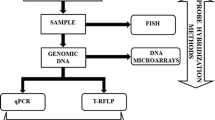
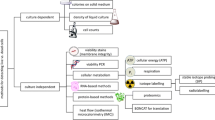
Abstract
Traditional cultivation-based methods to quantify microbial abundance are not suitable for analyses of microbial communities in environmental or medical samples, which consist mainly of uncultured microorganisms. Recently, different cultivation-independent quantification approaches have been developed to overcome this problem. Some of these techniques use specific fluorescence markers, for example ribosomal ribonucleic acid targeted oligonucleotide probes, to label the respective target organisms. Subsequently, the detected cells are visualized by fluorescence microscopy and are quantified by direct visual cell counting or by digital image analysis. This article provides an overview of these methods and some of their applications with emphasis on (semi-)automated image analysis solutions.

Similar content being viewed by others
References
Alfreider A et al (1996) Community analysis of the bacterial assemblages in the winter cover and pelagic layers of a high mountain lake by in situ hybridization. Appl Environ Microbiol 62:2138–2144
Amann RI, Ludwig W, Schleifer K-H (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Amann R, Snaidr J, Wagner M, Ledwig W, Schleifer KH (1996) In situ visualization of high genetic diversity in a natural microbial community. J Bacteriol 178: 3496–3500
Banadda EN, Smets IY, Jenne R, Van Impe JF (2005) Predicting the onset of filamentous bulking in biological wastewater treatment systems by exploiting image analysis information. Bioprocess Biosyst Eng 27:339–348
Belser LW (1979) Population ecology of nitrifying bacteria. Annu Rev Microbiol 33:309–333
Beyenal H, Donovan C, Lewandowski Z, Harkin G (2004) Three-dimensional biofilm structure quantification. J Microbiol Methods 59:395–413
Binder BJ, Liu YC (1998) Growth rate regulation of rRNA content of a marine Synechococcus (cyanobacterium) strain. Appl Environ Microbiol 64:3346–3351
Bjerrum L, Kjaer T, Ramsing NB (2002) Enumerating ammonia-oxidizing bacteria in environmental samples using competitive PCR. J Microbiol Methods 51:227–239
Blackburn N, Hagström A, Wikner J, Cuadros-Hansson R, Bjornsen PK (1998) Rapid determination of bacterial abundance, biovolume, morphology, and growth by neural network-based image analysis. Appl Environ Microbiol 64:3246–3255
Bloem J, Veninga M, Shepherd J (1995) Fully automatic determination of soil bacterium numbers, cell volumes, and frequencies of dividing cells by confocal laser scanning microscopy and image analysis. Appl Environ Microbiol 61:926–936
Boon N, Top EM, Verstraete W, Siciliano SD (2003) Bioaugmentation as a tool to protect the structure and function of an activated-sludge microbial community against a 3-chloroaniline shock load. Appl Environ Microbiol 69:1511–1520
Bouchez T et al (2000) Ecological study of a bioaugmentation failure. Environ Microbiol 2:179–190
Chi F, Shen SH, Cheng HP, Jing YX, Yanni YG, Dazzo FB (2005) Ascending migration of endophytic rhizobia, from roots to leaves, inside rice plants and assessment of benefits to rice growth physiology. Appl Environ Microbiol 71:7271–7278
Contreras EM, Giannuzzi L, Zaritzky NE (2004) Use of image analysis in the study of competition between filamentous and non-filamentous bacteria. Water Res 38:2621–2630
Coskuner G et al (2005) Agreement between theory and measurement in quantification of ammonia-oxidizing bacteria. Appl Environ Microbiol 71:6325–6334
Costerton JW et al (1987) Bacterial biofilms in nature and disease. Annu Rev Microbiol 41:435–464
Cottrell MT, Kirchman DL (2000) Community composition of marine bacterioplankton determined by 16S rRNA gene clone libraries and fluorescence in situ hybridization. Appl Environ Microbiol 66:5116–5122
da Motta M, Pons MN, Roche N (2003) Monitoring filamentous bulking in activated sludge systems fed by synthetic or municipal wastewater. Bioprocess Biosyst Eng 25:387–393
Daims H, Brühl A, Amann R, Schleifer K-H, Wagner M (1999) The domain-specific probe EUB338 is insufficient for the detection of all Bacteria: Development and evaluation of a more comprehensive probe set. Syst Appl Microbiol 22:434–444
Daims H, Nielsen JL, Nielsen PH, Schleifer KH, Wagner M (2001a) In situ characterization of Nitrospira-like nitrite-oxidizing bacteria active in wastewater treatment plants. Appl Environ Microbiol 67:5273–5284
Daims H, Purkhold U, Bjerrum L, Arnold E, Wilderer PA, Wagner M (2001b) Nitrification in sequencing biofilm batch reactors: lessons from molecular approaches. Water Sci Technol 43:9–18
Daims H, Ramsing NB, Schleifer K-H, Wagner M (2001c) Cultivation-independent, semiautomatic determination of absolute bacterial cell numbers in environmental samples by fluorescence in situ hybridization. Appl Environ Microbiol 67:5810–5818
Daims H, Lücker S, Wagner M (2006a) daime, a novel image analysis program for microbial ecology and biofilm research. Environ Microbiol 8:200–213
Daims H, Taylor MW, Wagner M (2006b) Wastewater treatment: a model system for microbial ecology. Trends Biotechnol 24:483–489
DeLong EF, Wickham GS, Pace NR (1989) Phylogenetic stains: ribosomal RNA based probes for the identification of single cells. Science 243:1360–1363
Egli K, Langer C, Siegrist HR, Zehnder AJ, Wagner M, van der Meer JR (2003) Community analysis of ammonia and nitrite oxidizers during start-up of nitritation reactors. Appl Environ Microbiol 69:3213–3222
Fernandez AS et al (2000) Flexible community structure correlates with stable community function in methanogenic bioreactor communities perturbed by glucose. Appl Environ Microbiol 66:4058–4067
Fiencke C, Bock E (2004) Genera-specific immunofluorescence labeling of ammonia oxidizers with polyclonal antibodies recognizing both subunits of the ammonia monooxygenase. Microb Ecol 47:374–384
Freese HM, Karsten U, Schumann R (2006) Bacterial abundance, activity, and viability in the eutrophic River Warnow, northeast Germany. Microb Ecol 51:117–127
Gans J, Wolinsky M, Dunbar J (2005) Computational improvements reveal great bacterial diversity and high metal toxicity in soil. Science 309:1387–1390
Glöckner FO et al (1996) An in situ hybridization protocol for detection and identification of planctonic bacteria. Syst Appl Microbiol 19:403–406
Harmsen HJ, Raangs GC, He T, Degener JE, Welling GW (2002) Extensive set of 16S rRNA-based probes for detection of bacteria in human feces. Appl Environ Microbiol 68:2982–2990
Hatzinger PB, Palmer P, Smith RL, Penarrieta CT, Yoshinari T (2003) Applicability of tetrazolium salts for the measurement of respiratory activity and viability of groundwater bacteria. J Microbiol Methods 52:47–58
Herndl GJ et al (2005) Contribution of Archaea to total prokaryotic production in the deep Atlantic Ocean. Appl Environ Microbiol 71:2303–2309
Heydorn A et al (2000) Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 146:2395–2407
Holland PM, Abramson RD, Watson R, Gelfand DH (1991) Detection of specific polymerase chain reaction product by utilizing the 5′->3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc Natl Acad Sci USA 88:7276–7280
Howard CV, Reed MG (2004) Unbiased stereology, 2nd edn. Taylor & Francis, Abingdon
Hug T, Gujer W, Siegrist H (2005) Rapid quantification of bacteria in activated sludge using fluorescence in situ hybridization and epifluorescence microscopy. Water Res 39:3837–3848
Jansen GJ, Wildeboer-Veloo AC, Tonk RH, Franks AH, Welling GW (1999) Development and validation of an automated, microscopy-based method for enumeration of groups of intestinal bacteria. J Microbiol Methods 37:215–221
Junge K, Eicken H, Deming JW (2004) Bacterial activity at −2 to −20 degrees C in arctic wintertime sea ice. Appl Environ Microbiol 70:550–557
Juretschko S, Loy A, Lehner A, Wagner M (2002) The microbial community composition of a nitrifying–denitrifying activated sludge from an industrial sewage treatment plant analyzed by the full-cycle rRNA approach. Syst Appl Microbiol 25:84–99
Kittler J, Illingworth J, Föglein J (1985) Threshold selection based on a simple image statistic. Comput Vis Graph Image Process 30:125–147
Kragelund C et al (2006) Ecophysiology of different filamentous Alphaproteobacteria in industrial wastewater treatment plants. Microbiology 152:3003–3012
Kuehn M, Hausner M, Bungartz H-J, Wagner M, Wilderer PA, Wuertz S (1998) Automated confocal laser scanning microscopy and semiautomated image processing for analysis of biofilms. Appl Environ Microbiol 64:4115–4127
Lebedeva EV, Alawi M, Fiencke C, Namsaraev B, Bock E, Spieck E (2005) Moderately thermophilic nitrifying bacteria from a hot spring of the Baikal rift zone. FEMS Microbiol Ecol 54:297–306
Licht TR, Tolker-Nielsen T, Holmstrom K, Krogfelt KA, Molin S (1999) Inhibition of Escherichia coli precursor-16S rRNA processing by mouse intestinal contents. Environ Microbiol 1:23–32
Liu J, Dazzo FB, Glagoleva O, Yu B, Jain AK (2001) CMEIAS: A computer-aided system for the image analysis of bacterial morphotypes in microbial communities. Microb Ecol 41:173–194
Lopez C, Pons MN, Morgenroth E (2005) Evaluation of microscopic techniques (epifluorescence microscopy, CLSM, TPE-LSM) as a basis for the quantitative image analysis of activated sludge. Water Res 39:456–468
Ludwig W, Schleifer KH (2000) How quantitative is quantitative PCR with respect to cell counts? Syst Appl Microbiol 23:556–562
Maixner F et al (2006) Nitrite concentration influences the population structure of Nitrospira-like bacteria. Environ Microbiol 8:1487–1495
Manser R, Muche K, Gujer W, Siegrist H (2005) A rapid method to quantify nitrifiers in activated sludge. Water Res 39:1585–1593
Manz W, Wagner M, Amann R, Schleifer K-H (1994) In situ characterization of the microbial consortia active in two wastewater treatment plants. Water Res 28:1715–1723
Manz W, Wendt-Potthoff K, Neu TR, Szewzyk U, Lawrence JR (1999) Phylogenetic composition, spatial structure, and dynamics of lotic bacterial biofilms investigated by fluorescent in situ hybridization and confocal laser scanning microscopy. Microb Ecol 37:225–237
Morgenroth E, Obermayer A, Arnold E, Brühl A, Wagner M, Wilderer PA (2000) Effect of long-term idle periods on the performance of sequencing batch reactors. Water Sci Technol 41:105–113
Nielsen JL, Aquino de Muro M, Nielsen PH (2003) Evaluation of the redox dye 5-cyano-2,3-tolyl-tetrazolium chloride for activity studies by simultaneous use of microautoradiography and fluorescence in situ hybridization. Appl Environ Microbiol 69:641–643
Nunan N et al (2001) Quantification of the in situ distribution of soil bacteria by large-scale imaging of thin sections of undisturbed soil. FEMS Microbiol Ecol 36:67–77
Oerther DB, Pernthaler J, Schramm A, Amann R, Raskin L (2000) Monitoring precursor 16S rRNAs of Acinetobacter spp. in activated sludge wastewater treatment systems. Appl Environ Microbiol 66:2154–2165
Pearce DA (2003) Bacterioplankton community structure in a maritime antarctic oligotrophic lake during a period of holomixis, as determined by denaturing gradient gel electrophoresis (DGGE) and fluorescence in situ hybridization (FISH). Microb Ecol 46:92–105
Pernthaler J, Alfreider A, Posch T, Andreatta S, Psenner R (1997) In situ classification and image cytometry of pelagic bacteria from a high mountain lake (Gossenköllesee, Austria). Appl Environ Microbiol 63:4778–4783
Pernthaler J, Pernthaler A, Amann R (2003) Automated enumeration of groups of marine picoplankton after fluorescence in situ hybridization. Appl Environ Microbiol 69:2631–2637
Poppert S, Essig A, Marre R, Wagner M, Horn M (2002) Detection and differentiation of chlamydiae by fluorescence in situ hybridization. Appl Environ Microbiol 68:4081–4089
Porter KG, Feig YS (1980) The use of DAPI for identifying and counting aquatic microflora. Limnol Oceanogr 25:943–948
Poulsen LK, Licht TR, Rang C, Krogfelt KA, Molin S (1995) Physiological state of Escherichia coli BJ4 growing in the large intestines of streptomycin-treated mice. J Bacteriol 177:5840–5845
Ramsing NB, Fossing H, Ferdelman TG, Andersen F, Thamdrup B (1996) Distribution of bacterial populations in a stratified fjord (Mariager Fjord, Denmark) quantified by in situ hybridization and related to chemical gradients in the water column. Appl Environ Microbiol 62:1391–1404
Reddy CK, Dazzo FB (2004) Computer-assisted segmentation of bacteria in color micrographs. Microsc Anal 18:5–7
Rizzuto R, Carrington W, Tuft RA (1998) Digital imaging microscopy of living cells. Trends Cell Biol 8:288–292
Schloter M, Assmus B, Hartmann A (1995) The use of immunological methods to detect and identify bacteria in the environment. Biotechnol Adv 13:75–90
Schmid M, Schmitz-Esser S, Jetten M, Wagner M (2001) 16S-23S rDNA intergenic spacer and 23S rDNA of anaerobic ammonium-oxidizing bacteria: implications for phylogeny and in situ detection. Environ Microbiol 3:450–459
Schmid M et al (2003) Characterization of activated sludge flocs by confocal laser scanning microscopy and image analysis. Water Res 37:2043–2052
Schmid M et al (2000) Molecular evidence for genus-level diversity of bacteria capable of catalyzing anaerobic ammonium oxidation. Syst Appl Microbiol 23:93–106
Schramm A, de Beer D, van den Heuvel JC, Ottengraf S, Amann R (1999) Microscale distribution of populations and activities of Nitrosospira and Nitrospira spp. along a macroscale gradient in a nitrifying bioreactor: quantification by in situ hybridization and the use of microsensors. Appl Environ Microbiol 65:3690–3696
Sekar R, Fuchs BM, Amann R, Pernthaler J (2004) Flow sorting of marine bacterioplankton after fluorescence in situ hybridization. Appl Environ Microbiol 70:6210–6219
Sieracki ME, Reichenbach SE, Webb KL (1989) Evaluation of automated threshold selection methods for accurately sizing microscopic fluorescent cells by image analysis. Appl Environ Microbiol 55:2762–2772
Sogin ML et al (2006) Microbial diversity in the deep sea and the underexplored “rare biosphere”. Proc Natl Acad Sci USA 103:12115–12120
Stoecker K et al (2006) Cohn’s Crenothrix is a filamentous methane oxidizer with an unusual methane monooxygenase. Proc Natl Acad Sci USA 103:2363–2367
Stubner S (2002) Enumeration of 16S rDNA of Desulfotomaculum lineage 1 in rice field soil by real-time PCR with SybrGreen detection. J Microbiol Methods 50:155–164
Thiel R, Blaut M (2005) An improved method for the automated enumeration of fluorescently labelled bacteria in human faeces. J Microbiol Methods 61:369–379
Thurnheer T, Gmür R, Giertsen E, Guggenheim B (2001) Automated fluorescent in situ hybridization for the specific detection and quantification of oral streptococci in dental plaque. J Microbiol Methods 44:39–47
Tolker-Nielsen T, Molin S (2000) Spatial organization of microbial biofilm communities. Microb Ecol 40:75–84
Ullrich S, Karrasch B, Hoppe H, Jeskulke K, Mehrens M (1996) Toxic effects on bacterial metabolism of the redox dye 5-cyano-2,3-ditolyl tetrazolium chloride. Appl Environ Microbiol 62:4587–4593
Van Vliet LJ, Boddeke FR, Sudar D, Young IT (1998) Image detectors for digital image microscopy. In: Wilkinson MHF, Schut F (eds) Digital image analysis of microbes, 1st edn. Wiley, Chichester
Viles CL, Sieracki ME (1992) Measurement of marine picoplankton cell size by using a cooled, charge-coupled device camera with image-analyzed fluorescence microscopy. Appl Environ Microbiol 58:584–592
Wagner M et al (1994) Development of an rRNA-targeted oligonucleotide probe specific for the genus Acinetobacter and its application for in situ monitoring in activated sludge. Appl Environ Microbiol 60:792–800
Wagner M, Rath G, Amann R, Koops H-P, Schleifer K-H (1995) In situ identification of ammonia-oxidizing bacteria. Syst Appl Microbiol 18:251–264
Wagner M, Horn M, Daims H (2003) Fluorescence in situ hybridization for the identification of prokaryotes. Curr Opin Microbiol 6:302–309
Wallner G, Erhart R, Amann R (1995) Flow cytometric analysis of activated sludge with rRNA-targeted probes. Appl Environ Microbiol 61:1859–1866
Wallner G, Fuchs B, Spring S, Beisker W, Amann R (1997) Flow sorting of microorganisms for molecular analysis. Appl Environ Microbiol 63:4223–4231
Wilkinson MHF (1998a) Automated and manual segmentation techniques in image analysis of microbes. In: Wilkinson MHF, Schut F (eds) Digital image analysis of microbes, 1st edn. Wiley, Chichester, pp 135–171
Wilkinson MHF (1998b) Optical systems for image analysed microscopy. In: Wilkinson MHF, Schut F (eds) Digital image analysis of microbes, 1st edn. Wiley, Chichester, pp 65–91
Xavier JB, White DC, Almeida JS (2003) Automated biofilm morphology quantification from confocal laser scanning microscopy imaging. Water Sci Technol 47:31–37
Yang X, Beyenal H, Harkin G, Lewandowski Z (2000) Quantifying biofilm structure using image analysis. J Microbiol Methods 39:109–119
Yang X, Beyenal H, Harkin G, Lewandowski Z (2001) Evaluation of biofilm image thresholding methods. Water Res 35:1149–1158
Acknowledgments
The development of image analysis approaches and software by our group has been financially supported by the Deutsche Forschungsgemeinschaft (DFG; grant Schl 120/14-1) and the Vienna Science and Technology Fund (WWTF; grant LS 216).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Daims, H., Wagner, M. Quantification of uncultured microorganisms by fluorescence microscopy and digital image analysis. Appl Microbiol Biotechnol 75, 237–248 (2007). https://doi.org/10.1007/s00253-007-0886-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-0886-z