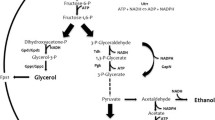
Abstract
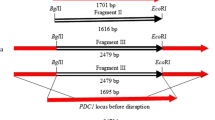
We constructed two recombinant strains of Saccharomyces cerevisiae in which the GPD2 gene was deleted using a one-step gene replacement method to minimize formation of glycerol and improve ethanol production. In addition, we also over-expressed the GLT1 gene by a two-step gene replacement method to overcome the redox-imbalancing problem in the genetically modified strains. The result of anaerobic batch fermentations showed that the rate of growth and glucose consumption of the KAM-5 (MATα ura3 gpd2Δ::RPT) strain were slower than the original strain, and the KAM-13 (MATα ura3 gpd2Δ::RPT P PGK-GLT1) strain, however, was indistinguishable compared to the original strain using the same criteria, as analyzed. On the other hand, when compared to the original strain, there were 32 and 38% reduction in glycerol formation for KAM-5 and KAM-13, respectively. Ethanol production increased by 8.6% for KAM-5 and 13.4% for KAM-13. Dramatic reduction in acetate and pyruvic acid was also observed in both mutants compared to the original strains. Although gene GPD2 is responsible for the glycerol synthesis, the mutant KAM-13, in which glycerol formation was substantially reduced, was able to cope and maintain osmoregulation and redox balance and have increased ethanol production under anaerobic fermentations. The result verified the proposed concept of increasing ethanol production in S. cerevisiae by genetic engineering of glycerol synthesis and over-expressing the GLT1 gene along with reconstituted nicotinamide adenine dinucleotide metabolism.



Similar content being viewed by others
References
Bakker BM, Overkamp KM, Van Maris AJ, Kötter P, Luttik MA, van Dijken JP, Pronk JT (2001) Stoichiometry and compartmentation of NADH metabolism in Saccharomyces cerevisiae. FEMS Microbiol Rev 25:15–37
Björkqvist S, Ansell R, Adler L, Lidén G (1997) Physiological response to anaerobicity of glycerol-3-phosphate dehydrogenase mutants of Saccharomyces cerevisiae. Appl Environ Microbiol 63:128–132
Burke D, Dawson D, Stearns T (2000) Methods in yeast genetics. A cold spring harbor laboratory course manual 174. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press
De Vries S, van Witzenburg R, Grivell LA, Marres CAM (1992) Primary structure and import pathway of the rotenone-insensitive NADH-ubiquinone oxidoreductase of mitochondria from Saccharomyces cerevisiae. Eur J Biochem 203:587–592
Hohmann S (2002) Osmotic stress signaling and osmoadaptation in yeasts. Microbiol Mol Biol Rev 66:300–372
Kong QX, Gu JG, Cao LM, Zhang AL, Chen X, Zhao XM (2006) Improved production of ethanol by deleting FPS1 and over-expressing GLT1 in Saccharomyces cerevisiae. Biotechnol Lett 28:2033–2038
Kong QX, Cao LM, Zhang AL, Chen X (2007) Overexpressing GLT1 in gpd1Δ mutant to improve the production of ethanol of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 73:1382–1386
Michnick S, Roustan JL, Remize F, Barre P (1997) Modulation of glycerol and ethanol yields during alcoholic fermentation in Saccharomyces cerevisiae strains overexpressed or disrupted for GPD1 encoding glycerol 3-phosphate dehydrogenase. Yeast 13:783–793
Nissen TL, Schulze U, Nielsen J, Villandsen J (1997) Flux distributions in anaerobic, glucose-limited continuous cultures of Saccharomyces cerevisiae. Microbiol 143:203–218
Nissen TL, Kielland-Brandt MC, Nielsen J, Villadsen J (2000) Optimization of ethanol production in Saccharomyces cerevisiae by metabolic engineering of the ammonium assimilation. Metab Eng 2:69–77
Norbeck J, Pahlman AK, Akhtar N, Blomberg A, Adler L (1996) Purification and characterization of two isoenzymes of DL-glycerol-3-phosphatase from Saccharomyces cerevisiae. J Biol Chem 271:13875–13881
Oner ET, Oliver SG, Kirdar B (2005) Production of ethanol from starch by respiration-deficient recombinant Saccharomyces cerevisiae. Appl Environ Microbiol 71:6443–6445
Pahlman AK, Granath K, Ansell R, Hohmann S, Adler L (2001) The yeast glycerol 3-phosphatases Gpp1p and Gpp2p are required for glycerol biosynthesis and differentially involved in the cellular responses to osmotic, anaerobic and oxidative stress. J Biol Chem 276:3555–3563
Remize F, Roustan JL, Sablayrolles JM, Barre P, Dequin S (1999) Glycerol overproduction by engineered Saccharomyces cerevisiae wine yeast strains leads to substantial changes in by-product formation and to a stimulation of fermentation rate in stationary phase. Appl Environ Microbiol 65:143–149
Remize F, Barnavon L, Dequin S (2001) Glycerol export and glycerol-3-phosphate dehydrogenase, but not glycerol phosphatase, are rate limiting for glycerol production in Saccharomyces cerevisiae. Metab Eng 3:301–312
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning-a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Torben L, Nissen TL, Morten C, KiellandBrandt, Nielsen J (2000) Optimization of ethanol production in Saccharomyces cerevisiae by metabolic engineering of the ammonium assimilation. Metab Eng 2:69–77
Valadi H, Larsson C, Gustafsson L (1998) Improved ethanol production by glycerol-3-phosphate dehydrogenase mutants of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 4:434–439
Verduyn C, Stouthamer AH, Scheffers WA, van Dijken JP (1991) A theoretical evaluation of growth yields of yeasts. Antonie Van Leeuwenhoek 59:49–63
Acknowledgements
The authors would like to thank Prof. Pingsheng, Ma for his valuable instruction throughout this study. This work was financially supported by the National High-Tech R&D Program (2002AA647040).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kong, QX., Zhang, AL., Cao, LM. et al. Over-expressing GLT1 in a gpd2Δ mutant of Saccharomyces cerevisiae to improve ethanol production. Appl Microbiol Biotechnol 75, 1361–1366 (2007). https://doi.org/10.1007/s00253-007-0948-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-0948-2