compounds, acetovanillone, p-coumaric acid, ferulic acid, syringaldehyde, and vanillin were the most efficient mediators, as effective as the synthetic mediator 1-hydroxybenzotriazole. Characterization of MG transformation products by HPLC, UV–Vis, and liquid chromatography-mass spectrometry-electrospray ionization analysis revealed that N-demethylation was the key mechanism of decolorization of MG by laccase. Growth inhibition test based on mycelial growth inhibition of white rot fungus Phanerochaete chrysosporium revealed that treatment with laccase plus natural mediators effectively reduced the growth inhibitory levels of MG than that of untreated one. Among all the tested compounds, syringaldehyde showed the highest enhanced decolorization, as a consequence reduced growth inhibition was observed in syringaldehyde-treated samples. The results of the present study revealed that the natural phenolic compounds could alternatively be used as potential redox mediators for effective laccase-mediated decolorization of MG.






Similar content being viewed by others
References
Abadulla E, Tzanov T, Costa S, Robra KH, Cavaco-Paulo A, Gubitz GM (2000) Decolorization and detoxification of textile dyes with a laccase from Trametes hirsute. Appl Environ Microbiol 66:3357–3362
Beta T, Nam S, Dexter JE, Sapirstein HD (2005) Phenolic content and antioxidant activity of pearled wheat and roller-milled fractions. Cereal Chem 82:390–393
Camarero S, Ibarra D, Martinez MA, Martinez AT (2005) Lignin-derived compounds as efficient laccase mediators for decolorization of different types of recalcitrant dyes. Appl Environ Microbiol 71:1775–1784
Canas AI, Alcalde M, Plou F, Martiänez MJ, Martiänez AT, Camarero S (2007) Transformation of polycyclic aromatic hydrocarbons by laccase is strongly enhanced by phenolic compounds present in soil. Environ Sci Technol 41:2964–2971
Cha CJ, Doerge DR, Cerniglia CE (2001) Biotransformation of Malachite Green by the Fungus Cunninghamella elegans. Appl Environ Microbiol 67:4358–4360
Chen CC, Lu CS, Chung YC, Jan JL (2007) UV light induced photodegradation of malachite green on TiO2 nanoparticles. J Hazard Mater 141:530–528
Claus H, Faber G, König H (2002) Redox-mediated decolorization of synthetic dyes by fungal laccases. Appl Microbiol Biotechnol 59:672–678
Clemmensen SJ, Jensen JC, Meyer O, Olsen P, Wurtzen G (1984) Toxicological studies on malachite green: a triphenylmethane dye. Arch Toxicol 56:43–45
Culp SJ, Beland FA (1996) Malachite green: a toxicological review. J Am Coll Toxicol 15:219–238
d'Acunzo F, Galli C (2003) First evidence of catalytic mediation by phenolic compounds in the laccase-induced oxidation of lignin models. Eur J Biochem 270:3634–3640
Fernandes C, Lalitha VS, Rao KVK (1991) Enhancing effect of malachite green on the development of hepatic pre-neoplastic lesions induced by N-nitrosodiethylamine in rats. Carcinogenesis 12:839–845
Fernández-Sánchez C, Tzanov T, Gübitz GM, Cavaco-Paulo A (2002) Voltammetric monitoring of laccase-catalysed mediated reactions. Bioelectrochemistry 58:149–156
García O, Camarero S, Colom JF, Martínez ÁT, Martínez MJ, Monje R, Vidal T (2003) Optimization of a laccase-mediator stage for TCF bleaching of flax pulp. Holzforschung 57:513–519
Gupta VK, Mittal A, Krishnan L, Gajbe V (2004) Adsorption kinetics and column operations for the removal and recovery of malachite green from wastewater using ash. Sep Purif Technol 40:87–96
Henderson AL, Schmitt TC, Heinze TM, Cerniglia CE (1997) Reduction of malachite green to leucomalachite green by intestinal bacteria. Appl Environ Microbiol 63:4099–4101
Jadhav JP, Govindwar SP (2006) Biotransformation of malachite green by Saccharomyces cerevisiae MTCC 463. Yeast 23:315–323
Jeon JR, Murugesan K, Kim YM, Kim EU, Chang YS (2008) Synergistic effect of laccase mediators on pentachlorophenol removal by Ganoderma lucidum laccase. Appl Microbiol Biotechnol 81:783–790
Jones JJ, Falkinham JO III (2003) Decolorization of malachite green and crystal violet by waterborne pathogenic Mycobacteria. Antimicrob Agents Chemothe 47:2323–2326
Kim KH, Tsao R, Yang R, Cui SW (2006) Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effect of hydrolysis conditions. Food Chem 95:466–473
Kumar KV, Ramamurthi V, Sivanesan S (2006) Dyes and pigments: biosorption of malachite green a cationic dye onto Pithophora sp., a fresh water algae. Dyes Pigments 69:74–79
Li KC, Helm RF, Eriksson KEL (1998) Mechanistic studies of the oxidation of a non-phenolic lignin model compound by the laccase/1-hydroxybenzotriazole redox system. Biotechnol Appl Biochem 27:239–243
Murugesan K, Arulmani M, Nam IH, Kim YM, Chang YS, Kalaichelvan PT (2006) Purification and characterization of laccase produced by a white rot fungus Pleurotus sajor-caju under submerged culture condition and its potential in decolorization of azo dyes. Appl Microbiol Biotechnol 72:939–946
Murugesan K, Dhamija A, Nam IH, Kim YM, Chang YS (2007a) Decolorization of reactive black 5 by laccase: optimization by response surface methodology. Dyes Pigments 75:176–184
Murugesan K, Nam IH, Kim YM, Chang YS (2007b) Decolorization of reactive dyes by a thermostable laccase produced by Ganoderma lucidum in solid state culture. Enzy Microbial Technol 40:1662–1672
Panadiker A, Fernandes C, Rao KVK (1992) The cytotoxic properties of malachite green are associated with the increased demethylase, aryl hydrocarbon hydroxylase and lipid peroxidation in primary cultures of Syrian hamster embryo cells. Cancer Lett 67:93–101
Papinutti VL, Forchiassin F (2004) Modification of malachite green by Fomes sclerodermeus and reduction of toxicity to Phanerochaete chrysosporium. FEMS Microbiol Lett 231:205–209
Parshetti G, Kalme S, Saratale G, Govindwar S (2006) Biodegradation of Malachite Green by Kocuria rosea MTCC 1532. Acta Chim Slov 53:492–498
Pointing SB (2001) Feasibility of bioremediation by white-rot fungi. Appl Microbiol Biotechnol 57:20–33
Pointing SB, Vrijmoed LLP (2000) Decolorization of azo and triphenylmethane dyes by Pycnoporus sanguineous producing laccase as the sole phenol oxidase. World J Microbiol Biotechnol 16:317–318
Reddy CA (1995) The potential for white rot fungi in the treatments of pollutants. Curr Opin Biotechnol 6:320–328
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Meth Enzymol 299:152–178 Article| PDF (1500K)
Vasdev K, Kuhad RC, Saxena RK (1995) Decolorization of triphenylmethane dyes by the bird’s nest fungus Cyathus bulleri. Curr Microbiol 30:269–272
Wang J, Sun B, Cao Y, Tian Y, Li X (2008) Optimization of ultrasound-assisted extraction of phenolic compounds from wheat bran. Food Chem 106:804–810
Wolfenden BS, Wilson RL (1982) Radical cations as reference chromogens in studies of one-electron transfer reactions: pulse radio analysis studies of 2,2’azinobis-(3-ethlbenzthiazoline-b-ulfonate). J Chem Soc Perkin Trans II:805–812
Yuzhu F, Viraraghavan T (2001) Fungal decolorization of dye wastewaters: a review. Bioresource Technol 79:251–262
Acknowledgments
This work was supported by the Ministry of the Environment of the Republic of Korea as “The GAIA Project” and the Brain Korea 21 project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murugesan, K., Yang, IH., Kim, YM. et al. Enhanced transformation of malachite green by laccase of Ganoderma lucidum in the presence of natural phenolic compounds. Appl Microbiol Biotechnol 82, 341–350 (2009). https://doi.org/10.1007/s00253-008-1819-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1819-1