Abstract
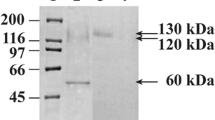
The cell wall of Candida albicans is composed of mannoproteins associated to glycan polymers. Most of these proteins are retained in this compartment through a phosphodiester linkage between a remnant of their glycosylphosphatidylinositol anchor and the β-1,6-glucan polymer. A pure β-1,6-glucanase is thus required in order to release them. In this paper, we report the expression/secretion by the yeast Yarrowia lipolytica of an Aspergillus fumigatus enzyme homologous to previously described β-1,6-glucanases. The coding sequence was expressed under the control of a strong promoter and the recombinant enzyme was targeted to the secretory pathway using the signal sequence of a well-known major secretory protein in this host. Addition of a FLAG epitope at the C-terminus allowed its efficient purification from culture supernatant following batch adsorption. The purified enzyme was characterized as a β-1,6-glucanase and was shown to be active on C. albicans cell walls allowing the release of a previously described cell wall protein.




Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bom IJ, Dielbandhoesing SK, Harvey KN, Oomes SJCM, Klis FM, Brul S (1998) A new tool for studying the molecular architecture of the fungal cell wall: one-step purification of recombinant Trichoderma β-(1-6)-glucanase expressed in Pichia pastoris. BBA 1425:419–424
de Groot PW, de Boer AD, Cunningham J, Dekker HL, de Jong L, Hellingwerf KJ, de Koster C, Klis FM (2004) Proteomic analysis of Candida albicans cell walls reveals covalently bound carbohydrate-active enzymes and adhesins. Eukaryot Cell 3:955–965
De la Cruz J, Llobell A (1999) Purification and properties of a basic endo-b-1,6-glucanase (BGN16.1) from the antagonistic fungus Trichoderma harzianum. Eur J Biochem 265:145–151
De la Cruz J, Pintor-Toro JA, Benítez T, Llobell A (1995) Purification and characterization of an endo-β-1,6-glucanase from Trichoderma harzianum that is related to its mycoparasitism. J Bacteriol 177:1864–1871
Djonovic S, Pozo MJ, Kenerley CM (2006) Tvbgn3, a β-1,6-glucanase from the biocontrol fungus Trichoderma virens, is involved in mycoparasitism and control of Pythium ultimum. Appl Environ Microbiol 72:7661–7670
Fontaine T, Simenel C, Dubreucq G, Adam O, Delepierre M, Lemoine J, Vorgias CE, Diaquin M, Latgé JP (2000) Molecular organization of the alkali-insoluble fraction of Aspergillus fumigatus cell wall. J Biol Chem 275:27594–27607
Fonzi WA, Irwin MY (1993) Isogenic strain construction and gene mapping in Candida albicans. Genetics 134:717–728
Kapteyn JC, Montijn RC, Vink E, de la Cruz J, Llobell A, Douwes JE, Shimoi H, Lipke PN, Klis FM (1996) Retention of Saccharomyces cerevisiae cell wall proteins through a phosphodiester-linked β-1,3-/β-1,6-glucan heteropolymer. Glycobiology 6:337–345
Kollár R, Reinhold BB, Petráková E, Yeh HJ, Ashwell G, Drgonová J, Kapteyn JC, Klis FM Cabib E (1997) Architecture of the yeast cell wall. β(1→6)-glucan interconnects mannoprotein, β(1→)3-glucan, and chitin. J Biol Chem 272:17762–17775
Liu TT, Lee RE, Barker KS, Lee RE, Wei L, Homayouni R, Rogers PD (2005) Genome-wide expression profiling of the response to azole, polyene, echinocandin, and pyrimidine antifungal agents in Candida albicans. Antimicrob Agents Chemother 49:2226–2236
Lora JM, de la Cruz J, Llobell A, Benitez T, Pintor-Toro JA (1995) Molecular characterization and heterologous expression of an endo-β-1,6-glucanase gene from the mycoparasitic fungus Trichoderma harzianum. Mol Gen Genet 247:639–645
Madzak C, Tréton B, Blanchin-Roland S (2000) Strong hybrid promoters and integrative expression/secretion vectors for quasi-constitutive expression of heterologous proteins in the yeast Yarrowia lipolytica. J Mol Microbiol Biotechnol 2:207–216
Madzak C, Gaillardin C, Beckerich JM (2004) Heterologous protein expression and secretion in the non-conventional yeast Yarrowia lipolytica: a review. J Biotechnol 109:63–81
Madzak C, Otterbein L, Chamkha M, Moukha S, Asther M, Gaillardin C, Beckerich JM (2005) Heterologous production of a laccase from the basidiomycete Pycnoporus cinnabarinus in the dimorphic yeast Yarrowia lipolytica. FEMS Yeast Res 6–7:635–646
Montero M, Sanz L, Rey M, Monte E, Llobell A (2005) BGN16.3, a novel acidic β-1,6-glucanase from mycoparasitic fungus Trichoderma harzianum CECT 2413. FEBS J 272:3441–3448
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng 10:1–6
Oyama S, Yamagata Y, Abe K, Nakajima T (2002) Cloning and expression of an endo-1,6-β-D-glucanase gene (neg1) from Neurospora crassa. Biosci Biotechnol Biochem 66:1378–1381
Richard ML, Plaine A (2007) Comprehensive analysis of glycosylphosphatidylinositol-anchored proteins in Candida albicans. Euk Cell 6:119–133
Ruiz-Herrera JM, Elorza V, Valentín E, Sentandreu R (2006) Molecular organization of the cell wall of Candida albicans and its relation to pathogenicity. FEMS Yeast Res 6:14–29
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boisramé, A., Gaillardin, C. Heterologous expression and characterization of a β-1,6-glucanase from Aspergillus fumigatus . Appl Microbiol Biotechnol 82, 663–669 (2009). https://doi.org/10.1007/s00253-008-1780-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1780-z