Abstract
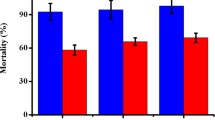
From the culture filtrate of the fungus Dactylellina varietas (syn. Dactylella varietas), an extracellular protease (designed Dv1) was purified by cation exchange and hydrophobic interaction chromatography. The purified protease showed a molecular mass of approximately 30 kDa and displayed an optimal activity at pH 8 and 60.5°C (more than 20 min). This protease could degrade a broad range of substrates including casein, gelatin, BSA (bovine serum albumin), and nematode cuticle. However, its proteolytic activity was highly sensitive to the serine protease inhibitor Phenylmethylphonylfuoride (1 mM), indicating that it belongs to the serine-type peptidase group. This protease could immobilize the free-living nematodes Panagrellus redivivus and Caenorhabditis elegans and hydrolyze the purified cuticle of P. redivivus, suggesting it may play a role in infection against nematodes. The encoding gene of Dv1 and its promoter sequence were cloned using degenerate primers and the DNA walking technology. Its open-reading frame contains 1,224 base pairs and without any intron. The deduced amino-acid sequence shared low identity to serine proteases from other nematode-trapping fungi. Our report identified a novel pathogenic protease from the nematode-trapping fungus D. varietas, and the three-dimensional structure of this protease was predicted using the Swiss-Prot method.






Similar content being viewed by others
References
Åhman J, Ek B, Rask L, Tunlid A (1996) Sequence analysis and regulation of a gene encoding a cuticle-degrading serine protease from the nematophagous fungus Arthrobotrys oligospora. Microbiology 142:1605–1616
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: signalP 3.0. J Mol Biol 340:783–795
Bonants PJ, Fitters PF, Thijs H, den Belder E, Waalwijk C, Henfling JW (1995) A basic serine protease from Paecilomyces lilacinus with biological activity against Meloidogyne hapla eggs. Microbiology 141:775–784
Bradford MM (1976) A rapid and sensitive for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cox GN, Kusch M, Edgar RS (1981) Cuticle of Caenorhabditis elegans: its isolation and partial characterization. J Cell Biol 90:7–17
Felsenstein J (1991) PHYLIP: phylogeny inference package, version 3.5. University of Washington, Seattle, WA
Guex N, Peitsch MC (1997) SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modelling. Electrophoresis 18:2714–2723
Huang XW, Zhao NH, Zhang KQ (2004) Extracellular enzymes serving as virulence factors in nematophagous fungi involved in infection of the host. Res Microbiol 155:811–816
Jansson HB, Nordbring-Hertz B (1988) Poinar GO Jr, Jansson H-B (eds) Infection mechanisms in the fungus-nematode system in diseases of nematodes, vol. II. CRC, Boca Raton, pp 59–72
Joshi L, St Leger RJ, Bidochka MJ (1995) Cloning of a cuticle-degrading protease from the entomopathogenic fungus, Beauveria bassiana. FEMS Microbiol Lett 125:211–217
Kraut J (1977) Serine proteases structure and mechanism of catalysis. Annu Rev Biochem 46:331–358
Laemmli EK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li Y, Jeewon R, Hyde KD, Mo MH and Zhang KQ (2006) Two new species of nematode-trapping fungi: relationships inferred from morphology, rDNA and protein gene sequence analyses. Mycol Res 110:790–800
Lopez-Llorca LV (1990) Purification and properties of extracellular protease produced by the nematophagous fungus Verticillium suchlasporium. Can J Microbiol 36:530–537
Maizels RM, Blaxter ML, Selkirk ME (1993) Forms and functions of nematode surfaces. Exp Parasitol 77:380–384
Morton CO, Hirsch PR, Peberdy JP, Kerry BR (2003) Cloning of and genetic variation in protease VCP1 from the nematophagous fungus Pochonia chlamydosporia. Mycol Res 107:38–46
Peitsch MC (1995) Protein modeling by e-mail. Bio/Technology 13:658–660
Schwede T, Kopp J, Guex N, Peitsch MC (2003) SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res 31:3381–3385
Segers R, Butt TM, Kerry BR, Peberdy JF (1994) The nematophagous fungus Verticillium chlamydosporium produces a chymoelastase-like protease which hydrolyses host nematode proteins in situ. Microbiology 140:2715–2723
Siddiqui ZA, Mahmood I (1996) Biological control of plant parasitic nematodes by fungi: a review. Bioresour Technol 58:229–239
Siezen RJ, Leunissen JAM (1997) Subtilases: the superfamily of subtilisin like serine protease. Protein Sci 6:501–523
Smith CA, Toogood HS, Baker HM, Daniel RM, Baker EN (1999) Calcium-mediated thermostability in the subtilisin superfamily: the crystal structure of Bacillus Ak.1 protease at 1.8 Å resolution. J Mol Biol 294:1027–1040
St Leger RJ, Frank DC, Roberts DW, Staples RC (1992) Molecular cloning and regulatory analysis of the cuticle-degrading protease structural gene from the entomopathogenic fungus Metarhizium anisopliae. Eur J Biochem 204:991–1001
Subramanian CV (1963) Dactylella, Monacrosporium and Dactylina. J Indian Bot Soc 42:291–300
Tunlid A, Jansson S (1991) Proteases and their involvement in the infection and immobilization of nematodes by the nematophagous fungus Arthrobotrys oligospora. Appl Environ Microbiol 57:2868–2872
Tunlid A, Rosen S, EK B, Rask L (1994) Purification and characterization of an extracellular serine protease from the nematode-trapping fungus Arthrobotrys oligospora. Microbiology 140:1687–1695
Wang M, Yang JK, Zhang KQ (2006) Characterization of an extracellular protease and its cDNA from the nematode-trapping fungus Monacrosporium microscaphoides. Can J Microbiol 52:130–139
Yang JK, Huang XW, Tian BY, Wang M, Niu QH, Zhang KQ (2005a) Isolation and characterization of a serine protease from the nematophagous fungus Lecanicillium psalliotae, displaying nematicidal activity. Biotechnol Lett 27:1123–1128
Yang JK, Huang XW, Tian BY, Sun H, Duan JX, Wu WP, Zhang KQ (2005b) Characterization of an extracellular serine protease gene from the nematophagous fungus Lecanicillium psalliotae. Biotechnol Lett 27:1329–1334
Zhang D, Yang Y, Castlebury LA, Cerniglia CE (1996) A method for the large scale isolation of high transformation efficiency fungal genomic DNA. FEMS Microbiol Lett 145:216–265
Zhao ML, Mo MH, Zhang KQ (2004) Characterization of a neutral serine protease and its full-length cDNA from the nematode-traping fungus Arthrobotrys oligospora. Mycologia 96:16–22
Acknowledgement
We thank Drs. Chenggang Zhou, Xiaowei Huang and Ms Wei Zhou for their help and advice in our studies and in preparing this manuscript. This work was funded by projects from the National Natural Science Foundation of China (approved nos. 30630003 and 30660107), by the Department of Science and Technology of Yunnan Province (approval Nos. 2004C0001Z and 2005NG05) and by Yunnan University (grant no. 2005Q008B).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jinkui Yang and Lianming Liang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yang, J., Liang, L., Zhang, Y. et al. Purification and cloning of a novel serine protease from the nematode-trapping fungus Dactylellina varietas and its potential roles in infection against nematodes. Appl Microbiol Biotechnol 75, 557–565 (2007). https://doi.org/10.1007/s00253-007-0839-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-0839-6