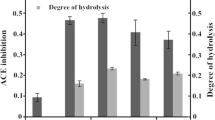
Abstract
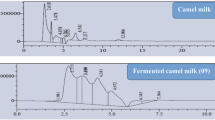
Acetes chinensis is an underutilized shrimp species thriving in Bo Hai Gulf of China. Its hydrolysate digested with protease SM98011 has been previously shown to have high angiotensin I-converting enzyme (ACE) inhibitory activity (He et al., J Pept Sci 12:726–733, 2006). In this article, A. chinensis were fermented by Lactobacillus fermentum SM 605 and the fermented sauce presented high ACE inhibitory activity. The minimum IC50 value (3.37 ± 0.04 mg/mL) was achieved by response surface methodology with optimized process parameters such as fermentation time of 24.19 h, incubation temperature at 38.10°C, and pH 6.12. Three ACE inhibitory peptides are purified by ultrafiltration, gel filtration, and reverse-phase high performance liquid chromatography. Identified by mass spectrometry, their amino acid sequences are Asp-Pro, Gly-Thr-Gly, and Ser-Thr, with IC50 values of 2.15 ± 0.02, 5.54 ± 0.09, and 4.03 ± 0.10 μM, respectively. Also, they are all novel ACE inhibitory peptides. Compared with protease digestion, fermentation is a simpler and cheaper method to produce ACE inhibitory peptides from shrimp A. chinensis.




Similar content being viewed by others
References
Abubakar A, Saito T, Kitazawa H, Kawai Y, Itoch T (1998) Structural analysis of new antihypertensive peptides derived from cheese whey protein by proteinase K digestion. J Dairy Sci 81:3131–3138
Box GEP, Hunter WG, Hunter JS (1978) Statistics for experimenters. Wiley, NY
Boyd PM, Barnaby N, Tan-Wilson A, Wilson KA (2002) Cleavage specificity of the subtilisin-like protease C1 from soybean. Biochim Biophys Acta 1596:269–282
Chun H, Sasaki M, Fujiyama Y, Bamba T (1996) Effect of peptide chain length on absorption and intact transport of hydrolyzed soybean peptide in rat intestinal everted sac. J Clin Biochem Nutr 21:131–140
Craft IL, Geddes D, Hyde CW, Wise IJ, Matthews DM (1968) Absorption and malabsorption of glycine and glycine peptides in man. J Gut 9:425–437
Dziuba J, Mimkiewicz P, Plitnik K (1996) Chicken meat proteins as potential precursors of bioactive peptides. Pol J Food Nutr Sci 4:85–96
FitzGerald RJ, Meisel H (2000) Milk protein-derived peptide inhibitors of angiotensin-I-converting enzyme. Br J Nutr 84:S33–S37
Gobbetti M, Ferranti P, Smacchi E, Goffredi F, Addeo F (2000) Production of angiotensin-I-converting-enzyme-inhibitory peptides in fermented milks started by Lactobacillus delbrueckii subsp. bulgaricus SS1 and Lactococcus lactis subsp. cremoris FT4. Appl Environ Microbiol 66:3898–3904
Gobbetti M, Stepaniak L, Angelis MD, Corsetti A, Cagno RD (2002) Latent bioactive peptides in milk proteins: proteolytic activation and significance in dairy processing. Crit Rev Food Sci Nutr 42:223–239
Harun-ur-Rashid M, Togo K, Ueda M, Miyamoto T (2007) Probiotic characteristics of lactic acid bacteria isolated from traditional fermented milk ‘Dahi’ in Bangladesh. Pakistan J Nutr 6:647–652
He HL, Chen XL, Sun CY, Zhang YZ, Zhou BC (2006) Analysis of novel angiotensin-I-converting enzyme inhibitory peptides from protease-hydrolyzed marine shrimp Acetes chinensis. J Pept Sci 12:726–733
Israili ZH, Hall WD (1992) Cough and angioneurotic edema associated with angiotensin-converting enzyme inhibitor therapy. A review of the literature and pathophysiology. Ann Intern Med 117:234–242
Je JY, Park PJ, Byun HG, Jung WK, Kim SK (2005) Angiotensin I converting enzyme (ACE) inhibitory peptide derived from the sauce of fermented blue mussel, Mytilus edulis. Bioresour Technol 96:1624–1629
Kempka AP, Lipke NL, da Luz Fontoura Pinheiro T, Menoncin S, Treichel H, Freire DM, Di Luccio M, de Oliveira D (2008) Response surface method to optimize the production and characterization of lipase from Penicillium verrucosum in solid-state fermentation. Bioprocess Biosyst Eng 31:119–125
Khuri AI, Cornell JA (1987) Response surfaces design and analysis. Dekker, NY
Kinoshita E, Yamakoshi J, Ikuchi M (1993) Purification and identification of an angiotensin I-converting enzyme inhibitor from soy sauce. Biosci Biotechnol Biochem 57:1107–1110
Kohumura M, Nio N, Kudo K, Minoshima Y, Munekata E, Ariyoshi Y (1989) Inhibition of angiotensin-converting enzyme by synthetic peptides of human b-casein. Agric Biol Chem 53:2107–2114
Kunji ERS, Mierau I, Hagting A, Poolman B, Konings N (1996) The proteolytic system of lactic acid bacteria. Antonie Van Leeuwenhoek 70:187–221
Maeno M, Yamamoto N, Takano T (1996) Identification of an antihypertensive peptide from casein hydrolysate produced by a proteinase from Lactobacillus helveticus CP790. J Dairy Sci 79:1316–1321
Maruyama S, Mitachi H, Awaya J, Kurono M, Tomizuka N, Suzuki H (1989) Angiotensin I-converting enzyme inhibitory activity of the C-terminal hexapeptide of as1-casein. Agric Biol Chem 53:2107–2114
Matsui T, Matsufuji H, Seki E, Osajima K, Nakashima M, Osajima Y (1993) Inhibition of angiotensin-I converting enzyme by Bacillus licheniformis alkaline protease hydrolysates derived from sardine muscle. Biosci Biotechnol Biochem 57:922–925
Matthews DM, Payne JW (1980) Transmembrane transport of small peptides. J Curr Top Membr Transp 14:331–425
Meisel H, Gunther S, Martin D, Schlimme E (1998) Apoptosis induced by modified ribonucleosides in human cell culture systems. FEBS Lett 433:265–268
Minervini F, Algaron F, Rizzello CG, Fox PF, Monnet V, Gobbetti M (2003) Angiotensin I-converting-enzyme-inhibitory and antibacterial peptides from Lactobacillus helveticus PR4 proteinase-hydrolyzed caseins of milk from six species. Appl Environ Microbiol 69:5297–5305
Miyoshi S, Ishikawa H, Kaneko T, Fukui F, Tanaka H, Maruyama S (1991) Structure and activity of angiotensin converting enzyme inhibitors in an a-zein hydrolysate. Agric Biol Chem 55:1313–1318
Nakamura Y, Yamamoto N, Sakai K, Okubo A, Yamazaki S, Takano T (1995a) Purification and characterization of angiotensin I-converting enzyme inhibitors from sour milk. J Dairy Sci 78:777–783
Nakamura Y, Yamamoto N, Sakai K (1995b) Antihypertensive effect of sour milk and peptides isolated from it that are inhibitors to angiotensin I-converting enzyme. J Dairy Sci 78:1253–1257
Narva M, Halleen J, Väänänen K, Korpela R (2004) Effects of Lactobacillus helveticus fermented milk on bone cells in vitro. Life Sci 75:1727–1734
Okamoto A, Hanagata H, Matsumoto E, Kawamura Y, Koizumi Y, Yanagida F (1995) Angiotensin I-converting enzyme inhibitory activities of various fermented foods. Biosci Biotechnol Biochem 59:1147–1149
Sipola M, Finckenberg P, Korpela R, Vapaatalo H, Nurminen ML (2002) Effect of long-term intake of milk products on blood pressure in hypertensive rats. J Dairy Res 69:103–111
Vercruysse L, Van Camp J, Smagghe G (2005) ACE inhibitory peptides derived from enzymatic hydrolysates of animal muscle protein: a review. J Agric Food Chem 53:8106–8115
Yamamoto N, Maeno M, Takano T (1999) Purification and characterization of an antihypertensive peptide from a yogurt-like product fermented by Lactobacillus helveticus CPN4. J Dairy Sci 82:1388–1393
Yano S, Suzuki K, Funatsu G (1996) Isolation from a-zein of thermolysin peptides with angiotensin I-converting enzyme inhibitory activity. Biosci Biotechnol Biochem 60:661–663
Acknowledgment
This work was supported by grants from the National High Technology Research and Development Program of China (Nos. 2001AA625040 and 2003AA625040).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the Electronic supplementary materials.
MOESM1
(DOC 94.5 kb)
Rights and permissions
About this article
Cite this article
Wang, YK., He, HL., Chen, XL. et al. Production of novel angiotensin I-converting enzyme inhibitory peptides by fermentation of marine shrimp Acetes chinensis with Lactobacillus fermentum SM 605. Appl Microbiol Biotechnol 79, 785–791 (2008). https://doi.org/10.1007/s00253-008-1489-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1489-z