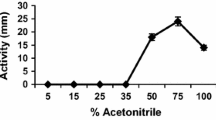
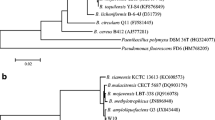
Abstract
Bacillus subtilis strain IB exhibiting inhibitory activity against the Fusarium head blight disease fungus Fusarium graminearum was isolated and identified. The major inhibitory compound was purified from the culture broth through anion exchange, hydrophobic interaction, and reverse phase high-performance liquid chromatography (RP-HPLC) steps. It was a 1,463-Da lipopeptide and had an amino acid composition consisting of Ala, Glx, Ile, Orn, Pro, Thr, and Tyr at a molar ratio of 1:3:1:1:1:1:2. Electrospray ionization mass spectrometry/mass spectrometry (ESI MS/MS) analyses of the natural and the ring-opened peptides showed the antagonist was fengycin, a kind of macrolactone molecule with antifungal activity produced by several Bacillus strains. Fluorescence microscopic analysis indicated this peptide permeabilized and disrupted F. graminearum hyphae.






Similar content being viewed by others
References
Ahn CY, Joung SH, Jeon JW, Kim HS, Yoon BD, Oh HM (2003) Selective control of cyanobacteria by surfactin-containing culture broth of Bacillus subtilis C1. Biotechnol Lett 25:1137–1142
Argyris J, Sanford DV, TeKrony D (2003) Fusarium graminearum infection during wheat seed development and its effect on seed quality. Crop Sci 43:1782–1788
Bais HP, Fall R, Vivanco JM (2004) Biocontrol of Bacillus subtilis against infection of Arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant Physiol 134:307–319
Besson F, Michel G (1987) Isolation and characterization of new iturins: iturin D and E. J Antibiot 40:437–442
Biemann K (1995) The coming of age of mass spectrometry in peptide and protein chemistry. Protein Sci 4:1920–1927
Buchanan RE, Gibbens NE (1984) Bergey’s manual of determinative bacteriology. Science Press, Beijing
Bujold I, Paulitz TC, Carisse O (2001) Effect of Microsphaeropsis sp. on the production of perithecia and ascospores of Gibberella zeae. Plant Dis 85:977–984
Chitarra GS, Breeuwer P, Nout MJ, van Aelst AC, Rombousts FM, Abee T (2003) An antifungal compound produced by Bacillus subtilis YM 10-20 inhibits germination of Penicillium roqueforti conidiospores. J Appl Microbiol 94:159–166
Cho SJ, Lee SK, Cha BJ, Kim YH, Shin KS (2003) Detection and characterization of the Gloeosporium gloeosporioides growth inhibitory compound iturin A from Bacillus subtilis strain KS03. FEMS Microbiol Lett 223:47–51
Eckart K (1994) Mass spectrometry of cyclic peptides. Mass Spectrom Rev 13:23–55
Fernandez MR (1992) The effect of Trichoderma harzianum on fungal pathogens infesting wheat and black oat straw. Soil Biol Biochem 24:1031–1034
Goswami R, Kistler H (2004) Heading for disaster: Fusarium graminearum on cereal crops. Mol Plant Pathol 5:515–525
Han QM, Cao LH (2003) Progress of the biological control on Fusarium graminearum Schw. of wheat. Journal of Triticeae Crops 23:128–131
Hu LB, Shi ZQ, Zhang T, Yang ZM (2007) Fengycin antibiotics isolated from B-FS01 culture inhibit the growth of Fusarium moniliforme Sheldon ATCC 38932. FEMS Microbiol Lett DOI https://doi.org/10.1111/j.1574-6968.2007.00743.x
Jitendra DD, Ibrahim MB (1997) Microbial production of surfactants and their commercial potential. Microbiol Mol Biol Rev 61:47–64
Khan NI, Schisler DA, Boehm MJ, Lipps PE, Slininger PJ (2004) Field testing of antagonists of Fusarium head blight incited by Gibberella zeae. Biol Control 29:245–255
Kim PI, Bai H, Bai D, Chae H, Chung S, Kim Y, Park R, Chi YT (2004) Purification and characterization of a lipopeptide produced by Bacillus thuringiensis CMB26. J Appl Microbiol 97:942–949
Klich MA, Arthur KS, Lax AR, Bland JM (1994) Iturin A: a potential new fungicide for stored grains. Mycopathology 127:123–127
Laemmli U (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Loeffler W, Tschen J, Vanittanakom N, Kugler M, Knorpp E, Hsieh TF, Wu TG (1986) Antifungal effects of bacilysin and fengycin from Bacillus subtilis F-29-3: a comparison with activity of other Bacillus antibiotics. J Phytopathol 115:204–213
Nishikiori T, Naganawa H, Muraoka Y, Aoyagi T, Umezawa H (1986) Plipastatins: new inhibitors of phospholipase A2, produced by Bacillus cereus BMG302-fF67. III. Structural elucidation of plipastatins. J Antibiot 39:755–761
Regine MD, Peypoux F (1994) Iturins, a special class of pore-forming lipopeptides: biological and physicochemical properties. Toxicology 87:151–174
Roepstorff P, Fohlman J (1984) Proposal for a common nomenclature for sequence ions in mass spectra of peptides. Biome Mass Spectrom 11:601
Stein T (2005) Bacillus subtilis antibiotics: structures, syntheses and specific functions. Mol Microbiol 56:845–857
Toure Y, Ongena M, Jacques P, Guiro A, Thonart P (2004) Role of lipopeptides produced by Bacillus subtilis GA1 in the reduction of grey mould disease caused by Botrytis cinerea on apple. J Appl Microbiol 96:1151–1160
Vanittanakom N, Loeffler W (1986) Fengycin—a novel antifungal lipopeptide antibiotics produced by Bacillus subtilis F-29-3. J Antibiot 39:888–901
Vater J, Kablitz B, Wilde C, Franke P, Mehta N, Cameotra SS (2002) Matrix-assisted laser desorption ionization-time of flight mass spectrometry of lipopeptide biosurfactants in whole cells and culture filtrates of Bacillus subtilis C-1 isolate from petroleum sludge. Appl Environ Microbiol 68:6210–6219
Williams BH, Hathout Y, Fenselau C (2002) Structural characterization of lipopeptide biomarkers isolated from Bacillus globigii. J Mass Spectrom 37:259–264
Yu GY, Sinclair JB, Hartman GL, Bertagnolli BL (2002) Production of iturin A by Bacillus amyloliquefaciens suppressing Rhizoctonia solani. Soil Biol Biochem 34:955–963
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, J., Liu, J., Chen, H. et al. Characterization of Fusarium graminearum inhibitory lipopeptide from Bacillus subtilis IB. Appl Microbiol Biotechnol 76, 889–894 (2007). https://doi.org/10.1007/s00253-007-1054-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-1054-1